
Periodontal debridement, particularly scaling and root planing (SRP), remains the cornerstone of nonsurgical periodontal therapy. Its effectiveness in reducing probing depths and controlling inflammation has been well documented for decades.1-3 However, despite its central role, the procedure presents significant variability in complexity. Factors such as pocket depth, furcation involvement, root concavities, calculus tenacity, and patient-related challenges can greatly influence the time, effort, and skill required to achieve adequate outcomes.4,5
At present, no standardized system exists to estimate the difficulty of debridement before treatment begins. Clinicians typically rely on subjective judgment or prior experience to allocate treatment time, which can lead to underestimation in complex cases or inefficiency in simpler ones. This lack of calibration may contribute to inconsistent patient outcomes, clinician fatigue, and challenges in scheduling, particularly in practices with multiple providers.6
Recognizing this gap, our study sought to define and prioritize the clinical, anatomical, and behavioural variables that most significantly affect debridement difficulty. A Modified Delphi process was selected to harness expert consensus from experienced dental hygienists and periodontists. While the panel size was intentionally small to ensure depth of expertise, this pilot round represents a first step toward developing a reproducible framework for complexity assessment.
To translate these consensus-derived factors into a usable tool, we designed the Debridement Difficulty Index (DDI). This scoring framework applies weighted values to validated difficulty factors and integrates tooth count to estimate case-specific treatment time. Importantly, the tool was implemented into a GPT-assisted interface, not as an autonomous AI predictor but as a guided calculator that supports clinicians in case assessment and scheduling.
This paper presents the development of the DDI as a proof-of-concept tool. Specifically, we describe:
- The Modified Delphi process used to establish consensus on difficulty factors.
- The design of the DDI scoring system and GPT interface.
- Preliminary retrospective calibration against clinical records.
- Future directions for prospective, multi-center validation.
By addressing the absence of standardized assessment, the DDI aims to improve clinical calibration, optimize appointment planning, and support more consistent patient-centered care in periodontal therapy.
Study aim
This study aimed to develop a proof-of-concept assessment tool that systematically identifies and measures the factors influencing the difficulty of nonsurgical periodontal debridement. Using expert consensus from a Modified Delphi process, we sought to organize these factors into a structured framework, the DDI.
The tool is designed to connect difficulty factors with estimated treatment time by considering both case complexity and tooth count. In its current pilot form, the DDI offers a reliable method for guiding case planning, aiding calibration among dental hygienists and dentists, and identifying situations where referral to a specialist might be necessary. Although retrospective calibration was conducted to evaluate feasibility, future prospective validation across various practice settings will be needed to verify its reliability and broader applicability.
Materials and design
Overall study design
This study was designed as a proof-of-concept project to assess the feasibility of creating a standardized framework for estimating the difficulty of debridement. The process commenced with a comprehensive literature review aimed at identifying clinical, anatomical, and patient-related factors known to influence the complexity of nonsurgical periodontal therapy. This review revealed a significant gap: while many studies document treatment outcomes, few directly address the variables that increase or decrease the difficulty of performing debridement.1,7
To bridge this gap, insights were gathered from experienced dental hygienists and periodontists, whose daily practice offered valuable context for identifying and prioritizing difficulty factors. These combined data sources formed the basis of a Modified Delphi study, in which an expert panel participated in three iterative rounds of structured surveys. The aim was to refine, rank, and achieve consensus on the most critical factors influencing debridement complexity. Although the panel size (n=9) was deliberately kept small to ensure a high level of expertise, this phase was regarded as a pilot Delphi round, with plans to expand in future studies.
The consensus-based factors were then used to develop a prototype scoring system, the DDI. The DDI combines weighted values for validated factors with the number of teeth requiring treatment to produce a case-specific complexity score and an estimated treatment time. To enhance usability, the scoring system was integrated into a GPT-assisted interface. In this setup, GPT does not act as an autonomous AI predictor but as a guided calculator that asks structured questions, applies the scoring rules, and delivers a case classification with an estimated treatment duration.
Finally, preliminary feasibility testing was conducted through retrospective calibration. Treatment records from a periodontal specialty practice were reviewed, and estimated debridement times generated by the DDI were compared with actual recorded appointment times. This calibration step provided early insight into the tool’s potential alignment with clinical practice, while also highlighting the need for prospective, multi-center validation to confirm accuracy and generalizability.
Literature review
An extensive review of the literature revealed a critical gap: while numerous studies have examined the clinical outcomes of nonsurgical periodontal therapy, few have systematically investigated the factors that make debridement more or less difficult to perform. This omission presents a challenge for clinicians, who must routinely estimate case complexity and allocate treatment time without the support of a validated, predictive framework.
Biofilm and calculus as drivers of difficulty
Subgingival biofilm is the primary etiologic factor in periodontal disease. As it matures and mineralizes, calculus becomes firmly attached to root surfaces, creating niches for further bacterial accumulation and disease progression.1,8 The presence of tenacious or burnished subgingival calculus is consistently associated with increased instrumentation time and reduced treatment efficiency.9,10
Operator skill and experience
Treatment outcomes are also strongly influenced by operator experience. Studies demonstrate that clinicians with advanced training achieve greater efficacy in sites with probing depths >5 mm, where access is restricted and calculus is difficult to remove.6,7 Without standardization, individual skill differences may lead to variability in treatment planning, inconsistent scheduling, and uneven patient outcomes.
Anatomical factors
Tooth and root morphology greatly increase the difficulty of debridement. Posterior teeth are especially challenging, with furcation involvement, root concavities, and developmental grooves restricting access and requiring specialized instruments and tactile accuracy.4,11 Maxillary molar furcations have been particularly noted as areas where non-surgical treatment is often less effective, even when performed by experienced clinicians.11,12
Balancing efficacy with tooth preservation
Another clinical challenge is balancing thorough calculus removal with preservation of cementum. Aggressive instrumentation risks unnecessary hard tissue removal, potentially affecting long-term stability. Factors such as burnished calculus, patient sensitivity, and bleeding on probing further complicate decision-making during SRP.13,14
Role of technology and adjuncts
Technological aids can mitigate some of these challenges. The use of magnification, enhanced lighting and ultrasonic instrumentation has been shown to improve calculus detection and removal, particularly in difficult-to-access sites.15,16 Conversely, the absence of such adjuncts may contribute to incomplete debridement, increased operator fatigue, and variability in outcomes.
The need for standardization
Despite widespread recognition of these contributing factors, no standardized index currently exists to quantify their combined effect on treatment difficulty. Current practice relies heavily on clinician judgment, which is subjective, variable, and difficult to calibrate across providers. A structured, evidence-based framework that translates difficulty factors into predictive treatment time estimates could improve clinical consistency, support equitable scheduling, and enhance patient-centered care.
This study responds to that need by combining evidence from the literature with expert consensus through a Modified Delphi process, providing the foundation for development of the DDI.
Modified Delphi Study
Panel selection and rationale
Building on the literature review, all referenced factors related to debridement difficulty were compiled and supplemented with clinical insights from practicing dental hygienists and periodontists.2,17,18
A Modified Delphi study was conducted. The panel included nine experts: seven Registered Dental Hygienists and two periodontists. Eligibility required a minimum of ten years of professional experience in periodontics through clinical practice, education, or research. Five hygienists were actively working in periodontal specialty settings, while two were involved in dental hygiene education. The average number of years in practice was 24.1 (range: 10–38 years), with four panel members having more than 30 years of experience. Although the panel size was small, it was purposely designed as a pilot Delphi round to focus on depth of expertise rather than breadth. Future rounds with larger panels are planned to enhance generalizability.
Delphi Process
The study proceeded through three iterative survey rounds. In total, 54 potential factors were evaluated for their impact on the difficulty and time required to complete nonsurgical periodontal therapy.
Round 1
Round 1 Procedures
In the first round, panelists were asked to rate each factor using a 5-point scale:
- 1 = Not at all relevant (should be excluded)
- 5 = Extremely relevant (critical to evaluating case complexity)
The factors were grouped into five domains (Table 1).
Table 1: Categories of potential difficulty factors (n=54)
| Domain | Examples of Factors |
| Patient-Specific | Anxiety, gag reflex, communication barriers, positioning challenges, low pain threshold, xerostomia, smoker |
| Deposit-Related | Tenacious extrinsic stain, supragingival calculus (moderate/heavy), subgingival calculus (moderate/heavy), burnished calculus, furcation involvement, irregular maintenance |
| Gingival/Periodontal | Friable tissue, bleeding/purulence on probing, mucogingival defects, gingival hyperplasia, pocket depths (1–10+ mm), sensitivity (mild–requiring anesthesia) |
| Oral Cavity/Tooth | Limited access (e.g., small opening, TMD), muscle/tongue interference, obstructive tori, tooth positioning anomalies, root concavities, root proximity, tooth mobility, narrow arch form, orthodontic appliances, implants/bridges, subgingival or poorly contoured restorations |
| Clinician/Environment | Skill with root anatomy, availability of specialized instruments, use of magnification and lighting, ergonomic limitations of operatory |
Data analysis and consensus criteria
Three metrics were calculated for each factor:
- Mean score (average relevance)
- % rated 4 or 5 (proportion viewed as highly significant)
- Interquartile Range (IQR) (consistency of agreement)
Consensus thresholds for Round 1 were defined as:
- Level 3 (High consensus/critical): Mean ≥ 4, ≥70% rated 4–5, IQR ≤ 1
- Level 2 (Moderate consensus): Mean ≥ 3.5, ≥50% rated 4–5, IQR ≤ 1.5
- Level 1 (Low consensus): Did not meet thresholds above
Round 1 outcomes
Based on these criteria, the 54 factors were distributed into three tiers of difficulty:
- Level 3: Critical factors with strong agreement (e.g., deep pocketing, furcation involvement, burnished calculus, limited access).
- Level 2: Moderately important factors with some agreement (e.g., irregular maintenance, moderate calculus, ergonomic limitations).
- • Level 1: Low consensus factors with minimal or inconsistent impact (e.g., tremors, extrinsic stain).
These classifications formed the basis for refinement in subsequent Delphi rounds.
Round 2
Refinement of factors
After analyzing the results of Round 1, the factor list was refined (Table 2). Items with low agreement or redundancy were removed, while two new variables were added for further review based on panellist feedback.
Table 2: New factors added in Round 2
| New Factor | Rationale for Addition |
| Age or fragility of the patient | Recognized as influencing treatment tolerance and efficiency |
| Additional time required for documentation, re-probing, patient management, and oral hygiene instruction (OHI) | Identified as a frequent, practical factor affecting overall chair time |
Survey instructions
Panellists received a summary of Round 1 findings and re-rated each factor on the same 5-point scale (1 = Not at all, 5 = Extremely relevant). They were also encouraged to suggest reclassification of factors between Levels 1–3 based on clinical judgment.
Expanded analysis metrics
To enhance the precision of consensus measurement, Round 2 analysis included:
- Mean score (average relevance)
- % rated 4–5 (proportion identifying as highly significant)
- % rated 3–5 (proportion identifying as moderately to highly relevant)
- Interquartile Range (IQR) (agreement consistency across panelists)
Consensus thresholds: Round 2
- Level 3 (Critical): Mean ≥ 4.0, ≥70% rated 4–5, IQR ≤ 1.0
- Level 2 (Moderate): Mean 3.5–3.9, <70% rated 4–5 but ≥80% rated 3–5, IQR ≤ 1.5
- Level 1 (Low): Mean < 3.5, <50% rated 4–5, <80% rated 3–5, or IQR > 1.5
These expanded thresholds allowed the panel to distinguish between critical, moderately important, and less impactful factors with greater granularity.
Round 2 outcomes
Analysis of Round 2 ratings led to the classification of 20 factors into Level 1 and Level 2 categories, while Level 3 was reserved for the most consistently critical variables. This refinement process prioritized those factors most likely to influence debridement complexity while filtering out items that contributed only minimal or inconsistent difficulty.
During Round 2, one participant inadvertently submitted duplicate responses. The duplication was identified through time-stamped survey data and confirmed with the respondent; the duplicate was removed prior to analysis to preserve data integrity.
Round 3
Purpose of Round 3
The third Delphi round sought to finalize consensus on the most clinically significant factors contributing to debridement difficulty. Panelists were asked to:
- Rank their top ten most challenging factors (Fig. 1).
- Confirm or challenge the classification of factors into Levels 1–3.
- Provide input on two additional questions:
- Should the DDI incorporate tooth count when calculating complexity?
- Should referral to a periodontist be recommended if certain clinical or environmental conditions cannot be met?
Fig. 1
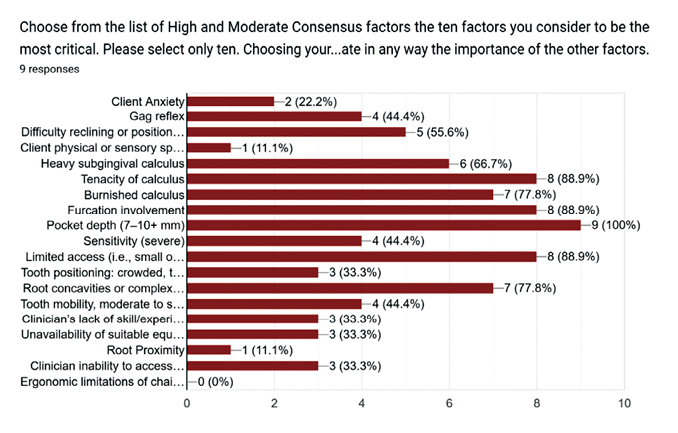
Open comment fields allowed participants to clarify reasoning and provide additional clinical nuance.
Consensus findings
Top ten most critical factors
When asked to rank their top ten most difficult factors, six items achieved high consensus (≥7 of 9 panelists) (Table 3):
- Pocket depth (7–10+ mm)
- Tenacity of calculus
- Furcation involvement
- Limited access (i.e., TMD, small opening)
- Burnished calculus
- Root concavities or complex morphology
Heavy subgingival calculus also emerged as near-critical with moderate consensus (6 of 9 panelists).
Table 3: Delphi Consensus results – Final classification of difficulty factors
| Domain | Examples of Factors | Consensus Agreement |
| Level 3 (Critical Difficulty Factors) | Gag reflex; difficulty reclining/positioning; patient anxiety; tenacity of calculus; heavy subgingival calculus; burnished calculus; pocket depth ≥7 mm; severe sensitivity; furcation involvement; limited access (e.g., TMD, restricted opening); root concavities; clinician inexperience with root anatomy; lack of suitable equipment; tooth mobility (Mobility II–III) | High consensus (7–9 of 9 panelists) |
| Level 2 (Moderately Important Factors) | Ergonomic limitations; root proximity; lack of magnification/lighting; patient cognitive/developmental challenges; moderate sensitivity; extrinsic stain; moderate calculus; xerostomia; smoker; bleeding on probing; narrow arch; large tori; OHI/documentation time; orthodontic appliances; implants/bridges; patient age/fragility; open contacts | High consensus (7 of 9 panelists) |
| Level 2 (Moderate Consensus Factors) | Tooth surface irregularities; irregular maintenance; purulence on probing; number of teeth present; wisdom teeth | Moderate consensus (6 of 9 panelists) |
| Level 1 (Low Consensus Factors) | Involuntary tremors/restlessness | Low consensus (5 of 9 panelists) |
Final polling results
Two final survey questions achieved unanimous agreement (9 of 9 panelists):
1. Should the DDI incorporate tooth count?
Yes. Panelists agreed that tooth count must be included to ensure individualized case planning that reflects both complexity and treatment extent.
2. Should consultation with a periodontal specialist be recommended when optimal treatment conditions cannot be achieved?
Yes. Consultation with a periodontal specialist should be considered when case complexity exceeds available resources or when optimal treatment outcomes may be compromised. Specific indicators for specialist consultation include:
- Case complexity exceeding available resources – When the combination of factors (deep pockets, furcations, complex anatomy) requires specialized expertise or extended treatment time beyond typical appointment scheduling
- Inadequate visualization or instrumentation – When magnification, specialized lighting, or specific instruments are necessary for case complexity are unavailable
- Facility limitations – When operatory design or equipment constraints prevent optimal patient positioning or clinician access
- Patient management challenges – When anxiety, medical complexity, or behavioural factors require specialized sedation protocols or modified treatment approaches
- Treatment duration concerns – When predicted treatment time exceeds practical appointment limits or patient tolerance
Tool development: DDI and GPT integration
Framework and scoring model
Following the Modified Delphi study, the validated factors were organized into a structured scoring framework: the DDI (Table 4). The goal was to create a reproducible tool that assigns weighted values to the most influential variables, incorporates tooth count, and outputs a projected treatment time.
Table 4: DDI factor weighting system
| Level | Examples of Factors | Weight |
| Level 3 (Critical) | Pocket depth ≥7 mm, furcation involvement, burnished calculus, limited access, root concavities | 2.0 |
| Level 2 (Moderate) | Root proximity, tooth positioning, ergonomic limitations, lack of magnification/lighting | 1.5 |
| Level 1 (Low) | Smoker, supragingival calculus, xerostomia, narrow arch, irregular maintenance | 1.0 |
| Referral (Non-negotiable) | Inadequate equipment, clinician inexperience with root anatomy | Flag Only |
The weighting system was based on Delphi consensus strength rather than arbitrary assignment:
- Level 3 (Critical factors): Highest consensus (mean ≥4, ≥70% rated 4–5) ➞ 2 points each.
- Level 2 (Moderately important factors): Moderate consensus (mean 3.5–3.9, <70% rated 4–5 but ≥80% rated 3–5) ➞ 1.5 points each.
- Level 1 (Mild factors): Lower consensus (mean <3.5 but ≥50% rated 3–5) ➞ 1 point each.
- Referral-triggering conditions: Flagged separately, not scored.
Calibration with clinical data
To provide initial context in real-world practice, the scoring system was calibrated retrospectively using clinical records from a periodontal specialty clinic. Over two decades of scheduling data showed that treatment times for full-mouth debridement ranged from roughly 45 minutes (simple cases) to 180 minutes (complex cases).
The DDI maps scores to five difficulty categories, each with corresponding time estimates (Table 5).
Table 5: DDI score-to-time calibration
| Category | Score Range | Estimated Treatment Time |
| Low | 0-20 | 45-60 min |
| Moderate | 21-40 | 61-100 min |
| High | 41-60 | 101-140 min |
| Complex | >60 | 141+ min |
This retrospective calibration demonstrated feasibility but does not replace the need for prospective, multi-center validation.
GPT integration
To improve usability, the DDI was integrated into a GPT-based clinical assistant. In this application, GPT functions as an interactive intake and calculator, guiding the clinician through a structured process:
- Asking for the number of teeth requiring debridement.
- Prompting selection of relevant difficulty factors across Levels 1–3.
- Automatically calculating the weighted DDI score and adjusting for tooth count.
- Classifying the case into a difficulty category and estimating treatment time.
- Flagging referral conditions if essential resources or clinician expertise are lacking.
This interface reduces subjectivity, ensures consistent application of the scoring system, and supports chairside decision-making without replacing clinical judgment.
Access and clinical usability
The DDI-GPT is available as a custom GPT within ChatGPT. Clinicians can access it by searching “Dental Debridement Difficulty Index” in the GPT library. The tool is currently free to use with a ChatGPT Plus account and provides step-by-step support in an interactive question-and-answer format. Early clinician feedback highlighted the intuitive workflow, time-saving calculations, and safeguard value of referral prompts as key benefits.
Results
The development and initial testing of the DDI support its feasibility as a practical clinical tool. The Modified Delphi process established consensus on the most influential factors contributing to debridement difficulty, which were stratified into weighted levels and integrated into a GPT-assisted scoring interface. Retrospective calibration demonstrated preliminary alignment between predicted and actual treatment times, and clinician feedback confirmed usability.
Delphi Consensus
Across three Delphi rounds, the expert panel refined an initial list of 54 factors into a validated framework. Six factors consistently emerged as critical predictors of difficulty, achieving high consensus (Table 6):
- Pocket depth ≥7 mm
- Tenacity of calculus
- Furcation involvement
- Limited access (e.g., TMD, restricted opening)
- Burnished calculus
- Root concavities or complex morphology
Heavy subgingival calculus was also identified as a near-critical factor (moderate consensus).
Table 6: Final consensus of difficulty factor categories from Delphi rounds
| Level | Consensus Strength | Examples of Factors |
| Level 3 (Critical) | High (≥7/9 panelists) | Pocket depth ≥7 mm, furcation involvement, burnished calculus, limited access, root concavities |
| Level 2 (Moderate) | High–moderate (≥6/9) | Root proximity, ergonomic limitations, lack of magnification |
| Level 1 (Low) | Moderate–low (<6/9) | Smoking, xerostomia, supragingival calculus, irregular maintenance |
| Referral (Non-negotiable) | Unanimous (9/9) | Clinician inexperience with root anatomy, lack of instruments |
To preserve comprehensiveness, moderate and mild factors were retained in the model, while referral-triggering conditions (e.g., lack of suitable instruments, inadequate magnification, clinician inexperience) were flagged separately to support patient safety.
Integration into GPT
The consensus-derived factors were programmed into a custom GPT-based assistant designed to function as an interactive calculator. The workflow included:
- Input of the number of teeth requiring debridement.
- Selection of relevant difficulty factors from Levels 1–3.
- Automatic calculation of a weighted DDI score, adjusted for tooth count.
- Classification of the case into one of four categories (Low ➞ Complex).
- Output of an estimated treatment time based on retrospective calibration.
- Flagging of referral conditions when non-negotiable thresholds were identified.
This integration transformed the DDI from a conceptual framework into an interactive, real-time decision support tool.
Retrospective calibration
To test alignment with real-world practice, the DDI was applied retrospectively to 25 treatment records from a periodontal specialty clinic. Each case was scored using documented factors, and predicted times were compared against actual appointment durations (Table 7).
Table 7: Predicted vs actual treatment times (Retrospective sample)
| Category | N | Mean Predicted | Mean Actual | Mean Difference | Mean Absolute |
| Complex (141–180 min) | 6 | 170.0 | 160.3 | 9.7 | 11.3 |
| High (101–140 min) | 7 | 125.1 | 112.9 | 12.3 | 12.3 |
| Moderate (61–100 min) | 9 | 81.1 | 79.3 | 1.8 | 6.2 |
| Low (≤60 min) | 3 | 53.3 | 52.3 | 1.0 | 11.0 |
Findings showed that the DDI estimates aligned closely with recorded treatment times:
- Simple cases (low scores) consistently aligned with 45–60 minutes.
- Moderate cases typically fell within 60–100 minutes.
- Complex cases (multiple Level 3 factors) exceeded 140 minutes.
Quantitative analysis confirmed this alignment. Across all cases, the mean predicted time was 111.4 minutes, the actual time lasted between 70 and 104.9 minutes, and the mean absolute error was 9.7 minutes. Correlation analysis showed a very strong relationship between predicted and actual times (r = 0.96). (Note: values represent feasibility testing; prospective validation with larger datasets is required.)
While some variability was observed in mid-range scores (Moderate–High categories), the calibration demonstrated that the DDI could reliably distinguish simple from complex cases and provide clinically meaningful time estimates.
Clinical usability
Clinicians who piloted the GPT interface reported:
- The stepwise questioning was an intuitive and streamlined assessment.
- Automated calculation reduced subjectivity in estimating difficulty.
- Referral flags were viewed as a valuable safeguard for patient-centred care and recognition of professional limitations.
Summary of results
The DDI has progressed from consensus-derived factors to a fully functional GPT-based assistant. Retrospective testing demonstrated strong feasibility, with predicted times aligning well with clinical records, particularly at the extremes of case complexity. Clinician feedback further supported its potential for improving scheduling accuracy, intra-operator calibration, and patient-centred decision-making.
Discussion
This study introduced the DDI, a proof-of-concept framework designed to estimate the complexity of nonsurgical periodontal debridement and guide treatment time. By combining expert consensus with a GPT-supported interface, the DDI demonstrates how predictive AI can be used not as a “black box,” but as a structured, calculator-style tool to enhance clinical calibration.
Interpretation of findings
The Modified Delphi process confirmed what clinicians often observe: factors such as deep pockets, furcation involvement, burnished calculus, limited access, and root concavities consistently make treatment more difficult. These findings support previous research that links probing depth, root shape, and calculus hardness with lower success rates for SRP.1,7
By stratifying these variables into weighted levels and including tooth count, the DDI offers a reproducible scoring system that surpasses subjective judgment. Retrospective calibration indicated that predicted time ranges generally aligned with clinical records, particularly in differentiating simple from complex cases. Although preliminary, these findings underscore the potential of combining expert consensus with a structured digital interface to promote more consistent treatment planning.
Clinical implications
The DDI offers a practical, chairside method for anticipating debridement difficulty. By guiding clinicians step-by-step through factor selection, the tool provides individualized time estimates, supports more accurate scheduling, enhances intra-team calibration and patient education. The inclusion of referral thresholds further strengthens patient safety by ensuring that complex cases are redirected to specialists when necessary.
Crucially, GPT in this context does not generate new clinical knowledge. Instead, it serves as a predictive interface, applying consensus-based rules in an interactive, calculator-like format. This pragmatic use of AI may reduce variability among clinicians while maintaining professional oversight in decision-making.
Application across practice settings
While calibration data were derived from a periodontal specialty clinic, the majority of nonsurgical periodontal therapy occurs in general practice settings. Practice environments vary significantly in available resources, case complexity, and appointment structure, which may influence treatment time estimates.
In diverse practice settings, the DDI serves multiple functions:
- Treatment Planning Support – helping practitioners determine optimal appointment structure and duration based on case complexity
- Resource Assessment – identifying when case factors exceed available practice resources or would benefit from collaborative care
The DDI framework recognizes that effective periodontal therapy depends on matching case complexity with appropriate resources and expertise. This collaborative approach ensures patients receive optimal care through the most suitable provider and setting, whether in general practice or specialty care.
Ethical and practical considerations
The introduction of AI into dentistry necessitates attention to data privacy, transparency, and clinician responsibility. The DDI does not replace clinical expertise but provides structured support that complements direct assessment and judgment. Clinicians remain accountable for care decisions, and the GPT interface functions solely as an assistive technology, not as an autonomous diagnostic system.
Limitations
Several limitations must be highlighted. The Delphi panel was small (n=9) and skewed toward dental hygienists, with limited geographic diversity. Consensus thresholds, while consistent with some Delphi precedents, lack psychometric validation. Calibration relied on retrospective data from a single specialty clinic, limiting external generalizability. Finally, the scoring system weights (2.0, 1.5, and 1.0 points) were clinically reasoned but not statistically validated. These limitations underscore the need for prospective, multi-center testing.
Future directions
Future research should focus on expanding the expert panel to 15-20 participants from general practice, specialty, and academic settings. This should be followed by prospective validation across multiple clinical sites, comparing real-time DDI scoring with actual treatment times. Key next steps include conducting inter-rater reliability testing and validating DDI performance across different practice types, along with developing calibration factors specific to each practice setting. Medium-term goals should aim to create simple visual aids for chairside use and explore integration with practice management software for automated scheduling suggestions. Long-term applications might involve studying how patient-reported outcomes relate to DDI scores and considering machine learning improvements, but only after establishing solid baseline validation across various clinical environments.
Conclusion
The DDI provides the first systematic framework for standardizing the assessment of periodontal debridement complexity. Through a structured Modified Delphi process, this study successfully identified and weighted six critical factors that consistently predict treatment difficulty: deep pockets (≥7mm), calculus tenacity, furcation involvement, limited access, burnished calculus, and complex root morphology.
The resulting proof-of-concept tool translates expert consensus into a reproducible scoring system accessible through a GPT-supported interface. Retrospective validation demonstrated strong correlation (r=0.96) between predicted and actual treatment times across 25 cases, with a mean absolute error of 9.7 minutes. While preliminary, these findings suggest the DDI can reliably differentiate case complexity and provide clinically meaningful time estimates.
For practicing clinicians, the DDI addresses a longstanding gap by providing structured guidance for treatment planning, scheduling optimization, and specialist consultation decisions. The tool’s referral thresholds ensure complex cases receive appropriate care while supporting calibration across providers.
This study demonstrates how expert clinical knowledge can be systematically captured, weighted, and delivered through accessible artificial intelligence interfaces. While prospective validation across diverse practice settings remains essential, the DDI establishes a foundation for evidence-based complexity assessment that could enhance consistency and efficiency in periodontal care delivery.
By combining traditional clinical expertise with structured predictive AI tools, this work marks an advancement toward more standardized, data-driven periodontal therapy planning.
Key takeaways
What this means for your practice
- Six factors consistently predict longer debridement times: deep pockets (≥7mm), tenacious calculus, furcations, limited access, burnished calculus, and complex root anatomy
- Multiple difficulty factors require extended scheduling: Consider longer appointments or multiple visits when several factors are present
- Clear consultation indicators: Consider specialist referral when specialized instruments, magnification, or complex root expertise are needed
- Better appointment planning: More accurate time predictions support optimal scheduling and resource allocation
- Improved patient experience: Realistic time estimates reduce waiting, rushed treatments, and appointment overruns
- Enhanced practice management: Structured assessment supports consistent treatment planning across providers
- Your clinical judgment validated: This study confirms what experienced clinicians already recognize about case complexity
- Apply these findings today: Consider how these validated difficulty factors can improve your current case assessment and scheduling practices.
Oral Health welcomes this original article.
AI attribution disclosure: The development of the DDI and portions of the manuscript were supported by the use of generative AI (ChatGPT, OpenAI, 2025). AI assistance was used for drafting, editing, and summarizing text, while all clinical content, data interpretation, and final revisions were performed and verified by the authors.
References
- Greenstein G. Periodontal response to mechanical non-surgical therapy: a review. J Periodontol. 1992;63(2):118-130.
- Drisko CH. Root instrumentation. Power-driven versus manual scalers, which one? Dent Clin North Am. 1998;42(2):229-244.
- Cobb CM, Sottosanti JS. A re-evaluation of scaling and root planing. J Periodontol. 2021;92(10):1370-1378.
- Ruhling A, Schlemme H, Konig J, Kocher T, Schwahn C, Plagmann HC. Learning root debridement with curettes and power-driven instruments. Part I: a training program to increase effectivity. J Clin Periodontol. 2002;29(7):622-629.
- Meissner G, Oehme B, Strackeljan J, Kocher T. Clinical subgingival calculus detection with a smart ultrasonic device: a pilot study. J Clin Periodontol. 2008;35(2):126-132.
- Preshaw PM, Holliday R, Law H, Heasman PA. Outcomes of non-surgical periodontal treatment by dental hygienists in training: impact of site- and patient-level factors. Int J Dent Hyg. 2013;11(4):273-279.
- Van der Weijden GAF, Dekkers GJ, Slot DE. Success of non-surgical periodontal therapy in adult periodontitis patients: A retrospective analysis. Int J Dent Hyg. 2019;17(4):309-317.
- Harrel SK, Cobb CM, Sheldon LN, Rethman MP, Sottosanti JS. Calculus as a Risk Factor for Periodontal Disease: Narrative Review on Treatment Indications When the Response to Scaling and Root Planing Is Inadequate. Dent J (Basel). 2022;10(10).
- Fleischer HC, Mellonig JT, Brayer WK, Gray JL, Barnett JD. Scaling and root planing efficacy in multirooted teeth. J Periodontol. 1989;60(7):402-409.
- Wylam JM, Mealey BL, Mills MP, Waldrop TC, Moskowicz DC. The clinical effectiveness of open versus closed scaling and root planing on multi-rooted teeth. J Periodontol. 1993;64(11):1023-1028.
- Al-Shammari KF, Kazor CE, Wang HL. Molar root anatomy and management of furcation defects. J Clin Periodontol. 2001;28(8):730-740.
- Zitzmann NU, Krastl G, Hecker H, Walter C, Weiger R. Endodontics or implants? A review of decisive criteria and guidelines for single tooth restorations and full arch reconstructions. Int Endod J. 2009;42(9):757-774.
- Miremadi SR, De Bruyn H, Steyaert H, Princen K, Cosyn J. A randomized controlled trial comparing surgical and non-surgical periodontal therapy: a 3-year clinical and cost-effectiveness analysis. J Clin Periodontol. 2015;42(8):748-755.
- Chung WC, Huang CF, Feng SW. Clinical Benefits of Minimally Invasive Non-Surgical Periodontal Therapy as an Alternative of Conventional Non-Surgical Periodontal Therapy-A Pilot Study. Int J Environ Res Public Health. 2022;19(12).
- Joda T, Waltimo T, Probst-Hensch N, Pauli-Magnus C, Zitzmann NU. Health Data in Dentistry: An Attempt to Master the Digital Challenge. Public Health Genomics. 2019;22(1-2):1-7.
- Cyris M, Festerling J, Kahl M, Springer C, Dorfer CE, Graetz C. Guided biofilm therapy versus conventional protocol-clinical outcomes in non-surgical periodontal therapy. BMC Oral Health. 2024;24(1):1105.
- Rabbani GM, Ash MM, Jr., Caffesse RG. The effectiveness of subgingival scaling and root planing in calculus removal. J Periodontol. 1981;52(3):119-123.
- Tomasi C, Leyland AH, Wennstrom JL. Factors influencing the outcome of non-surgical periodontal treatment: a multilevel approach. J Clin Periodontol. 2007;34(8):682-690.
About the authors

Colleen C. Porter is a Registered Dental Hygienist working in a periodontal specialty office and serving as part-time Professor at Niagara College Canada. She earned her Certificate in Dental Hygiene from Niagara College and her Bachelor of Education in Adult Education from Brock University.

Catherine P. Hamm is a Registered Dental Hygienist working in a periodontal specialty practice. She graduated from Niagara College and earned Certificates in Local Anesthetic Techniques from University of Manitoba and Saskatchewan Polytechnic. Passionate about advancing oral health and giving back to the community, Catherine is preparing to volunteer with the Build Your Smile Dental Foundation in Uganda in January 2026.

Dr. Peter Fritz is a pioneering periodontist and implant surgeon who blends clinical excellence with a deep understanding of the legal and ethical implications of emerging digital technologies in dentistry. He holds adjunct appointments at the Eastman Institute for Oral Health (University of Rochester), McMaster University, and Brock University, and directs a summer residency in AI-Enhanced Periodontics for dental students at his Fonthill, Ontario clinic..
