Summary
Shwachman-Diamond Syndrome (SDS) is a rare autosomal recessive disorder characterized by various systemic manifestations, including pancreatic insufficiency, bone marrow dysfunction, and skeletal abnormalities. The dental management of patients with SDS is an emerging area of interest and this narrative review aims to provide a comprehensive overview of SDS, its symptoms, and the implications for dental management.
Oral disease disproportionately affects patients with SDS, significantly impacting their quality of life. Patients with SDS have been found to have significantly more caries in primary and permanent dentitions, delayed dental development, recurrent oral ulcerations, gingival bleeding upon brushing, and pain while eating, compared to healthy controls.
We suggest dental care is provided by a pediatric dentist, with close communication with the patient’s medical team and other dental specialist, like orthodontists, due to the complicating systemic factors and medical considerations involved in the dental management of these patients.
However, general dentists ought to be aware of SDS and its associated dental management considerations, as not all SDS patients will be able to see pediatric dentists. Further research is also required to better understand the pathophysiology of SDS, its oral manifestations, and optimal treatment modalities.
Shwachman-Diamond Syndrome (SDS) is a rare autosomal recessive disorder characterized by pancreatic insufficiency, bone marrow dysfunction, and skeletal abnormalities.1 SDS was first described in 1964 by Bodian et al.2, and it’s prevalence is estimated to be 1:100,000 to 1:200,000 live births with a median life expectancy of 35 years.3,4 Eighty-nine percent of cases involve a mutation in the SBDS gene, located on chromosome 7q11, which results in a truncated protein usually responsible for RNA metabolism and processing.1 SDS presents with a spectrum of clinical manifestations, including delayed growth, pancreatic insufficiency, neutropenia, thrombocytopenia, and increased risk of developing leukemia.3,5
While the systemic effects of SDS have been well-documented, its impact on oral health and dental management is an emerging area of interest. Patients with SDS may experience unique challenges related to dental care with the burden of oral disease having a significant impact on their quality of life, with dental abscesses being the most common infections in these patients and resulting in reduced appetite, due to pain while eating.2,6
The dental management of patients with SDS requires a multidisciplinary approach, involving collaboration between dentists, physicians, and other healthcare providers. Early identification of oral manifestations, tailored preventive strategies, and careful consideration of pharmacological interactions are essential components of effective dental care for individuals with SDS.
This narrative review aims to provide a comprehensive overview of SDS, its symptoms, and the implications for dental management. By synthesizing existing literature and highlighting key clinical considerations, this paper seeks to enhance understanding and promote optimal oral health outcomes for patients with this rare genetic syndrome.
Fig. 1
Fig. 2
Systemic manifestations
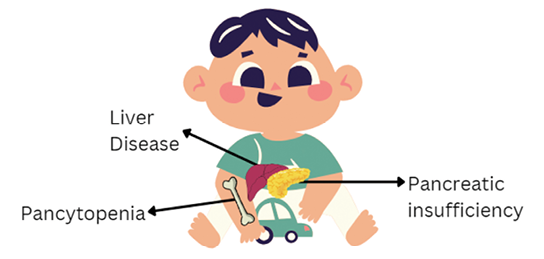
Pancreatic insufficiency: Approximately 90% of SDS patients are estimated to have pancreatic dysfunction within the first year of life.7 Histological examination found SDS patients had fat cells replacing acini cells with increased interstitial connective tissue, dilation of some ductules, while the isle of Langerhans was preserved.8 Pancreatic insufficiency associated with SDS can be diagnosed using the classification and regression tree (CART) analysis for patients over 3 years of age, in conjunction with MRI used to detect the fatty tissue replacement.9 Clinically, the secretion of proteolytic enzymes is severely decreased leading to steatorrhea, with 50% of cases requiring treatment with pancreatic enzyme supplements.10
Liver disease: SDS presents with enlarged liver, with increased serum liver enzymes in 75% of infants and children.11 Fifty-eight percent of patients were found to have mild to moderate elevation of serum bile acide.11 However, these conditions resolve at approximately 5 years of age and appear not to have significant effects in adulthood, with magnetic resonance imaging (MRI) suggesting no evidence of hepatic steatosis, cirrhosis, or fibrosis.11 However, patients may have mild cholestasis in the long term and have a tendency to develop hepatic and renal microcysts.11
Hematologic manifestations: Various hematologic conditions present with SDS, including anemia, raised fetal haemoglobin, neutropenia, impaired neutrophil chemotaxis, thrombocytopenia, and increased risk of myelodysplastic syndrome and acute myeloid leukemia (AML).5,12 See Table 1 for estimated prevalence of these conditions and note that due to the rare nature of SDS the epidemiologic data on it is heavily biased due to the small sample sizes, which may over or under estimate its true prevalence.
Table 1: Prevalence of Haematologic abnormalities in SDS Patients5,12
| Abnormality | Estimated Prevalence (%) |
| Neutropenia | 88-100 |
| Anemia (normocytic and normochromic) | 42-80 |
| Fetal hemoglobin elevation | 44-80 |
| Thrombocytopenia | 24-88 |
| Pancytopenia | 10-65 |
| Bone marrow dysplasia | 0-54 |
| Bone marrow hypoplasia | 39-100 |
| Myelodysplastic syndrome | 0-44 |
| Acute myeloid leukemia | 5-24 |
Approximately one third of neutropenia cases are chronic, while two thirds are intermittent, and may be characterized by poor chemotaxis, migration, and mobility of neutrophils.5 The majority of cases of thrombocytopenia are mild; however, some cases are severe and usually associated with AML.5
Haematological conditions are the primary source of morbidity and mortality for SDS patients. SDS patients have an estimated median survival of over 35 years old.12 The mechanism and pathophysiology regarding haematologic conditions remains unclear and may be multifactorial, with further research necessary to discover pathways for targeted therapy.12
Oral manifestations
Patient with SDS have been found to have significantly more caries in primary and permanent dentitions, delayed dental development, recurrent oral ulcerations, gingival bleeding upon brushing, and pain while eating, compared to healthy controls.4
Dental caries is a multifactorial disease and in patients with SDS neutropenia may play a role in its development. The human neutrophil peptides 1-3, found within neutrophils and normally present in gingival crevicular fluid, are involved in nonoxidative microbial elimination, and their absence may allow for propogation of cariogenic bacteria, leading to increased caries in SDS patients.13 However, this relationship is not well established and requires further investigation.
Another factor that may play a role in the increased caries risk of SDS patients is the frequent use of cariogenic sugared antibiotic syrups. Oral hygiene may also be compromised due to pain while brushing the teeth, due to gingival inflammation or oral ulceration. Moreover, the use of drugs like antiemetics, gastric acid secretion inhibitors, and decongestants may result in xerostomia as an adverse effect, further increasing the caries risk.
SDS patients are more likely to have delayed eruption which may be related to delayed skeletal age.4 Other anomalies, like dental dysplasia, impaction, hypodontia, hyperdontia, and transposition rarely occurred in patients with SDS and are likely not a manifestation of the syndrome.
Recurrent oral ulcerations are well known to be associated with neutropenia and were found to be present in 66% of SDS patients, often occurring over four times per year.4 The exact pathophysiology in relation to neutrophils is unclear and requires further investigation. Other factors, like stress, nutritional deficiencies, acute infections, immunodeficiency, and trauma, may also play a role in ulcer development in these patients.
Gingival bleeding may be associated with thrombocytopenia, poor oral hygiene, or secondary vitamin k deficiency following long term antibiotic use.5 The effects of neutropenia on periodontal disease are not clear. It is well documented that periodontitis can present as a manifestation of neutropenia, as the lack of immune cells results in impaired defense against gingival pathogens.14-20 However, some reports suggest neutropenia may act as a protective factor, as a lack of neutrophils and their associated lysosomal enzymes in the gingival tissue may result in reduced inflammation and breakdown of periodontal tissue.4
Fig. 3

Dental management
SDS patients are at elevated risk of dental disease and require careful consideration of systemic and psychosocial factors when preventing and treating infections and symptoms. Preventive measures to reduce caries risk, like regular recall visits, sealants, professional fluoride application, and oral hygiene instruction ought to be prioritized from a young age. If signs or symptoms of xerostomia are reported or observed, then saliva promoting or substitute measures, like xylitol containing gum/candies, should be considered, with patients being informed that excessive doses of xylitol may result in upset stomach.
Scaling and root planning in conjunction with systemic antibiotics has been found to be successful in treating most cases of periodontitis in patients with neutropenia.20 Treatment for management of SDS patient’s gingival condition should consider regular scaling visits every three months, periodic chlorhexidine mouth rinse, and prophylactic antibiotics.
Oral ulcerations ought to be investigated for potentiating factors and can be managed with topical anesthetics or benzydamine. Prior to prescribing or administering any drug, dentists should consider the weight of the patient, due to the slower rate of growth, and consider that SDS patients may reach toxic doses more quickly due to hepatic abnormalities. Acetaminophen is a common drug used for pain management in children, and patients with SDS should be warned about the possible risks associated with taking it in toxic doses, again, due to the liver abnormalities and slow growth pattern associated with SDS.11
Furthermore, the delayed growth pattern linked with delayed erruption of teeth should be followed by an orthodontist to ensure occlusion is not negatively impacted long term.
Prior to surgical procedures, a platelet count should be obtained, due to the risk of thrombocytopenia. Other precautionary measures, like treatment in hospital or replacement factors, may also be considered when appropriate.
Moreover, due to the increased risk of AML in SDS patients, they should be closely monitored for early signs of AML. Oral manifestations of leukemia include gingival hyperplasia with spontaneous bleeding, fungal infections, oral ulcers, and petechial haemorrhages of the tongue, lips and palate.21 Systemic signs and symptoms of leukemia include fatigue, lymphadenopathy, recurrent infection, bone and abdominal pain, and purpura.21 Monitoring for signs of ALM may prove to be difficult, as many of the signs are similar to the clinical manifestations of SDS, highlighting the importance of interdisciplinary care and communication with the patients’ medical team.
Conclusion
Oral disease disproportionately affects patients with SDS, significantly impacting their quality of life. The systemic manifestations associated with SDS make the dental management of these patient more complex. We suggest dental care is provided by a pediatric dentist, with close communication with the patient’s medical team and other dental specialist, like orthodontists, due to the complicating systemic factors and medical considerations involved in the dental management of these patients. However, general dentists ought to be aware of SDS and its associated dental management considerations, as not all SDS patients will be able to see pediatric dentists. Finally, further research is required to better understand the pathophysiology of SDS, its oral manifestations, and optimal treatment modalities. 
Oral Health welcomes this original article.
References
- Boocock GRB, Morrison JA, Popovic M, Richards N, Ellis L, Durie PR, et al. Mutations in SBDS are associated with Shwachman-Diamond syndrome. Nat Genet. 2003 Jan;33(1):97–101.
- Bodian M, Sheldon W, Lightwood R. Congenital Hypoplasia of the Exocrine Pancreas. Acta Paediatr. 1964 May;53(3):282–93.
- Kathuria R, Poddar U, Yachha SK. Shwachman-Diamond Syndrome: are we missing many? Indian Pediatr. 2012 Sep;49(9):748–9.
- Ho W, Cheretakis C, Durie P, Kulkarni G, Glogauer M. Prevalence of oral diseases in Shwachman‐Diamond syndrome. Spec Care Dentist. 2007 Mar;27(2):52–8.
- Smith OP, Hann IM, Chessells JM, Reeves BR, Milla P. Haematological abnormalities in Shwachman-Diamond syndrome. Br J Haematol. 1996 Aug;94(2):279–84.
- Ginzberg H, Shin J, Ellis L, Morrison J, Ip W, Dror Y, et al. Shwachman syndrome: Phenotypic manifestations of sibling sets and isolated cases in a large patient cohort are similar. J Pediatr. 1999 Jul;135(1):81–8.
- Ikuse T, Kudo T, Arai K, Fujii Y, Ida S, Ishii T, et al. Shwachman–Diamond syndrome: Nationwide survey and systematic review in Japan. Pediatr Int. 2018 Aug;60(8):719–26.
- Aggett PJ, Cavanagh NP, Matthew DJ, Pincott JR, Sutcliffe J, Harries JT. Shwachman’s syndrome. A review of 21 cases. Arch Dis Child. 1980 May;55(5):331–47.
- Ip WF, Dupuis A, Ellis L, Beharry S, Morrison J, Stormon MO, et al. Serum pancreatic enzymes define the pancreatic phenotype in patients with Shwachman-Diamond syndrome. J Pediatr. 2002 Aug;141(2):259–65.
- Mack D, Forstner G, Wilschanski M, Freedman M, Durie P. Shwachman syndrome: Exocrine pancreatic dysfunction and variable phenotypic expression. Gastroenterology. 1996 Dec;111(6):1593–602.
- Toiviainen-Salo S, Durie PR, Numminen K, Heikkilä P, Marttinen E, Savilahti E, et al. The Natural History of Shwachman-Diamond Syndrome–Associated Liver Disease from Childhood to Adulthood. J Pediatr. 2009 Dec;155(6):807-811.e2.
- Dror Y, Freedman MH. Shwachman–Diamond Syndrome. Br J Haematol. 2002 Sep;118(3):701–13.
- Antonio AG, Alcantara PCDC, Ramos MEB, De Souza IPR. The importance of dental care for a child with severe congenital neutropenia: a case report. Spec Care Dentist. 2010 Nov;30(6):261–5.
- Deasy MJ, Vogel RI, Macedo-Sobrinho B, Gertzman G, Simon B. Familial benign chronic neutropenia associated with periodontal disease. A case report. J Periodontol. 1980 Apr;51(4):206–10.
- Stabholz A, Soskolne V, Machtei E, Or R, Soskolne WA. Effect of benign familial neutropenia on the periodontium of Yemenite Jews. J Periodontol. 1990 Jan;61(1):51–4.
- van Winkelhoff AJ, Schouten-van Meeteren AY, Baart JA, Vandenbroucke-Grauls CM. Microbiology of destructive periodontal disease in adolescent patients with congenital neutropenia. A report of 3 cases. J Clin Periodontol. 2000 Nov;27(11):793–8.
- Carrassi A, Abati S, Santarelli G, Vogel G. Periodontitis in a patient with chronic neutropenia. J Periodontol. 1989 Jun;60(6):352–7.
- Baehni PC, Payot P, Tsai CC, Cimasoni G. Periodontal status associated with chronic neutropenia. J Clin Periodontol. 1983 Mar;10(2):222–30.
- Zaromb A, Chamberlain D, Schoor R, Almas K, Blei F. Periodontitis as a manifestation of chronic benign neutropenia. J Periodontol. 2006 Nov;77(11):1921–6.
- Schmidt JC, Walter C, Rischewski JR, Weiger R. Treatment of periodontitis as a manifestation of neutropenia with or without systemic antibiotics: a systematic review. Pediatr Dent. 2013;35(2):E54-63.
- Guan G, Firth N. Oral manifestations as an early clinical sign of acute myeloid leukaemia: a case report. Aust Dent J. 2015 Mar;60(1):123–7.
About the Authors

Dr. Mohammed Inam Ullah Khan is a clinical instructor at the Faculty of Dentistry, University of Toronto and works as a general dentist in private practice.

Dr. Saba Khuffash works as a general dentist in private practice.

Dr. Hassan El-Awour has won multiple awards for his teaching as a clinical instructor in the department of oral surgery at the Faculty of Dentistry, University of Toronto. He is an international speaker on various topics ranging from oral surgery, implantology, and bone biology and has won several awards for his research in oral surgery and oral medicine.