
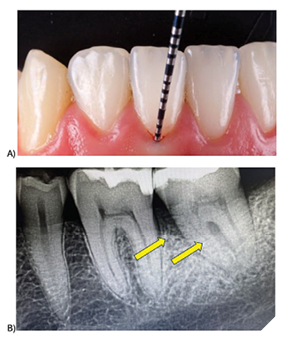
Periodontal disease remains one of the most prevalent yet preventable chronic conditions in adults, affecting nearly half of the adult population at some point in their lives. Yet for decades, dentists have relied on tools that largely tell the story of damage after it has already happened, through radiographs and periodontal probing, rather than giving an early warning before irreversible destruction occurs (Fig. 1). This diagnostic gap has meant that millions of patients worldwide progress to moderate or severe disease before interventions begin.
Fig. 1
From this frustration, PerioMonitor was born: a Canadian-made innovation designed to revolutionize the early detection and monitoring of gum disease, translating cutting-edge science directly to the dental chair. It is the result of a journey that combines scientific discovery, clinical need, and the dedication of a research team committed to improving patient outcomes.
Origins: The question that started it all
I have always been frustrated by the fact, that by the time we diagnose periodontal disease, bone loss is already there. We needed something that could see the invisible – the earliest inflammation.
As a periodontist with over 25 years of clinical experience, I see countless patients whose gum disease could have been prevented or slowed if only we and they had known earlier.
The concept emerged during my post-doctoral fellowship at Harvard Medical School, where I was studying white blood cells (neutrophils). I became fascinated by the fact that neutrophils, the body’s first-line immune defenders, were constantly present in the periodontium, even in health (Fig. 2). This suggested that subtle changes in their levels might signal early inflammation, long before visible damage.
Fig. 2A
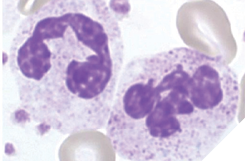
Fig. 2B
From idea to research: How the testing began
Step 1: Establishing the science
Back in my laboratory from 2003 to today (Fig. 3) at the University of Toronto Faculty of Dentistry, I began investigating whether oral neutrophil counts could be a reliable biomarker for gum inflammation.
Fig. 3

Our research plan involved multiple phases:
1. Recruitment – Patients across a spectrum of periodontal health were recruited from the patients attending the University of Toronto Faculty of Dentistry: those with healthy gums, patients with gingivitis, and those with moderate-to-severe periodontitis.
2. Sample collection – We developed a simple, non-invasive oral rinse method to collect cells from the oral cavity using a rapid oral rinse. The patient simply swished with a sterile saline solution for 30 seconds, which was then collected for testing.
3. Laboratory analysis – In the early stages, neutrophil counts were performed manually under a microscope by trained technicians in my laboratory (Fig. 4). As the project advanced, automated flow cytometry and staining methods were introduced for accuracy and reproducibility (Fig. 5).
Fig. 4A

Fig. 4B
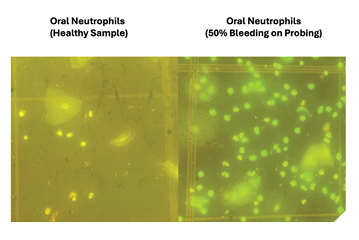
Fig. 5
4. Clinical correlation – Neutrophil levels were directly compared with gold-standard periodontal charting, probing depths, bleeding scores, and radiographs to confirm their diagnostic relevance.
Step 2: Early findings
The first pilot studies showed a clear pattern:
- Healthy gums demonstrated a stable baseline neutrophil count.
- Early gingivitis patients had a measurable spike – even with no clinical signs of inflammation and when pocket depths and radiographs appeared normal.
- Active periodontitis patients had dramatically elevated counts, correlating with bone loss.
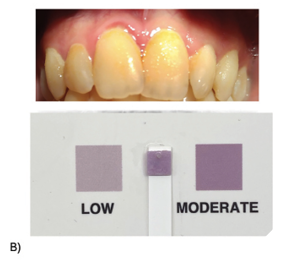
These findings confirmed that oral neutrophil monitoring could detect inflammation before clinical signs of destruction appeared, offering a truly preventive window of opportunity (Fig. 6).
Fig. 6A
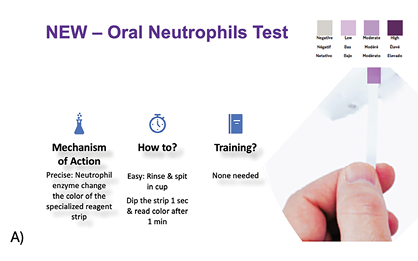
Fig. 6B
Step 3: Progression to real-world testing
Over several years, the method was refined into a true chairside test. The goals were clear: it had to be quick (<5 minutes), non-invasive, inexpensive, and easy for clinicians and patients to understand. Funding for much of this work came from the Ontario Centre for Innovation and Canadian Institutes of Health Research. We carried out pilot trials across Canada, testing:
- Ease of use for hygienists and dentists
- Repeatability across visits
- Patient comprehension and engagement with the results
Feedback from clinicians during these trials helped refine result displays, patient handouts, and integration with existing dental charting.
Clinical significance: Changing how we practice
The clinical relevance of PerioMonitor is profound. It provides a real-time inflammatory score that can be used in several key scenarios:
1. Early detection and prevention – In healthy patients, a sudden rise in neutrophil levels can highlight the need for preventive measures before structural damage occurs.
2. Monitoring high-risk patients – For diabetics, smokers, or immunocompromised patients, more frequent monitoring can help detect acute flare-ups.
3. Post-treatment evaluation – After scaling, root planing, or surgical intervention, PerioMonitor can confirm whether inflammation has resolved or if residual disease activity remains within weeks of the intervention. Current protocols require months of healing before probing can be used to assess results.
4. Longitudinal tracking – Over multiple visits, trends in neutrophil levels give a clear picture of disease control or progression, supporting evidence-based recall intervals.
5. Patient motivation – Showing patients their inflammation score engages them in their own care and often increases compliance with home hygiene recommendations.
From lab to chairside: The Canadian innovation story
Moving from a promising lab discovery to a Health Canada-approved clinical tool required collaboration. Oral Science, led by CEO Daniel Menard, managed manufacturing, marketing, and the rigorous regulatory process. Securing approval required large, multi-site clinical trials, comprehensive safety testing, and detailed manufacturing documentation.
This collaboration highlights the strength of the Canadian innovation ecosystem: academic research, industry expertise, and a healthcare system ready to adopt home made evidence-based solutions.
Future directions: AI and systemic health integration
The next chapter for PerioMonitor includes integrating artificial intelligence to predict a patient’s future periodontal risk trajectory and combining their neutrophil trends with other health data. The link between gum health and systemic diseases like diabetes and cardiovascular disease means that PerioMonitor will eventually become a valuable screening tool beyond dentistry.
PerioMonitor is more than a device – it is a bridge between laboratory science and everyday dental practice. It embodies the belief that with the right question, meticulous research, and the courage to innovate, we can dramatically improve patient outcomes. This is not only a story of periodontal health but also of Canadian scientists translating discovery into life-changing clinical tools. 
Oral Health welcomes this original article.
About the author

Dr. Michael Glogauer is a clinician-scientist and leader in the fields of neutrophil biology, innate immunity, oral microbiome, and periodontology. He is a Fellow of the Canadian Academy of Health Sciences. Dr. Glogauer is also a Professor at the Faculty of Dentistry, University of Toronto (since 2002), Head of Dentistry for University Health Network and Chief of Dental Oncology at Princess Margaret Cancer Centre.
