A retrospective case-control pilot study
Abstract
The use of transnasal sphenopalatine ganglion (SPG) block has expanded in the field of pain management due to its minimally invasive delivery and potential benefit for craniofacial pain. In a case-control pilot evaluation, patients receiving SPG block via the SphenoCath catheter system demonstrated numerically larger reductions in temporomandibular disorder (TMD) pain compared to those treated with a night guard, although both groups reported meaningful improvement following treatment. Patient-reported improvement and tolerability were high, with no serious adverse events noted. These preliminary findings are consistent with previous clinical use of SPG-targeted approaches in other craniofacial pain conditions. Larger, adequately powered clinical trials are warranted to confirm efficacy and guide standardized clinical protocols.
Literature review
Temporomandibular disorders (TMDs) are among the most common causes of chronic orofacial pain globally, with prevalence estimates ranging from 5% to 12% depending on the diagnostic criteria and population studied.2 Patients frequently experience functional limitations of the jaw, masticatory impairment, and psychosocial distress, which contribute to reduced quality of life.3,4 Heightened clinical awareness and efforts to develop validated patient-reported outcome measures reflect the growing burden of TMD on individuals and health systems.4
TMD pathophysiology is multifactorial, involving complex interactions among biological, psychological, and social determinants.5,6 Centralized pain mechanisms play a significant role in symptom persistence, as patients with chronic TMD show patterns of altered nociceptive processing and heightened pain sensitivity.7 Emotional distress, parafunctional behaviors such as bruxism, and sleep disturbances further exacerbate symptom severity.3,7 These multidimensional contributors lead to diagnostic heterogeneity and variation in treatment response across patient subgroups.5,8
Conservative treatments, including oral appliances, NSAIDs, physiotherapy, and behavioural strategies, are typically first-line in clinical practice.5,9 Although often beneficial, a substantial proportion of patients experience inadequate or incomplete symptom relief despite prolonged management.6,8 As a result, there is increasing interest in minimally invasive interventions targeting neuromodulation and craniofacial pain pathways.6,8
The sphenopalatine ganglion (SPG) is a parasympathetic ganglion located in the pterygopalatine fossa and is closely associated with sensory branches of the trigeminal nerve.10,11 Its role in cranial autonomic modulation and nociceptive signal transmission has been widely studied in facial pain and headache conditions.11,12 Shared innervation pathways between the trigeminal nerve and cranial parasympathetic fibers in the sphenopalatine ganglion suggest that modulating SPG activity may influence nociceptive processing in TMD-related pain.11,12 SPG-mediated processes involving vasodilation and neurogenic inflammation have been implicated in migraine and other facial pain syndromes.12
Transnasal SPG block has emerged as a promising clinical intervention for primary headache disorders.13,14 Randomized controlled trials using the Tx360 device have demonstrated rapid analgesic onset and sustained pain reduction in chronic migraine populations.15,16 Retrospective evaluation of the SphenoCath device in acute migraine also showed that most patients achieved meaningful pain relief within 15 minutes, with mild and transient adverse effects.17 Clinical adoption continues to increase among headache specialists, although consensus on standardized protocols is still evolving.18 Radiofrequency-based SPG interventions further support the ganglion as a clinically relevant pain management target.19-21
Multiple SPG block techniques exist, including topical, drip, and catheter-guided intranasal approaches.10,22 Catheter-guided delivery is designed to optimize anesthetic distribution within the pterygopalatine fossa.10 Digital infrared thermal imaging (DITI) has shown utility as a non-invasive indicator of successful SPG block, with increased facial temperature reflecting autonomic block.23,24 Across studies, SPG block appears safe, well-tolerated, and acceptable to patients.17,18
Despite its success in headache medicine, the therapeutic potential of SPG block for TMD-related pain has not been rigorously evaluated. Given shared trigeminal pathways, the presence of autonomic features in TMD, and high rates of refractory pain, SPG block may represent a valuable addition to multidisciplinary management for selected patients.8,9, 12 Pilot studies are essential to address this evidence gap and provide feasibility data to inform future randomized controlled trials, including appropriate sample size estimation.25,26
Case-control pilot study
Eight patients with chronic TMD-related pain were included (6 females, 2 males; mean age: 45.3 years; age range: 33–54 years). Baseline demographic and clinical characteristics for both groups are summarized in Table 1. All patients met diagnostic criteria for myofascial or arthrogenous TMD, with persistent pain for longer than three months and documented incomplete response to conservative therapies, including oral appliances and NSAIDs.
Table 1: Baseline demographic and clinical characteristics of patients in the SphenoCath and night guard groups.
| Group | Mean Age (years) | Sex (F/M) | Mean Duration (years) | Diagnosis: Myofascial n (%) |
| SphenoCath (n=4) | 47.0 | 3/1 | 5.5 | 3 (75%) |
| Night Guard (n=4) | 43.5 | 3/1 | 14.8 | 3 (75%) |
Eligibility was confirmed through standardized chart review. Inclusion required a DC/TMD diagnosis specifically identifying myofascial involvement, along with ICOP-defined chronic pain of at least 3 months’ duration. Pain characteristics including primary location within the masticatory muscles or temporomandibular joint, frequency, and identifiable triggers were documented at baseline to ensure diagnostic consistency across patients. All participants were adults (≥18 years) and able to provide informed consent.
Patients were excluded if they had any contraindications to SPG block, including allergy to local anesthetics, recent nasal or sinus infection, coagulopathy, or a history of facial trauma or surgery that could alter intranasal anatomy. Additional exclusions included pregnancy, inability to consent or read English, and diagnoses limited strictly to arthrogenous or other non-myofascial TMD presentations, as these were outside the scope of the pilot intervention.
Four patients received transnasal SPG block using the SphenoCath device, while four matched controls continued a standardized conservative management protocol consisting of customized night guard wear and routine follow-up. To preserve comparability between groups, no additional interventions were provided to control patients during the observation period. Local anesthetic (2% lidocaine) was administered intranasally for SPG block. Pain levels were measured pre- and post-intervention using a validated Visual Analog Scale (VAS) ranging from 0 (no pain) to 10 (worst pain).
SPG-treated patients demonstrated greater average pain reduction compared to controls, with most achieving a post-treatment VAS score ≤ 1. No serious adverse effects were reported.
Procedure
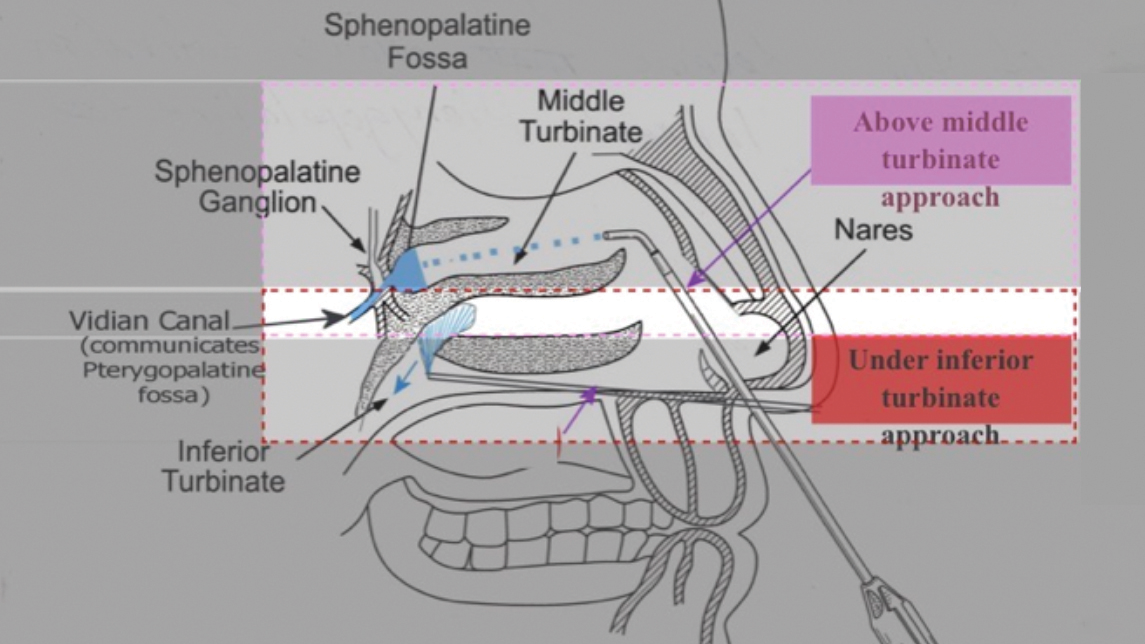
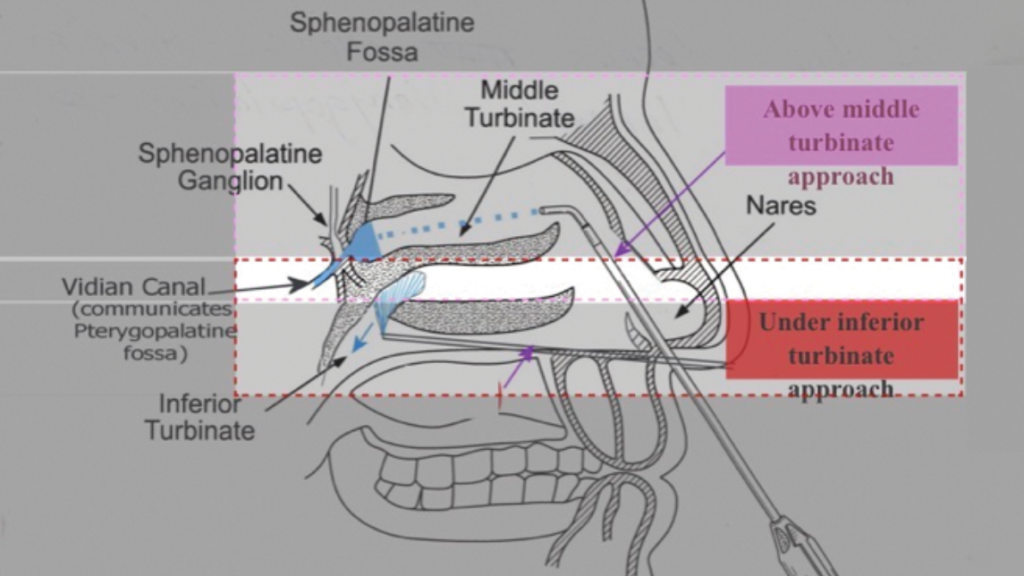
Patients in the treatment group received transnasal SPG blocks using the SphenoCath catheter (Part Number: DT1001GP-3). A topical application of 2% viscous lidocaine was first administered intranasally to anesthetize the nasal passage. The SphenoCath catheter was then advanced to the posterior nasal cavity to deliver lidocaine hydrochloride 2% (100 mg/5 mL; 20 mg/mL; DIN: 02421992) directly to the SPG. The transnasal placement and delivery technique of the SphenoCath catheter are illustrated in Figure 1.1
Fig. 1
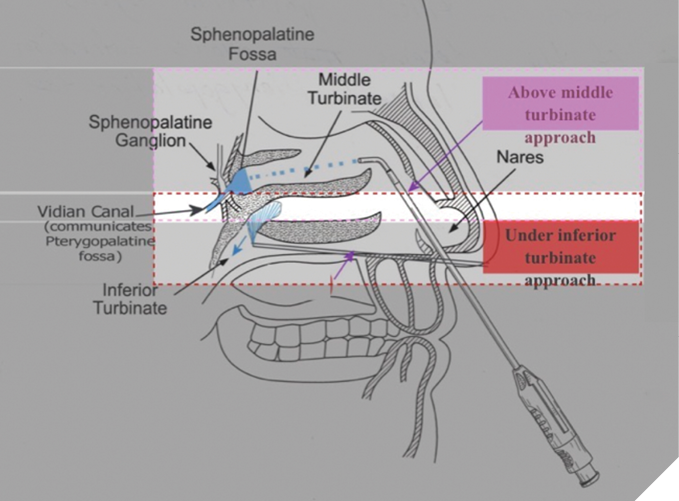
Facial temperature changes were assessed immediately following the procedure using digital infrared thermal imaging (DITI) to confirm an autonomic response indicative of successful block. Pain was measured pre- and post-intervention using the Visual Analog Scale (VAS). A follow-up assessment was conducted within 20 minutes post-treatment to measure short-term effects. The 20-minute observation period was used to confirm procedural efficacy, as indicated by facial temperature change, and to ensure that patients did not experience any immediate adverse effects.
In addition to the immediate post-procedure assessment, patients were followed clinically according to routine practice. Among patients receiving SPG block, follow-up assessments occurred at variable intervals after the initial SphenoCath treatment, with some patients undergoing a second treatment session at the same follow-up visit. However, several patients reported sufficient symptom relief after the initial treatment and did not return for a second session. The mean follow-up interval from the first SphenoCath treatment to subsequent assessment was approximately 3.8 weeks.
Matched control patients were followed according to a standardized conservative management protocol consisting of customized night guard wear and scheduled reassessments. No additional therapeutic modalities were introduced during the study window to preserve comparability between the intervention and control groups.
All patients had previously attempted conservative care that yielded only partial relief, ensuring clinical justification for either continued conservative management (controls) or minimally invasive intervention (SPG block). Informed consent was obtained from all participants.
Statistical analysis
Baseline characteristics were summarized using descriptive statistics. Continuous variables (age, duration, VAS pain scores) are presented as means with corresponding ranges where available, while categorical variables (sex, diagnosis) are reported as counts and percentages.
Within-group changes in VAS were assessed using paired t-tests and, given the small sample (n=4 per group), Wilcoxon signed-rank tests were also computed to provide a non-parametric complement. Between-group differences in change scores (pre–post) were evaluated using Welch’s t-test and Mann–Whitney U test to account for potential inequality of variance and non-normality. The effect size for the between-group difference was expressed as Cohen’s d.
Ninety-five percent confidence intervals (95% CIs) were calculated for mean change scores to quantify the precision of observed effects.
Sample size estimates for a future randomized controlled trial (RCT) were obtained using two-sample t-test power analysis (α=0.05, 80% power) and were informed by the effect size measured in this pilot study. Because the sample was limited, all statistical tests were considered exploratory.
Results and discussion
Eight patients were included: four treated with SphenoCath and four with a night guard. Mean age was 47.0 years in the SphenoCath group and 43.5 years in the night guard group; sex distributions were similar (3F/1M in each group). Mean symptom duration was 5.5 vs 14.8 years, respectively.
In the SphenoCath group, mean VAS decreased from 6.00 to 1.50 in the SphenoCath group (mean change -4.50); paired t-test p=0.042, Wilcoxon p=0.125. In the night guard group, mean VAS decreased from 5.50 to 1.75 (mean change -3.75); paired t-test p=0.065, Wilcoxon p=0.109. Between-group comparison of change scores was not statistically significant (Welch’s t-test p=0.702; Mann–Whitney p=0.661). Treatment outcomes, including mean pre- and post-treatment VAS scores, are summarized in Table 2. Post-treatment, 3/4 SphenoCath patients and 2/4 night guard patients achieved VAS ≤ 1. Group-level changes in mean VAS scores are illustrated in Figure 2, and individual patient trajectories are shown in Figure 3.
Table 2: Treatment outcomes based on mean change (pre–post) treatment VAS scores in the SphenoCath and night guard groups.
| Group | Mean VAS Before | Mean VAS After | Mean Change | N with VAS ≤ 1 (post) | Total N |
| SphenoCath | 6.0 | 1.5 | -4.5 | 3 | 4 |
| Night Guard | 5.5 | 1.75 | -3.75 | 2 | 4 |
Fig. 2
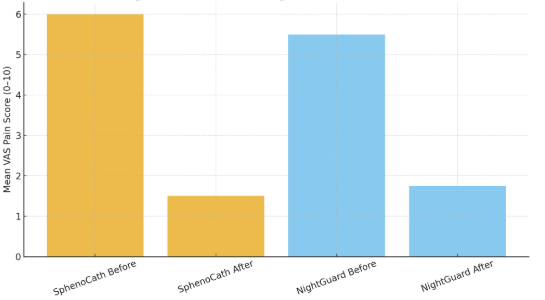
Fig. 3
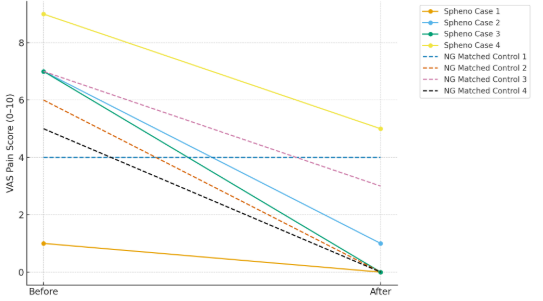
Both groups experienced notable reductions in pain, with the SphenoCath group showing a numerically larger decrease. Although the study was not powered to detect statistically significant between-group differences, the direction of change suggests a potential added benefit of SPG block beyond standard conservative therapy.
These findings are consistent with prior reports describing rapid analgesic effects following SPG-targeted treatments in craniofacial pain. The short-term improvements observed here indicate that SPG block may serve as a useful adjunct for patients who obtain only partial relief from oral appliances or other first-line measures.
The absence of serious adverse events and consistent physiologic response on thermographic imaging support the feasibility and tolerability of this intervention. These early results provide a preliminary signal justifying further evaluation in a larger, adequately powered randomized controlled trial. As a feasibility study, the goal was not to demonstrate statistical significance but to evaluate procedure consistency, tolerability, and short-term response patterns. Follow-up assessments beyond the immediate post-procedure period were available for several patients; however, follow-up timing varied and was not standardized as part of the pilot design. Although symptom duration differed between groups, both cohorts met standardized DC/TMD diagnostic criteria and had demonstrated incomplete response to conservative therapy, supporting clinical comparability for this pilot evaluation. The very short follow-up window limits conclusions regarding durability of effect and should be interpreted as an assessment of immediate response rather than sustained analgesia.
Sample size justification for future RCT
The observed between-group effect size for change in VAS was small (Cohen’s d = 0.28). Based on this effect size, an estimated 196 participants per group would be required to achieve 80% power at α=0.05 for a two-sided test. For planning purposes, alternative scenarios suggest that detecting a moderate effect (d = 0.5) would require ~64 per group, d = 0.6 ≈ 45 per group, and d = 0.8 ≈26 per group. A pilot RCT may therefore target 25–30 participants per arm to provide stable estimates of variability and treatment effects.
Conclusions
In summary, this pilot retrospective case–control study demonstrates that transnasal sphenopalatine ganglion (SPG) block using the SphenoCath device is a feasible and well-tolerated intervention and may be associated with short-term pain reduction in patients with chronic temporomandibular disorder (TMD) who have not achieved adequate relief with conservative therapy. Both the SPG-treated and control groups experienced clinically meaningful improvements in pain, with a numerically greater reduction observed in the SPG group. Although the study was not powered to detect statistically significant between-group differences, the observed trends, favorable safety profile, and consistent physiologic responses support the plausibility of SPG block as a potential adjunctive treatment for selected patients. Importantly, these findings highlight the value of pilot studies in generating preliminary effect estimates, assessing feasibility, and informing the design of future randomized controlled trials. Larger, adequately powered prospective studies with longer follow-up are warranted to more definitively evaluate efficacy, durability of effect, and patient subgroups most likely to benefit.
Future research
Given the promising pain reduction observed in this pilot evaluation, a larger randomized controlled trial is warranted to more definitively assess the clinical efficacy of SphenoCath-guided SPG block in patients with chronic TMD-related pain. Future studies should incorporate an adequately powered sample, guided by effect size estimates generated in the present analysis, to detect clinically meaningful between-group differences. Enhanced blinding procedures, broader inclusion criteria, and stratification by TMD subtype and duration of symptoms may further clarify which patient populations derive the greatest benefit.
In addition to pain measures, future research should integrate objective physiologic outcomes, such as digital infrared thermal imaging, to verify autonomic block and evaluate correlations between thermographic changes and clinical response. Longitudinal follow-up is also needed to determine optimal treatment intervals and durability of effect. Comparative studies examining SPG block in combination with standard conservative therapies may help define its role within a multidisciplinary care pathway for refractory TMD. 
Oral Health welcomes this original article.
References
- Eldredge S. Transnasal sphenopalatine ganglion block using the SphenoCath catheter. SphenoCath instructional presentation. Canada; 2019. URL: https://sphenocath.ca/wp-content/uploads/2019/05/AA11Canadatactile.pptx
- Zieliński G, Pająk-Zielińska B, Ginszt M. A meta-analysis of the global prevalence of temporomandibular disorders. J Clin Med. 2024;13(5):1365.
- Dan R, Li J, Xie T, Luo M, Lau RS, Hu S, et al. Impact of different types of temporomandibular disorders on jaw functional limitation and psychological distress in orthodontic patients. J Oral Rehabil. 2023;50(8):644–654.
- Elstad E, Bocell FD, Owens TC, Logan D, Melluso E, Viscione C, et al. Focus groups to inform the development of a patient-reported outcome measure (PROM) for temporomandibular joint disorders (TMDs). Patient. 2023;16(3):265–276.
- Kapos FP, Exposto FG, Oyarzo JF, Durham J. Temporomandibular disorders: a review of current concepts in aetiology, diagnosis, and management. Oral Surg. 2020;13(4).
- Patel K, Eley KA, Cascarini L, Watt-Smith S, Larkin M, Lloyd T, et al. Temporomandibular disorders—review of evidence-based management and a proposed multidisciplinary care pathway. Oral Surg Oral Med Oral Pathol Oral Radiol. 2023.
- Harper DE, Schrepf A, Clauw DJ. Pain mechanisms and centralized pain in temporomandibular disorders. J Dent Res. 2016;95(10):1102–1108.
- Garstka AA, Kozowska L, Kijak K, Brzózka M, Gronwald H, Skomro P, et al. Accurate diagnosis and treatment of painful temporomandibular disorders: a literature review supplemented by own clinical experience. Pain Res Manag. 2023;2023:1002235.
- Ferneini EM. Temporomandibular joint disorders (TMD). J Oral Maxillofac Surg. 2021;79(10):2171–2172.
- Alexander CE, Dua A. Sphenopalatine ganglion block. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2022.
- Piagkou M, Demesticha T, Troupis T, Vlasis K, Skandalakis P, Makri A, et al. The pterygopalatine ganglion and its role in various pain syndromes: from anatomy to clinical practice. Pain Pract. 2011;12(5):399–412.
- Robbins MS, Robertson CE, Kaplan E, Ailani J, Charleston L, Kuruvilla D, et al. The sphenopalatine ganglion: anatomy, pathophysiology, and therapeutic targeting in headache. Headache. 2016;56(2):240–258.
- Loder E, Weizenbaum E. Sphenopalatine ganglion block for headache disorders. UpToDate. 2020.
- Mojica J, Mo B, Ng A. Sphenopalatine ganglion block in the management of chronic headaches. Curr Pain Headache Rep. 2017;21(6).
- Cady R, Saper JR, Dexter K, Manley HR. A double-blind, placebo-controlled study of repetitive transnasal sphenopalatine ganglion blockade with Tx360® as acute treatment for chronic migraine. Headache. 2014;55(1):101–116.
- Cady R, Saper JR, Dexter K, Cady RJ, Manley HR. Long-term efficacy of repetitive sphenopalatine blockade with bupivacaine vs saline using the Tx360® device for chronic migraine. Headache. 2015;55(4):529–542.
- Binfalah M, Alghawi E, Shosha E, Alhilly A, Bakhiet M. Sphenopalatine ganglion block for the treatment of acute migraine headache. Pain Res Treat. 2018;2018:1–6.
- Burkett JG, Robbins MS, Robertson CE, Mete M, Saikali NP, Halker Singh RB, et al. Sphenopalatine ganglion block in primary headaches. Neurol Clin Pract. 2019;10(6):503–509.
- Ho KWD, Przkora R, Kumar S. Sphenopalatine ganglion: block, radiofrequency ablation and neurostimulation—a systematic review. J Headache Pain. 2017;18(1).
- Tolba R, Weiss AL, Denis DJ. Sphenopalatine ganglion block and radiofrequency ablation: technical notes and efficacy. Ochsner J. 2019;19(1):32–37.
- Shah RJ, Dixon B, Padalia D. Sphenopalatine ganglion radiofrequency thermocoagulation. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2023.
- Pattinson B. Sphenopalatine ganglion block in the relief of pain. J Orthop Med. 1995;17(3):104–109.
- Kim NE, Park B, Moon YR, Lee SY, Gil HY, Kim S, et al. Changes in facial temperature measured by digital infrared thermal imaging in patients after transnasal sphenopalatine ganglion block. Medicine. 2019;98(15):e15084.
- Kim NE, Kim JE, Lee SY, Gil HY, Min SK, Park B, et al. Comparison of temperature and pain changes between drip and topical methods of administering the transnasal sphenopalatine ganglion block. J Pers Med. 2022;12(5):830.
- Hertzog MA. Considerations in determining sample size for pilot studies. Res Nurs Health. 2008;31(2):180–191.
- Julious SA. Sample size of 12 per group rule of thumb for a pilot study. Pharm Stat. 2005;4(4):287–291.
About the authors

Avery Kirchmann is a third-year undergraduate student at McGill University, majoring in Political Science with a minor in the Sociology of Medicine. Her academic interests lie at the intersection of healthcare, medicine, and social policy.

Dr. Sherif Elsaraj is the current President of the Ottawa Dental Society. He earned his Doctor of Dental Medicine degree and a Master’s degree in Oral Biology from the University of Manitoba, and later completed a PhD in Craniofacial Pain and Health Sciences at McGill University. He is currently an adjunct professor in the Department of Family Medicine at the University of Ottawa.