More than 100 years ago, the famed physician Charles Mayo, MD, stated that the future of medicine and disease prevention lay in the hands of and must come from the dental profession. Historically, the dental profession has not used or monitored patient wellness via traditional diagnostic testing as is common in medicine. Recently, there has been a movement among dental providers to become more intimately involved in the overall systemic wellness of their patients. A key means to this end is to perform basic diagnostic testing to monitor and detect disease. This movement is bringing those prescient words from Dr. Mayo to fruition. For the efficient re-engagement of dentistry back into medicine, a mechanism that allows for diagnostic insight and active engagement with medical providers can be achieved using saliva as a vehicle for patient monitoring, screening, diagnosis, and collaboration.
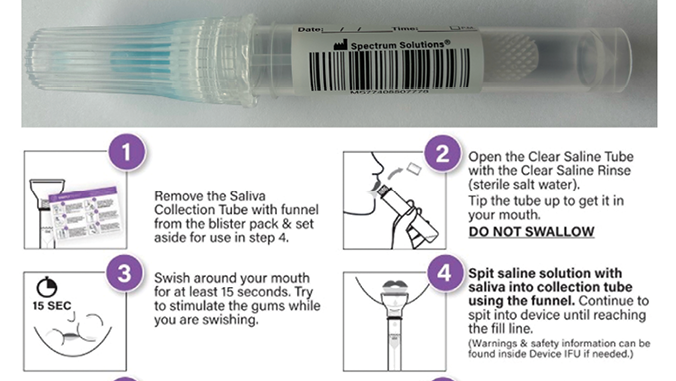
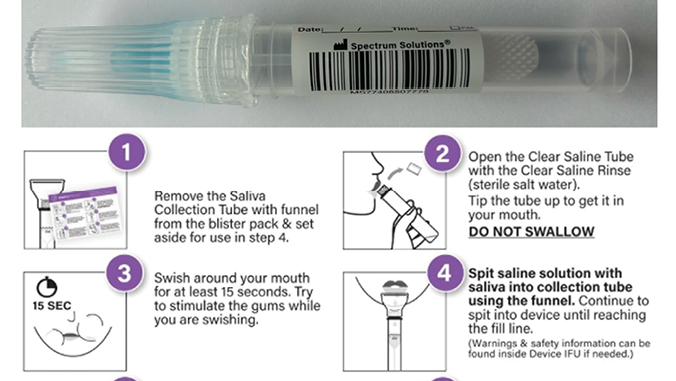
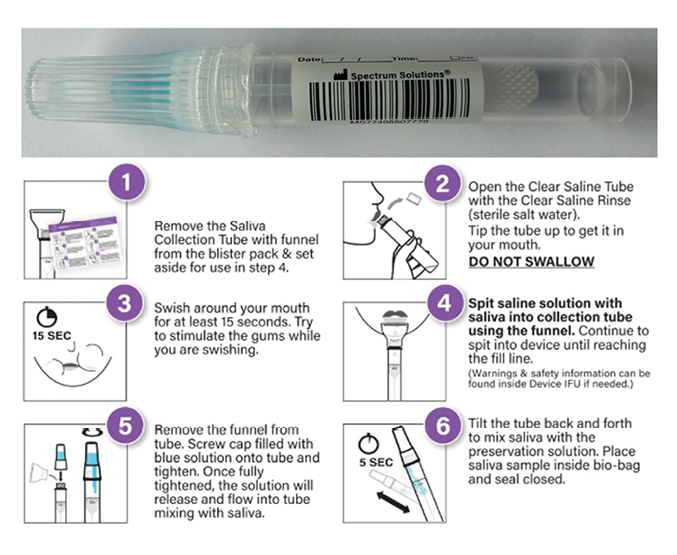
Saliva is a clear, heterogeneous biofluid consisting of water, proteins, nucleic acids, and inorganic components. On average, individuals produce between 0.5 and 1.5 liters daily.1 The multifunctional nature of saliva allows for the facilitation of digestion, swallowing, tasting, lubrication, and as a protective barrier against pathogens. The presence of proteins, peptides, nucleic acids, and macromolecules make saliva an ideal sample type for the screening, diagnosis, and monitoring of disease (Fig. 1). Over the last decade, it has become clear that saliva can serve as a mirror of the body since most of its fluid component originates from the blood. It is, therefore, a reflection of the natural substances and macromolecules produced during health and disease. Like saliva, blood is a complex bodily fluid that contains a wide range of molecular analytes, including hormones, antibodies, inflammatory markers, and defensive molecules. Although blood, serum, and plasma are traditionally considered the gold standard for measuring biomarkers, saliva is a viable and often equivalent alternate biosample. The procedures required to collect, ship, store, and eventually analyze blood for medical and dental purposes can be expensive, problematic, and physically intrusive. Comparatively, saliva carries the advantages of simple collection and can be performed by anyone without specialized training or equipment. The procedure for collection is non-invasive and pain-free, and samples are safer to handle, especially with collection devices that have preservative and stabilization solutions that neutralize pathogens and preserve the analytes of interest (Fig. 2). Once stabilized, samples can be shipped at room temperature, and storage is simple. Overall, the procedures for collecting, shipping, storing, and analyzing saliva are cheaper than those for blood and decrease the overall costs for patients and providers.
Fig. 1
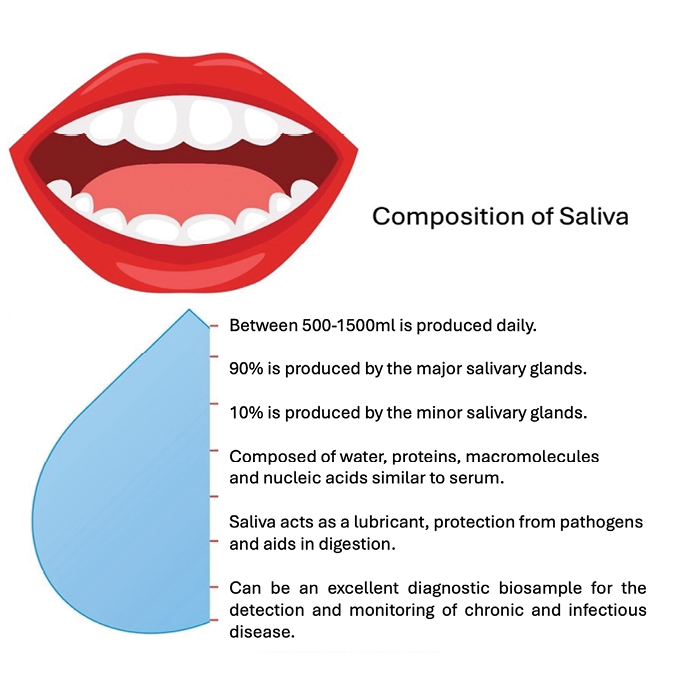
Fig. 2
Saliva can effectively be used in place of blood for various diagnostic purposes. The term “salivaomics” was initially coined by Professor DT Wong and others in 2008 and 20122 as a reflection of the ability of saliva to measure a broad range of essential macromolecules associated with health and disease. For example, saliva is a viable sample type for the screening and diagnosis of a variety of diseases such as cancer,3 heart disease and diabetes. Saliva is also an excellent sample type for the monitoring of drug therapy4 in depression and anxiety.5 Saliva can be used to determine infection status,6 as in respiratory disease or periodontal disease. The measurement of inflammation7 and markers of inflammation such as C reactive protein are commonly performed using saliva samples. Finally, several investigators have used saliva as a means to test for traumatic brain injury.8 Employing saliva as a medium for biomarker development and evaluation alleviates patient discomfort by providing a safe, non-invasive, and inexpensive method for disease detection and monitoring. The discovery and publication of saliva-based proteomics, microbiomics, metabolomics, genomics, and transcriptomics have opened the door to non-invasive discovery for clinical and translational applications in personalized medicine and dentistry.
Diagnostics
The ability of a health care practitioner to deliver effective care depends on their ability to accurately identify the cause of a patient’s problem or concern. In the medical model, more than 80% of decision-making is based on laboratory and diagnostic testing results.9,10 Over the last few decades, our understanding of the various disease mechanisms has increased and furthered our ability to make more accurate and specific diagnoses. These advancements have been mainly the result of improvements and expansion of molecular diagnostics and pathology. Prior to the COVID-19 pandemic, the use of molecular diagnostics and, in particular, a powerful technique called polymerase chain reaction (PCR) assay was underutilized. The use of PCR techniques to rapidly and accurately detect SARS-CoV-2 during the pandemic allowed the healthcare community to more broadly accept and adopt molecular diagnostics to better help their patients. Now, more healthcare providers are familiar with the technique, and the power of rapid, inexpensive, and accurate diagnostics is available to the medical and dental communities. A broad spectrum of molecular diagnostics and techniques provide critical additional information in disease detection, diagnosis, and prognosis. While the COVID-19 pandemic may have brought terms like PCR and molecular diagnostics into the mainstream, the field of diagnostics has steadily evolved behind the scenes to provide for: faster identification and treatment of infectious disease, increased antibiotic stewardship, improved management of chronic disease, advancement in point of care solutions, expanded options for direct-to-consumer testing, and improved maternal and fetal testing. Each of these advancements has highlighted the laboratory’s value to healthcare, particularly the unique opportunity for the dental community to reunite with medicine. The overall impact on population health is that molecular diagnostics in the dental community can allow for earlier treatment and better patient care, reduce or eliminate unnecessary and inadequate treatment, reduce overall healthcare costs, and reduce patient morbidity and mortality.
Periodontal applications
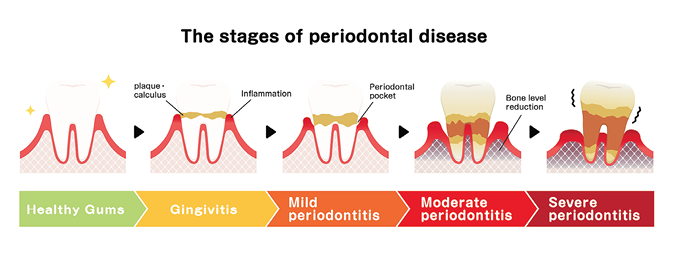
The most impactful application of diagnostics in the future of dentistry is the ability to detect the bacteria that are responsible for the development of gingivitis and periodontitis (Fig. 3). Periodontal disease is one of the most significant and pervasive public health concerns that can dramatically impact the development and progression of a variety of chronic diseases, such as cardiovascular disease, fatty liver disease, diabetes, neurodegeneration, and cancer. Additionally, periodontal disease has a significant impact on pregnancy risk and fetal health. The ability of a dental provider to conduct basic diagnostic testing can afford the ability to dramatically impact the welfare of every patient who visits the dentist. It is well known anecdotally that people see the dentist 2-3 times per year, whereas they visit their medical provider perhaps only once for a physical. An Agency for Healthcare Research and Quality study showed that 37% (121.2 million) of Americans visited a dentist and physician at least once a year. Another 9% (28.2 million) only visited a dentist11. Data from the CDC and the American Dental Association suggested that from 2019 to 2020, 62.7% of Americans saw a dentist, and 50% saw their dental provider at least twice.12
Fig. 3
Recent diagnostic improvements and developments include tests for the detection of bacteria that are most responsible for gingivitis, periodontal disease, and peri-implantitis. Data over the last several decades has identified the following organisms as having the most significance in dental medicine. To make reading easier, each organism is commonly referred to using their two-letter abbreviation. (Aggregatibacter actinomycetemcomitans abbreviated as Aa, Prevotella intermedia abbreviated as Pi, Campylobacter rectus abbreviated as Cr, Tannerella forsythia abbreviated as Tf, Treponema denticola abbreviated as Td, Fusobacterium nucleatum abbreviated as Fn, and Porphyromonas gingivalis abbreviated as Pg). Peri-implantitis is commonly associated with Pseudomonas aeruginosa abbreviated as Pa. Caries risk is often associated with the bacteria Streptococcus mutans abbreviated as Sm and Streptococcus sanguinis abbreviated as Ss is often a bacteria that is associated with a healthy oral cavity. Another very important test using saliva in the dental practice is for fungal co-infection arising from the yeast Candida albicans abbreviated as Ca. Additionally, there are several critically important viral pathogens Herpes simplex virus 1 and 2 abbreviated as HSV-1 and 2, Cytomegalovirus abbreviated as CMV, Epstein-Barr Virus abbreviated as EBV, and Human Papilloma Virus abbreviated as HPV. The report that is returned to the dentist or physician is a clear and concise analysis of the levels present of each of these organisms that may be contributing to oral disease in their patients (Fig. 4).
Fig. 4

In recent unpublished work from our laboratory, we established in a cohort of 3000 patients that co-infection between bacteria associated with periodontitis such as Aa, Pg and EBV and HSV-1 occurred greater than 50% of the time. Preliminary data suggested that the most aggressive oral bacteria, Aa and Pg, had a 60% and 58% incidence of herpesviral co-infection, respectively, consistent with previous literature.13-15 The relevance of this data is that there is a largely unrecognized synergy between oral bacteria and the herpesviruses we frequently have present in the oral cavity. By the time humans are into their teens, we are nearly 100% seropositive. This means that as these viruses come and go during our lifetime, they cooperate with oral bacteria to make oral and systemic diseases worse. This illustrates that without diagnostic testing, we do not see the problems lurking and, as a result, cannot fix the issues before more severe harm develops. Rapid and accurate detection of pathogens associated with oral disease is available more broadly. It can assist the dental provider in achieving more precise diagnosis and, most importantly, allow for more precision targeting of the root of the disease.
Systemic impact from the oral microbiome
Briefly, the systemic impact from the oral microbiome has been established in recent years. There is a clear association between chronic periodontitis and coronary artery disease.16,17 An association between neurogenerative disease and oral health has been previously established.18-20 Additionally, people with moderate to severe periodontitis have been found to have a higher risk of suffering from rheumatoid arthritis.21,22 Further, the association between periodontal disease and pregnancy risks has also been firmly established.23-25 Finally, people with diabetes and metabolic disease are at a higher risk of developing periodontal disease, and new research is suggesting that periodontal disease may be a risk factor for the development of diabetes.26-28
Saliva in the diagnosis and treatment of oral-systemic disease
Heart disease is the leading cause of death in the United States, and studies show a causal relationship between heart disease and gum disease.17 People with gum disease have an increased risk of having a heart attack, stroke, or other serious cardiovascular event.
The oral microbiome is the 2nd largest microbial community in the body, behind the gut. Resident microbes in the oral cavity play a critical role in homeostasis and pathogenesis. Data has shown that certain oral microbes, such as Pg, Fn, Td, and Aa, are enriched in patients with oral disease.29 Data further suggests that these same organisms are associated with systemic disease such as CVD, diabetes, and arthritis.17,21,28
Periodontitis induces systemic inflammation and contributes to the development of neurodegenerative disease. Chronic inflammation of the brain is a well-recognized cause and symptom of neurodegenerative disorders. Evidence suggests that systemic inflammation is a possible trigger for the development of neuroinflammation. P. gingivalis, for example, can travel from the mouth to the brain, and related gingipains can destroy brain neurons. Research led by scientists from the National Institute of Aging suggests that periodontal disease can cause chronic inflammation, which is associated with the development of Alzheimer’s disease and related dementias.30
Saliva diagnostics for the future
Saliva is an essential and relevant biological fluid with a wide range of research and clinical applications. It has a high potential to become the future of noninvasive diagnostics. A broad array of diseases can be diagnosed, prevented, and monitored via saliva, including cardiovascular, neurological, gastrointestinal, hormonal, metabolic, and perhaps most importantly, periodontal disorders.
As the field of saliva diagnostics continues to grow and expand with greater accuracy, specificity, and sensitivity, we will see saliva become the preferred sample type for various in-office and remote applications. To reflect on Charles Mayo, MD, the future of medicine, dentistry, and disease prevention is in the hands of the dentist and oral provider. 
Oral Health welcomes this original article.
Disclaimer: DV is the chief scientific officer for Spectrum Solutions, which manufactures and distributes saliva collection kits and performs molecular diagnostics for the medical and dental communities.
References
- Humphrey SP, Williamson RT. A review of saliva: normal composition, flow, and function. J Prosthet Dent. Feb 2001;85(2):162-9. doi:10.1067/mpr.2001.113778
- Wong DT. Salivaomics. J Am Dent Assoc. Oct 2012;143(10 Suppl):19S-24S. doi:10.14219/jada.archive.2012.0339
- Nguyen TA, Chen RH, Hawkins BA, et al. Can we Predict Drug Excretion into Saliva? A Systematic Review and Analysis of Physicochemical Properties. Clin Pharmacokinet. Aug 2024;63(8):1067-1087. doi:10.1007/s40262-024-01398-9
- Chundru VNS, Nirmal RM, Srikanth B, Bojji M, Midhun N, Lakshmi BJ. Salivaomics for Oral Cancer Detection: An Insight. J Pharm Bioallied Sci. Jun 2021;13(Suppl 1):S52-S56. doi:10.4103/jpbs.JPBS_529_20
- Kugler J. [Emotional status and immunoglobulin A in saliva–review of the literature]. Psychother Psychosom Med Psychol. Jun 1991;41(6):232-42. Emotionale Befindlichkeit und Immunglobulin A im Speichel–Eine Literaturubersicht.
- Teles FRF, Chandrasekaran G, Martin L, et al. Salivary and serum inflammatory biomarkers during periodontitis progression and after treatment. J Clin Periodontol. Aug 5 2024;doi:10.1111/jcpe.14048
- Relvas M, Mendes-Frias A, Goncalves M, et al. Salivary IL-1beta, IL-6, and IL-10 Are Key Biomarkers of Periodontitis Severity. Int J Mol Sci. Aug 1 2024;25(15)doi:10.3390/ijms25158401
- Kvist M, Valimaa L, Harel A, Malmi S, Tuomisto A. Glycans as Potential Diagnostic Markers of Traumatic Brain Injury in Children. Diagnostics (Basel). Jun 26 2023;13(13)doi:10.3390/diagnostics13132181
- de Araujo CS, da Silva ACL, Freitas-Fernandes LB, Maia LC, da Silva Fidalgo TK, Valente AP. Untargeted stimulated and unstimulated salivary metabolomics and saliva flow rate in children. Clin Oral Investig. Aug 17 2024;28(9):489. doi:10.1007/s00784-024-05883-0
- van Moll C, Egberts T, Wagner C, Zwaan L, Ten Berg M. The Nature, Causes, and Clinical Impact of Errors in the Clinical Laboratory Testing Process Leading to Diagnostic Error: A Voluntary Incident Report Analysis. J Patient Saf. Dec 1 2023;19(8):573-579. doi:10.1097/PTS.0000000000001166
- O’Kane M. The reporting, classification and grading of quality failures in the medical laboratory. Clin Chim Acta. Jun 2009;404(1):28-31. doi:10.1016/j.cca.2009.03.023
- Richard Manski D, MBA, PhD, Frederick Rohde, MA, and Timothy Ricks,, DMD M, FICD. Trends in the Number and Percentage of the Population with Any Dental or Medical Visits, 2003–2018. Updated October 2021. https://meps.ahrq.gov/data_files/publications/st537/stat537.pdf
- Amy E. Cha PD, M.P.H., and Robin A. Cohen, Ph.D. NCHS Data Brief.
- Slots J, Kamma JJ, Sugar C. The herpesvirus-Porphyromonas gingivalis-periodontitis axis. J Periodontal Res. Jun 2003;38(3):318-23. doi:10.1034/j.1600-0765.2003.00659.x
- Saygun I, Kubar A, Ozdemir A, Yapar M, Slots J. Herpesviral-bacterial interrelationships in aggressive periodontitis. J Periodontal Res. Aug 2004;39(4):207-12. doi:10.1111/j.1600-0765.2004.00728.x
- Fang T, Liu L, Mao S, Jiang Z, Cao Y, Pan J. Association between virus infection and periodontitis: Evidence from the National Health and Nutrition Examination Survey 2009-2014. J Med Virol. Jul 2024;96(7):e29784. doi:10.1002/jmv.29784
- Daily ZA, Al-Ghurabi BH. Accuracy of salivary biomarkers in the diagnosis of periodontal status and coronary heart disease. J Med Life. Apr 2024;17(4):442-448. doi:10.25122/jml-2023-0264
- Bale BF, Doneen AL, Vigerust DJ. High-risk periodontal pathogens contribute to the pathogenesis of atherosclerosis. Postgrad Med J. Apr 2017;93(1098):215-220. doi:10.1136/postgradmedj-2016-134279
- Han J, Liu Y, Guo X, Gao G, Wu Q. Research Trends in the Comorbidity Between Periodontitis and Neurodegenerative Diseases. Int Dent J. Aug 12 2024;doi:10.1016/j.identj.2024.07.1212
- Visentin D, Gobin I, Maglica Z. Periodontal Pathogens and Their Links to Neuroinflammation and Neurodegeneration. Microorganisms. Jul 18 2023;11(7)doi:10.3390/microorganisms11071832
- Wu YF, Lee WF, Salamanca E, et al. Oral Microbiota Changes in Elderly Patients, an Indicator of Alzheimer’s Disease. Int J Environ Res Public Health. Apr 15 2021;18(8)doi:10.3390/ijerph18084211
- Rak D, Kulloli AM, Shetty SK, et al. Correlation between rheumatoid arthritis and chronic periodontitis: a systematic review and meta-analysis. Minerva Dent Oral Sci. Jun 13 2024;doi:10.23736/S2724-6329.23.04891-X
- Raittio E, Nascimento GG, Lopez R, Baelum V. Exploring the Bidirectional Relationship Between Periodontitis and Rheumatoid Arthritis in a Large Danish Cohort. ACR Open Rheumatol. Jul 5 2024;doi:10.1002/acr2.11718
- Sharma M, Sunda U, Dubey P, Tilva H. From Oral Health to Obstetric Outcomes: A Comprehensive Review of Periodontal Disease and Its Implications for Preeclampsia. Cureus. Jun 2024;16(6):e62995. doi:10.7759/cureus.62995
- Wen P, Li H, Xu X, et al. A prospective study on maternal periodontal diseases and neonatal adverse outcomes. Acta Odontol Scand. Jun 11 2024;83:348-355. doi:10.2340/aos.v83.40836
- Gilani SI, Niaz A, Afridi S. Maternal periodontitis as a risk factor for preterm birth: A cross-sectional study. J Dent Res Dent Clin Dent Prospects. Winter 2024;18(1):72-76. doi:10.34172/joddd.40860
- Wei X, Zhang X, Chen R, et al. Impact of periodontitis on type 2 diabetes: a bioinformatic analysis. BMC Oral Health. May 29 2024;24(1):635. doi:10.1186/s12903-024-04408-1
- Chung YL, Lee JJ, Chien HH, Chang MC, Jeng JH. Interplay between diabetes mellitus and periodontal/pulpal-periapical diseases. J Dent Sci. Jul 2024;19(3):1338-1347. doi:10.1016/j.jds.2024.03.021
- Vlachou S, Loume A, Giannopoulou C, Papathanasiou E, Zekeridou A. Investigating the Interplay: Periodontal Disease and Type 1 Diabetes Mellitus-A Comprehensive Review of Clinical Studies. Int J Mol Sci. Jul 2 2024;25(13)doi:10.3390/ijms25137299
- Morrison AG, Sarkar S, Umar S, Lee STM, Thomas SM. The Contribution of the Human Oral Microbiome to Oral Disease: A Review. Microorganisms. Jan 26 2023;11(2)doi:10.3390/microorganisms11020318
- Beydoun MA, Beydoun HA, Hossain S, El-Hajj ZW, Weiss J, Zonderman AB. Clinical and Bacterial Markers of Periodontitis and Their Association with Incident All-Cause and Alzheimer’s Disease Dementia in a Large National Survey. J Alzheimers Dis. 2020;75(1):157-172. doi:10.3233/JAD-200064
About the Author

Dr. Vigerust received degrees from the University of Texas at El Paso, Texas Tech University, and Vanderbilt University SOM. He is faculty at Vanderbilt School of Medicine and Texas Tech University School of Dental Medicine, specializing in infectious disease, immunology, and pathology. He is also the chief scientific officer at Spectrum Solutions.