Abstract
Background: Peri-implantitis is a serious complication for both patients and clinicians. Affected implants typically show extensive bone loss in radiographs, are difficult to manage and usually lead to implant loss leaving significant alveolar bone defects.
Methods: In 2009 the patient reported here received two endosseous implants to replace his mandibular left second bicuspid and first molar teeth. After restoration by his general dentist, he did not appear again until 2014 at which time the first molar implant showed what appeared to be advanced bone loss extending almost to the implant apex. As the implant was asymptomatic, the patient declined treatment, preferring to wait until the implant became mobile but agreed to return in one year for follow-up. No documented treatment was provided by his restorative dentist.
Results: When the patient returned one year later, a radiograph showed a marked reduction in the peri-implant radiolucency which had resolved completely after a further year.
Conclusions: Peri-implant radiolucencies are not always indicative of impending implant failure, meaning that it is wise to investigate existing risk factors such as occlusal trauma and their resolution before settling on a diagnosis of peri-implantitis.
Dental implant replacement of missing or condemned teeth has become common treatment in both specialty and general dental practices. Planned and done properly, outcomes are generally successful in restoring function and fulfilling patient expectations.1, 2 Over the long-term however, implants can result in complications and failures as they are not as easy to maintain as teeth,3 even in the same mouths. The most serious complication of integrated implants is peri-implantitis, which usually presents as bleeding and/or suppuration on gentle probing, probing pocket depths of ≥ 6 mm, and crestal bone loss ≥ 3mm.4 It can be linked to a variety of factors including improper patient selection and/or operator errors in surgical and/or prosthetic management. Once peri-implantitis is established, it becomes a site of chronic inflammation and infection with progressive loss of supporting bone. Unfortunately, it is very difficult, indeed often impossible to treat successfully5, 6 in the end requiring implant removal and extensive, costly remedial treatment. This makes the case to be presented here interesting but difficult to explain since an apparent advanced peri-implantitis lesion apparently resolved spontaneously without treatment.
Clinical case
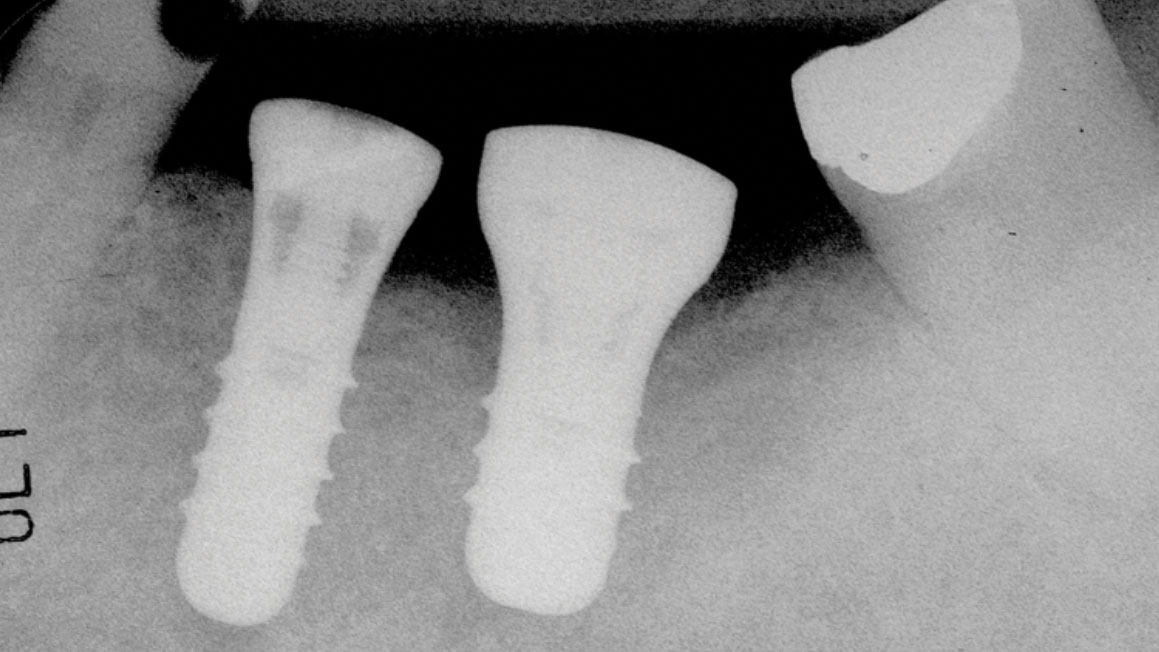
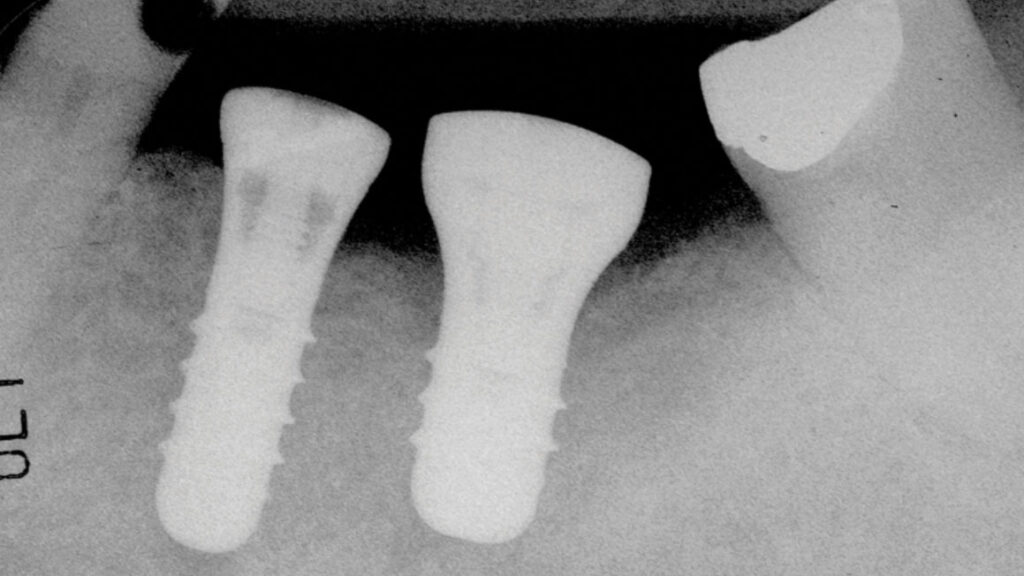
The patient was a 59-year-old gentleman who was a non-smoker with no medical issues contraindicating dental implant placement. In July 2009, he attended the office of an oral and maxillofacial surgeon (RL) and received two standard Straumann tissue level implants to replace his mandibular left second bicuspid and first molar teeth. The opposing arch had a fixed, tooth-supported bridge replacing the first molar. The bicuspid implant was a regular diameter, 10mm-long one, while the molar implant was 4.8 by 8mm-long (Fig. 1). The patient subsequently returned to his general practitioner for restoration with two separate crowns in late 2009 and wasn’t seen again until July 2014, at which time the molar implant showed an apparent, extensive peri-implantitis lesion extending almost to the implant apex. No implant mobility was detected but the patient was advised to have the implant removed. However, since it was not causing him any discomfort, he opted to let things progress until the implant became mobile. He was advised to return if mobility and/or pain developed, but in any event to return in one year’s time for follow-up. The patient did return as requested on May 15, 2015. A new radiograph was taken at that time and showed that the lesion appeared to be healing without any documented remedial treatment by his dentist (Fig. 3). A further follow-up in November 2016 showed even more bony healing in a radiograph (Fig. 4A) and clinically healthy soft tissues (Fig. 4B) with minimal probing depths and without associated bleeding upon probing.
Fig. 1
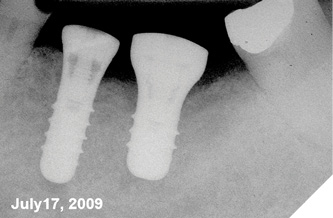
Fig. 2
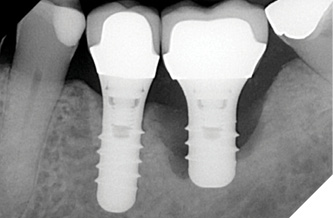
Fig. 3
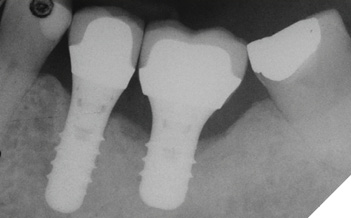
Fig. 4A
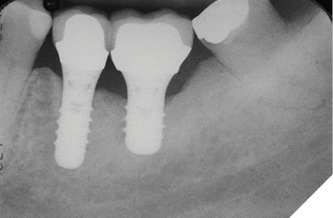
Fig. 4B
Discussion
Numerous predisposing factors have been implicated in the onset of peri-implantitis. While peri-implant mucositis often can be managed with things as simple as improved homecare, peri-implantitis is far more serious, often requiring implant removal. How is it possible then that the apparent peri-implantitis affecting the implant in the present report resolved without professional intervention?
Possible risk factors7 for peri-implantitis include uncontrolled, co-existing periodontitis, inadequate peri-implant keratinized soft tissues, suboptimal homecare with preceding peri-implant mucositis, inadequate profession maintenance, poor prosthesis design, bruxism, and/or deleterious patient habits such as smoking. Related gene health issues can include poor glycaemic control, poorly controlled diabetes or obesity with its generalized and constant, body-wide, hyper-inflammatory state.8 Most recently, biocorrosion of titanium with local release of titanium nano-particles also has been suggested as an initiator.9 Evidence of occlusal overload should always be considered as the presence of wear facets on implant-supported prostheses has been linked to a 2.4 times increase in peri-implantitis prevalence.10 Merin11 reported a case of a bruxer who after 14 months in function with a Straumann tissue level mandibular molar implant showed significant, funnel-shaped, crestal bone loss. There was no bleeding on probing, but heavy occlusal contacts were detected in centric occlusion and lateral excursions on both the working and non-working inclines. Occlusal adjustment was performed until only very light centric contact remained and the patient was urged to wear her nightguard. When she returned for follow-up at five months, the apparent bone loss appeared to have resolved.
Passanezi et al.12 presented a similar case in a 62-year-old, non-smoking, systemically healthy woman who had received a fixed, 3-unit prosthesis supported by two standard threaded implants in her left posterior mandible. After treatment completion, the patient did not return for 6 years. At that visit, she reported having undergone psychological treatment for 4 years and had been aware of bruxing and peri-implant bleeding. Examination revealed mean peri-implant probing depths of 4.75 millimeters (possibly pseudo-pocketing due to marginal tissue inflammatory enlargement/mucositis) at both implants, which was approximately 2.0 mm deeper than baseline at restoration, while intraoral radiographs showed apparent cratering bone loss extending to half the implant lengths. The prosthesis was removed and components cleaned with 0.2% chlorhexidine before replacing them. Subgingival debridement was attempted with plastic curettes, but no antibiotic prescribed. In response, the peri-implant inflammation resolved by 15 days and one month later, a flat-plane, acrylic occlusal bite guard was inserted. Five months later, the patient reported that she was no longer bruxing and at 10 months, the peri-implant radiolucencies were decreasing starting apically, and probing depths had decreased. By 4 years, there were no radiolucent lesions remaining.
A common finding with the peri-implantitis cases presented by Merin and by Passanezi et al. and another by Tawil13 was that occlusal overload seemed to have played a role given that occlusal adjustments and bruxism management resolved the peri-implant radiolucencies. Occlusal overload is commonly thought not to play a role in peri-implantitis or periodontitis unless local inflammation exists.14 15, 16 Peri-implantitis is generally preceded by mucositis,17, 18 an inflammatory condition of peri-implant soft tissues initiated by microbial biofilm. If mucositis becomes chronic, peri-implantitis may ensue with progressive bone loss exacerbated by existing occlusal trauma.
Wolff’s time-honoured experimental work determined a direct correlation between mechanical loading of bone and its remodeling.19 Bone microstructure including density and mineral content will adapt according to mechanical stresses received.20 If stresses remain within physiologic limits, bone will react by increasing in density. If on the other hand, stresses increase beyond these limits, damage to bone, such as micro-cracks, can occur leading to resorption by bone cells including osteocytes. Co-existing chronic inflammation will lead to high expression of RANKL and increased osteolytic activity,21-23 including bone demineralization24 and proteolysis of peri-lacunar and peri-canalicular organic bone matrix, a process termed “osteocytic osteolysis”.25 This is more or less what happens when cementless total hip replacements loosen aseptically due to chronic, low level inflammation caused by release of wear-related metal particles from the implant surface. This condition is referred to as “peri-prosthetic osteolysis”26 or “particle osteolysis” and can result in implant failure. Particle-stimulated macrophages, or in the case of dental implants, those reacting to bacterial plaque organisms, will release cytokines, chemokines, and other pro-inflammatory substances that induce bone resorption and suppress bone formation27 and activate the immune system.25
Unfortunately, whether or not the implant in the patient reported here was initially in hyper-occlusion was not recorded by the patient’s restorative dentist. Nevertheless, the initial peri-implant radiolucency seen likely was not yet an established peri-implantitis lesion but rather merely localized demineralization, which may have resolved in response to altered patient behavior after he was counselled that the implant was likely failing.
Conclusions
Peri-implant radiolucencies suggestive of major bone loss are not necessarily a sign of peri-implantitis. If the affected implant is otherwise asymptomatic, before making a definitive diagnosis it may be wise to explore and manage possible risk factors for bone resorption such as occlusal overload. 
Oral Health welcomes this original article.
References
- Howe MS, Keys W, Richards D. Long-term (10-year) dental implant survival: A systematic review and sensitivity meta-analysis. J Dent. 2019;84:9-21.
- Torres-Alemany A, Fernandez-Estevan L, Agustin-Panadero R, Montiel-Company JM, Labaig-Rueda C, Manes-Ferrer JF. Clinical Behavior of Short Dental Implants: Systematic Review and Meta-Analysis. J Clin Med. 2020;9(10).
- Sartoretto SC, Shibli JA, Javid K, Cotrim K, Canabarro A, Louro RS, et al. Comparing the Long-Term Success Rates of Tooth Preservation and Dental Implants: A Critical Review. J Funct Biomater. 2023;14(3).
- Berglundh T, Armitage G, Araujo MG, Avila-Ortiz G, Blanco J, Camargo PM, et al. Peri-implant diseases and conditions: Consensus report of workgroup 4 of the 2017 World Workshop on the Classification of Periodontal and Peri-Implant Diseases and Conditions. J Periodontol. 2018;89 Suppl 1:S313-S8.
- Khoury F, Keeve PL, Ramanauskaite A, Schwarz F, Koo KT, Sculean A, et al. Surgical treatment of peri-implantitis – Consensus report of working group 4. Int Dent J. 2019;69 Suppl 2(Suppl 2):18-22.
- Romandini M, Bougas K, Alibegovic L, Hosseini S, Carcuac O, Berglundh T, et al. Long-term outcomes and prognostic factors of surgical treatment of peri-implantitis – A retrospective study. Clin Oral Implants Res. 2024;35(3):321-9.
- Hashim DC, N. A Comprehensive Review of Peri-implantitis Risk Factors
- Current Oral Health Reports 2020;7:262-73.
- Rohm TV, Meier DT, Olefsky JM, Donath MY. Inflammation in obesity, diabetes, and related disorders. Immunity. 2022;55(1):31-55.
- Kandaswamy E, Sakulpaptong W, Guo X, Ni A, Powell HM, Tatakis DN, et al. Titanium as a Possible Modifier of Inflammation Around Dental Implants. Int J Oral Maxillofac Implants. 2022;37(2):381-90.
- Dalago HR, Schuldt Filho G, Rodrigues MA, Renvert S, Bianchini MA. Risk indicators for Peri-implantitis. A cross-sectional study with 916 implants. Clin Oral Implants Res. 2017;28(2):144-50.
- Merin RL. Repair of peri-implant bone loss after occlusal adjustment: a case report. J Am Dent Assoc. 2014;145(10):1058-62.
- Passanezi E, Sant’Ana AC, Damante CA. Occlusal trauma and mucositis or peri-implantitis? J Am Dent Assoc. 2017;148(2):106-12.
- Tawil G. Peri-implant bone loss caused by occlusal overload: repair of the peri-implant defect following correction of the traumatic occlusion. A case report. Int J Oral Maxillofac Implants. 2008;23(1):153-7.
- Isidor F. Loss of osseointegration caused by occlusal load of oral implants. A clinical and radiographic study in monkeys. Clin Oral Implants Res. 1996;7(2):143-52.
- Polson AM. Interrelationship of inflammation and tooth mobility (trauma) in pathogenesis of periodontal disease. J Clin Periodontol. 1980;7(5):351-60.
- Fu JH, Hsu YT, Wang HL. Identifying occlusal overload and how to deal with it to avoid marginal bone loss around implants. European journal of oral implantology. 2012;5 Suppl:S91-103.
- Jepsen S, Berglundh T, Genco R, Aass AM, Demirel K, Derks J, et al. Primary prevention of peri-implantitis: managing peri-implant mucositis. J Clin Periodontol. 2015;42 Suppl 16:S152-7.
- Berglundh T, Armitage G, Araujo MG, Avila-Ortiz G, Blanco J, Camargo PM, et al. Peri-implant diseases and conditions: Consensus report of workgroup 4 of the 2017 World Workshop on the Classification of Periodontal and Peri-Implant Diseases and Conditions. J Clin Periodontol. 2018;45 Suppl 20:S286-S91.
- Cowin SC. Wolff’s law of trabecular architecture at remodeling equilibrium. J Biomech Eng. 1986;108(1):83-8.
- Naveau A, Shinmyouzu K, Moore C, Avivi-Arber L, Jokerst J, Koka S. Etiology and Measurement of Peri-Implant Crestal Bone Loss (CBL). J Clin Med. 2019;8(2).
- Luo X, Wan Q, Cheng L, Xu R. Mechanisms of bone remodeling and therapeutic strategies in chronic apical periodontitis. Frontiers in cellular and infection microbiology. 2022;12:908859.
- Belanger LF. Osteocytic osteolysis. Calcif Tissue Res. 1969;4(1):1-12.
- Mbalaviele G, Novack DV, Schett G, Teitelbaum SL. Inflammatory osteolysis: a conspiracy against bone. J Clin Invest. 2017;127(6):2030-9.
- Bohatirchuk F. Calciolysis as the initial stage of bone resorption. A stain historadiographic study. Am J Med. 1966;41(5):836-46.
- Tsourdi E, Jahn K, Rauner M, Busse B, Bonewald LF. Physiological and pathological osteocytic osteolysis. J Musculoskelet Neuronal Interact. 2018;18(3):292-303.
- Engh CA, Jr., Sychterz CJ, Young AM, Pollock DC, Toomey SD, Engh CA, Sr. Interobserver and intraobserver variability in radiographic assessment of osteolysis. J Arthroplasty. 2002;17(6):752-9.
- Goodman SB, Gallo J. Periprosthetic Osteolysis: Mechanisms, Prevention and Treatment. J Clin Med. 2019;8(12).
About the authors

Douglas A Deporter, PhD, Professor, Discipline of Periodontology.

Robin Listrom, Instructor, Disciplines of Oral & Maxillofacial Surgery, Periodontics and Prosthodontics, Faculty of Dentistry, University of Toronto.