In the patient’s pursuit of restoring both aesthetics and function, dental implants have become one of the most widely used treatment modalities for the rehabilitation of partially and completely edentulous cases. Despite their high long-term success rates, dental implants remain susceptible to biologic complications such as peri-implant mucositis and peri-implantitis. These peri-implant diseases are primarily associated with bacterial biofilm, often in combination with patient and prosthesis related risk factors.1
Peri-implant mucositis is a reversible inflammatory condition limited to the peri-implant soft tissues, with no radiographic signs of bone loss.2 It is a common condition, with reported prevalence rates ranging from approximately 24% to 70%, depending on the study population and diagnostic criteria.2
Peri-implantitis is a non-reversible inflammatory condition affecting both the peri-implant soft tissues and supporting bone.2 Diagnosis typically involves bleeding and/or suppuration on probing, progressively increasing probing depths and radiographic bone loss.2 Measurement thresholds vary between studies, prevalence estimates range from 5% to 45%, but peri-implantitis is recognized as a relatively common biological complication of implant therapy,2 which can often be linked to the same reasons as initial tooth loss.
Here, we present the case of an asymptomatic patient with full-arch implant-supported fixed bridges who had not received professional hygiene care in over twenty-five years. This case highlights advanced peri-implant disease, complicated by improper use of bonding material over exposed implant threads. It underscores the essential role of regular supportive implant maintenance in preserving bone and preventing implant loss. Ongoing implant maintenance and patient education are necessary to ensure the patient understands the level of care required, both professionally and personally, to sustain health.
Case presentation
The patient, a 74-year-old male, was referred to our periodontal clinic for an implant maintenance visit. The patient is a non-diabetic and non-smoker, comorbidities include atrial fibrillation and hypertension. He is currently under the care of a physician for management of atrial fibrillation and is on long-term anticoagulant therapy. Cardiovascular disease, including atrial fibrillation, is recognized as a risk factor for periodontal disease through both infectious and inflammatory pathways, further underscoring the importance of ongoing periodontal maintenance.3 The patient lost his natural teeth due to a combination of periodontal disease, parafunction and failed root canal procedures. Following a discussion with his dentist, the decision was made to remove all natural teeth and replace them with dental implants.
The patient had 16 BioHex implants spanning both arches placed in 2000 by his dentist. The implants support two fixed full-arch prostheses. The patient reported receiving only occasional dental care during the first 15 years after implant placement, limited to prosthetic issues, and no implant maintenance or dental care in the past 10 years. His home care consisted of brushing his teeth with a manual toothbrush two times per day and rinsing with mouthwash.
The patient did not have a dental care provider and was referred to our clinic by a caring personal acquaintance who was also a dentist. The patient had previously sought care for their dental concerns (swelling in the upper left and lower anterior) but was unable to find a dentist willing to proceed with treatment due to the complexity of the case.
Despite the absence of professional follow-up, the patient reported being generally satisfied with the appearance and function of his prosthetics. His motivation for seeking care was preventive and to establish a provider for ongoing maintenance “in case of trouble later.”
Clinical findings
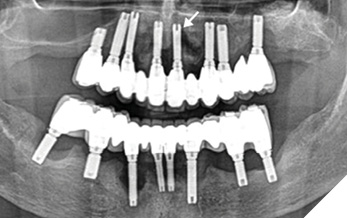
The patient reported no current pain or functional limitations but acknowledged occasional gingival sensitivity during home care. He expressed satisfaction with his chewing ability and prostheses stability. The panoramic radiograph from the referring dentist revealed generalized radiographic bone loss around all implants (Fig. 1).
Fig. 1
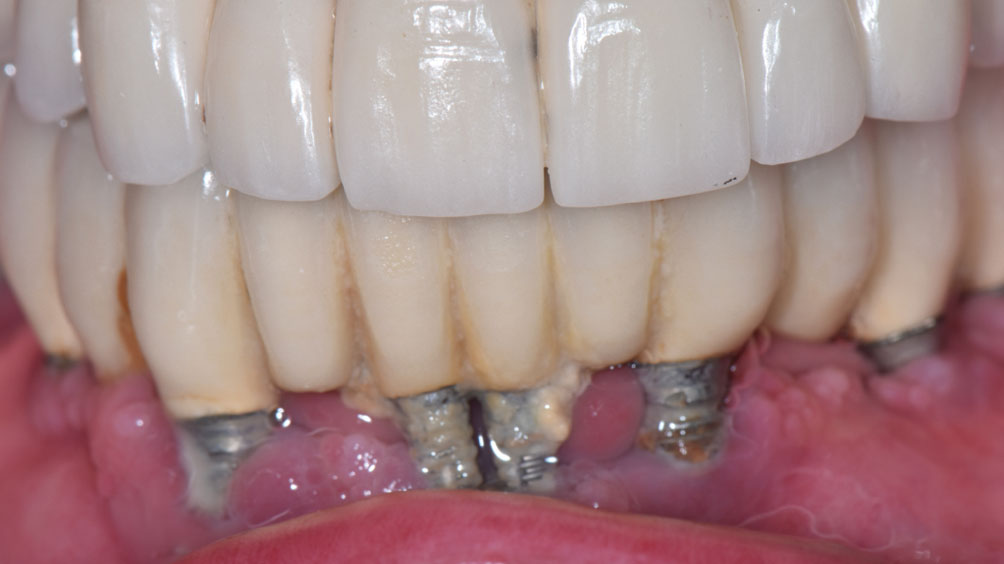
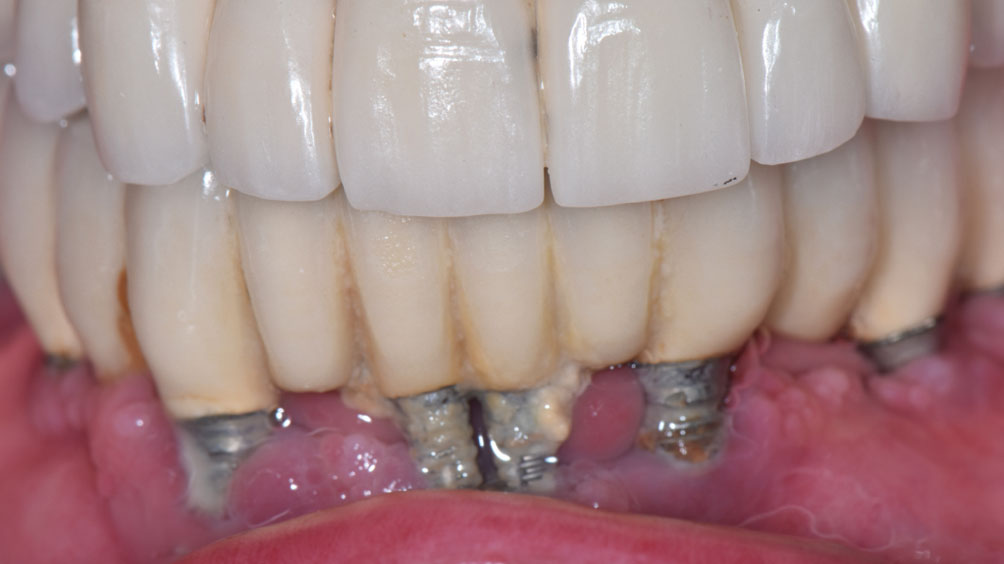
On physical examination, heavy biofilm and calculus deposits were present on all implants, with generalized bleeding upon debridement and suppuration noted at multiple sites (Fig. 2). Multiple implants exhibited advanced recession with exposed threads, most notable in the mandibular anterior region. Composite resin bonding material was noted on the implant threads at teeth 13, 33, and 41, which the patient reported was placed years earlier to mask exposed threads (Fig. 3).
Fig. 2
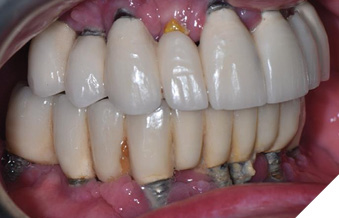
Fig. 3
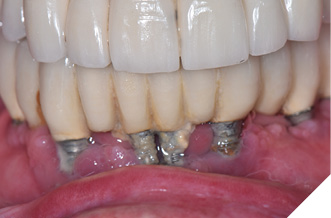
Keratinized tissue was generally thin and no clinical mobility of the implants was detected as they are splinted together. Due to the patient’s dental anxiety, blood pressure of 172/101 and tissue sensitivity, no probing depths were recorded at the initial appointment.
Diagnostic assessment
Given the patient’s limited history, suppuration around multiple implants, generalized inflammation, and bone loss, the primary diagnosis was generalized peri-implantitis. Generalized peri-implant bone loss averaging approximately 65% was observed, with the most severe bone loss found at the 21 implant, measuring 10.5 mm based on the panoramic radiograph (Fig. 1).
The presence of exposed threads, localized bonding material bonded to implant surfaces, heavy calculus and biofilm accumulation were identified as significant contributing factors to the peri-implantitis. Due to the advanced recession, bone loss and generalized bleeding on debridement, the long-term prognosis for all implants was considered doubtful.
The severity of bone loss and prosthetic design posed challenges for cleansability and access to implant surfaces during supportive implant maintenance procedures.
The patient was given the option to remove the superstructure, extract any failed implants, and modify or replace the superstructure. The patient was genuinely surprised that the condition was as serious and advanced as it was, considering he experienced little discomfort or restrictions in function. Before consenting to the removal of the superstructure, the patient wanted to proceed with non-surgical supportive periodontal maintenance and review the outcome of that treatment. This maintenance approach aligns with the Fonthill Implant Care Protocol, which emphasizes early detection and biofilm disruption to prevent peri-implant disease, reinforcing evidence that structured, evidence-based recall systems can significantly improve long-term implant prognosis.4 Consultation with his physician regarding his hypertension was also recommended.
Therapeutic intervention
The treatment objective for this initial appointment was not definitive therapy, but rather palliative care, focusing primarily on supragingival and limited subgingival debridement to reduce inflammation. The aim was also to foster a positive treatment experience, encouraging the patient’s commitment to ongoing care and to develop a more effective personalized home-care strategy.
In accordance with Guided Biofilm Therapy (GBT) principles, the instrumentation for this appointment included EMS AirFlow with low-abrasive erythritol powder for disrupting biofilm, Blue & Green Inc. titanium implant scalers, Mini-Gracey titanium curettes (PDT), and a carbon-coated PI Max Piezon ultrasonic tip to efficiently and safely remove calculus from the titanium implants. GBT emphasizes biofilm disclosure, minimally invasive air polishing, and implant-safe ultrasonic instrumentation to achieve thorough debridement while preserving hard and soft tissue integrity.5
Discussion
This case demonstrates how the absence of structured professional maintenance care predictably accelerates peri-implant disease progression. The patient had not been adequately educated on proper at-home implant care or the importance of consistent professional maintenance to disrupt biofilm around his complicated prosthetics. The lack of professional maintenance combined with inadequate home care contributed to heavy biofilm and calculus accumulation, generalized inflammation, bone loss and suppuration across both arches.
The use of composite resin to cover exposed implant threads in the maxillary and mandibular anterior region likely compromised surface integrity, leading to microscopic porosities, visible surface irregularities, and increased biofilm retention. Surface roughness within the intraoral environment significantly influences the initial adhesion and subsequent retention of microorganisms, with these effects being further amplified when the surfaces are located subgingivally.6 Additionally, the prosthetic design created inaccessible areas that hindered effective daily biofilm control at home.
Regular professional maintenance every three months is often recommended for high-risk implant patients to prevent the progression of peri-implant disease. Biofilm development occurs over several weeks and stabilizes as a mature subgingival biofilm at approximately 12 weeks, beyond which the inflammatory burden increases significantly.7
For dentists, hygienists, and lab technicians, this case emphasizes the need to clearly plan for long-term maintenance protocols for all implant patients. Regularly scheduled periodontal maintenance is critical in managing peri-implant biofilm, particularly in complex cases such as this one, which carries an underlying risk from previous tooth loss from periodontitis and limitations from personal hygiene.
While one might argue that this reconstruction has provided the patient with 25 years of acceptable function and esthetics, such an assessment overlooks the biologic burden of chronic peri-implant infection. The patient has lived for decades with a persistent source of inflammation and suppuration in his oral cavity, which almost certainly has systemic implications. Cardiovascular disease, including atrial fibrillation, has well-established links to periodontal and peri-implant infection through both infectious and inflammatory pathways. To illustrate the severity, if a patient presented with an ulcerated, suppurating lesion on their arm that persisted for 25 years, no clinician would consider this acceptable or compatible with health. The oral cavity should not be judged by a different standard. This case underscores the danger of equating prosthetic survival with treatment success, while ignoring the biological and systemic costs of unmanaged peri-implant disease.
Conclusion
This case of generalized peri-implantitis in a patient with long-standing maxillary and mandibular full-arch implant-supported prostheses emphasizes the consequences of extended neglect of professional maintenance. The combination of a history of tooth loss due to periodontitis, an inaccessible prosthetic design, substantial biofilm and calculus accumulation, and the use of bonding material over exposed threads created an environment that facilitated disease progression and impaired peri-implant health without the safety net of professional maintenance.
It highlights the importance of regular supportive implant maintenance intervals in preserving peri-implant bone and preventing implant failure. Dental professionals must ensure patients understand that an implant is not a one-time procedure but a lifelong commitment to maintenance. This includes thorough patient education on effective home care and the need for professional maintenance to control biofilm. Timely intervention, patient education, and proper referral for complex cases remain the standards of successful, long-term implant therapy. 
Oral Health welcomes this original article.
References
- Sun TC, Chen CJ, Gallucci GO. Prevention and management of peri-implant disease. Clin Implant Dent Relat Res. 2023 Apr 12. doi: 10.1111/cid.13206.
- Wada M, Mameno T, Otsuki M, Kani M, Tsujioka Y, Ikebe K. Prevalence and risk indicators for peri-implant diseases: A literature review. Jpn Dent Sci Rev. 2021 Jun 8;57:78-84. doi: 10.1016/j.jdsr.2021.05.002.
- Froum SJ, Hengjeerajaras P, Liu KY, Maketone P, Patel V, Shi Y. The link between periodontitis/peri-implantitis and cardiovascular disease: a systematic literature review. Int J Periodontics Restorative Dent. 2020 Nov-Dec;40(6):e229-e233. doi: 10.11607/prd.4591.
- Fritz PC, Lavoie DM, More R, Dakin LM, Nahli A, Longo AB. The Fonthill Implant Care Protocol: a critical update. Oral Health. 2019 Oct 8.
- Electro Medical Systems (EMS). The unique story of Guided Biofilm Therapy. Nyon, Switzerland: EMS Dental; [accessed 2025 Aug 9]. Available from: https://www.ems-dental.com/en/unique-story-gbt
- Rashid H. The effect of surface roughness on ceramics used in dentistry: A review of literature. Int J Dent. 2014;2014:852730. doi:10.1155/2014/852730. PMID: 25512743; PMCID: PMC4253118.
- Mocanu RC, Martu MA, Luchian I, Sufaru IG, Maftei GA, Ioanid N, et al. Microbiologic profiles of patients with dental prosthetic treatment and periodontitis before and after photoactivation therapy—randomized clinical trial. Microorganisms. 2021 Mar 30;9(4):713. doi:10.3390/microorganisms9040713.
About the authors

Meghan Demetraki Paleolog is pursuing her Doctor of Dental Surgery (DDS) as a third-year student at the University of Toronto. This past summer, Meghan participated in the AI-Enhanced Periodontal Residency at Dr. Peter C. Fritz’s clinic in Fonthill, Ontario.

Colleen C. Porter is a Registered Dental Hygienist working in a periodontal specialty office and serving as part-time Professor at Niagara College Canada. She earned her Certificate in Dental Hygiene from Niagara College and her Bachelor of Education in Adult Education from Brock University.

Dr. Peter Fritz is a pioneering periodontist and implant surgeon, blending clinical excellence with a deep understanding of the legal and ethical implications of emerging digital technologies in dentistry. Holding adjunct positions at McMaster University and Brock University, Dr. Fritz’s interdisciplinary research focuses on innovative approaches to enhancing patient care. His clinic in Fonthill, Ontario, is recognized for setting new standards in patient care through AI-driven practices. With advanced degrees in both dentistry and law, Dr. Fritz exemplifies a commitment to advancing dental science with a focus on innovation, ethics, and exploration.