
The World Health Organization (WHO) Global Oral Health Report Towards universal health coverage for oral health by 2030 states: “No other disease group affects humanity across the life cycle and across all countries in the way that oral disease do”.1 Indeed, oral diseases are more prevalent globally than mental health disorders, cardiovascular diseases, diabetes mellitus, chronic respiratory diseases, and cancers combined. Worldwide, approximately $710 B is spent on treatment of oral diseases with a resulting loss of $323 B in productivity.2 In Canada, available statistics showed that oral diseases account for productivity losses of over $1 billion per year.3 Because of global population growth and increased tooth retention throughout the age span, these numbers are expected to grow substantially, with high global economic impact. To address the major oral health problem globally, the abovementioned WHO report advocates a paradigm shift in oral health policy planning “…from a conventional model of restorative dentistry towards a promotive and preventive model”.1 Several preventive oral health therapeutics have been developed and tested over the years.4-6 Many of these approaches suffer from lack of high-quality laboratory evidence or rigorous clinical trials.
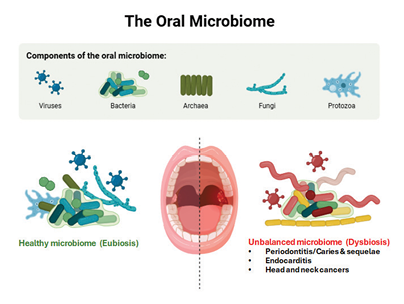
The oral microbiome and its modulation offer a powerful target for next-generation preventive oral care. Known as “the ecological community of commensal, symbiotic, and pathogenic microorganisms that literally share our body space”,7 the oral microbiome contains a community of microorganisms, including bacteria, fungi, viruses, archaea, and protozoa, and plays a major role in oral diseases8 (Fig. 1). In health, a complex equilibrium exists between the resident species in the oral cavity to maintain a healthy, or symbiotic state. Several factors, however, including diet, poor oral hygiene, tobacco smoking, and certain medications, among others, can disrupt this weak homeostatic balance to cause a dysbiotic state with potential implications on both oral and systemic health.9 Oral diseases have been consistently linked to dysbiosis of the oral microbiome, with a single or few species predominating and an associated increased risk of disease that drives inflammation and tissue damage10 (Fig. 1). For instance, increased colonization by mutans streptococci (MS), mainly of the species Streptococcus mutans and Streptococcus sobrinus, with significant acidogenic and aciduric properties, and oftentimes in synergy with other bacteria and fungi, e.g., Candida albicans,11 is primarily responsible for caries formation.12,13 The localized metabolic production of acids of these microorganisms decays the tooth through the demineralization of calcium and phosphorous from the enamel and dentin over several months, resulting in caries lesions.14
Fig. 1
Addressing the microbiome imbalance in the oral cavity requires targeted approaches, one that can actively recalibrate the microbiome toward a health-associated state. Pharmabiotics—defined as bacterial cells of human origin or their products with a proven pharmacological role in health and disease15—represent one such promising strategy. A key class of pharmabiotics is probiotics, defined as “live microorganisms which when administered in adequate amounts confer a health benefit on the host”.16 By reinforcing epithelial barriers, competing with pathogens, inhibiting harmful bacteria, and modulating the host immune response, probiotics provide mechanisms that directly counter microbiome-driven pathologies.17 Depending on the strain, delivery, and host context, these effects can be local within the oral cavity or systemic. A diverse range of bacteria—including acidogenic Lactobacilli, Bifidobacteria, and Streptococci—have demonstrated the capacity to rebalance dysbiotic communities and mitigate oral disease.18-22
Our research team comprising a dental clinician/scientist and an oral microbiologist at the University of Toronto Faculty of Dentistry collaboratively undertook a mission: To discover new probiotic strains specifically from the oral cavity to improve oral health. We focused on identifying strains of the oral commensal Streptococcus salivarius (Fig. 2) for a more sustainable and longer-term probiotic benefit compared to species from external sources (such as dairy products). S. salivarius is a species known to offer significant value to oral health. A pioneer colonizer of the oral cavity and a predominant member of the native microbiota that persists throughout the human life-span,23,24 S. salivarius are well adapted to growth in the mouth and oral biofilms. Importantly, they are known to produce anti-microbial substances against other bacteria.25 Additionally, many S. salivarius
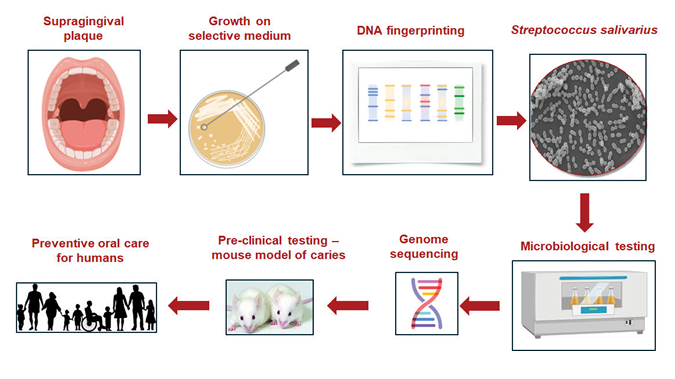
strains are regarded as having “generally recognized as safe” (GRAS) status in the USA25 and as natural health products by Health Canada.26 This means that they can be used in food or health products for human consumption. We focused specifically on identifying S. salivarius strains from the dental plaque of caries-free children, with the hypothesis that the highly structured plaque biofilm of caries-free children would harbor a high proportion of S. salivarius strains with strong inhibitory activities against cariogenic bacteria. As the inhibitory activities against MS are observed in only ~1% of S. salivarius strains,27,28 we followed a systematic screening approach to identify these potential candidates (outlined in Fig. 3). Each step of the experimental screen was designed, and validated, to eliminate less suitable options and refine the pool of strains to those showing the strongest inhibitory activities. Presumptive S. salivarius strains were first identified from the harvested plaque samples by cultivation in culture broth specific for their growth. Any positive clones were then “fingerprinted” to verify that they contained S. salivarius gene sequences and produced general inhibitory substances. Positive clones were further tested and ranked by their ability to show the strongest inhibitory activities against the major cariogenic group of bacteria, the MS. Those that met the pre-set criteria were selected for a more thorough testing via in vitro means, such as their ability to inhibit the growth of biofilms from monocultures of the major cariogenic S. mutans12 and dual cultures containing both S. mutans and C. albicans. Additionally, we also tested the ability of the select group of candidates for their ability to bind salivary-coated hydroxyapatite (enamel substitute) and dissected rodent incisors.
Fig. 2

Fig. 3
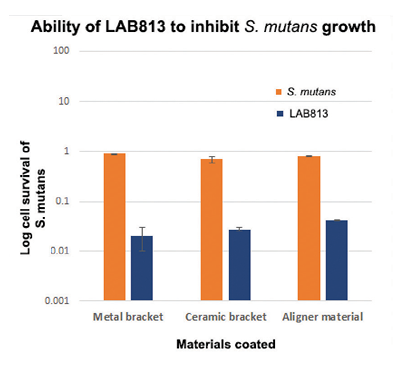
We screened approximately 600 S. salivarius strains from the supragingival plaque samples harvested from the buccal surfaces of teeth of 60 healthy children. Several potential candidates were identified and characterized. In vitro experiments showed that the killing efficiency of at least five probiotic strains against S. mutans biofilms approached 90% (as measured by their ability to inhibit the growth of S. mutans; Fig. 4). One candidate, LAB813, was especially efficient, with its inhibitory activities against the biofilms approaching 99%. Significantly, when grown on materials used in orthodontic treatment (such as metal and ceramic brackets and plastic aligner materials made of thermoplastic polyurethane/copolyester), the killing efficiency of LAB813 of S. mutans biofilms approached 99%29 (Fig. 4; orange bars). Indeed, when LAB813 was pre-formed on these materials, the killing efficiency against S. mutans increased further, approaching 99.9% (Fig. 4; blue bars). Furthermore, and of great clinical significance, is our finding that it only takes 2 hours for LAB813 to kill 99% of S. mutans biofilm cells, as shown in a time killing kinetic study. The inhibitory abilities of LAB813 are likely to be mediated via the production of anti-inhibitory molecules such as peptides and other compounds, a hypothesis that was strengthened after a thorough analysis of the sequenced LAB813 revealed several potential anti-bacterial peptide sequences.30
Fig. 4
Our in vitro data gives substantial support for commercializing these probiotic products as preventive oral health products for use in humans. Currently, we are actively engaged in planning and/or conducting activities that will move our products into the market. For example, we are planning a series of pre-clinical studies on mice, a powerful way to test the efficacy and safety of therapeutics before translating to humans. Fortunately, in dental research, there exists a well-established rodent model of caries where the formation of caries is induced in rodents by exposing the animals to a high sugar diet combined with inoculation of S. mutans.31 We plan to take advantage of this model and combine our studies with the powerful and highly sensitive micro-computed tomographic technique to generate ultrahigh-resolution images of rodent teeth to determine the impact of our probiotic administration on tooth cavitation (Fig. 5). Another immediate goal of our research is to develop innovative probiotic products that can be readily incorporated into current oral healthcare and treatment modalities for use in the oral cavity. For example, we are actively working with bioengineers and industrial partners to develop delivery formats including powder forms, tablets, and microcapsules (Fig. 6). We are also actively involved in conducting manufacturing, stability and efficacy testing of our finished products, conducting clinical trials to validate claims and safety, obtaining federal regulatory approval for human use, etc.
Fig. 5
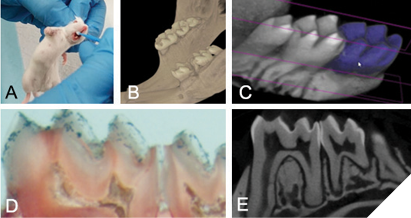
Fig. 6

We are excited and optimistic about the potential of our probiotic products to transform preventive oral health strategies for individuals across all age groups and for oral healthcare providers worldwide. Importantly, they hold promise for making a meaningful difference in the lives of those most at risk of dental caries, including Indigenous individuals, communities, and peoples—helping close critical gaps in oral health. 
Oral Health welcomes this original article.
Acknowledgement: We would like to acknowledge the significant help and contribution of numerous students (undergraduate, dental and graduate students, dental specialty students), postdoctoral fellows and research associates, who have worked tirelessly in our research labs at the Faculty of Dentistry, University of Toronto over the years. Credit also goes to dental students from Tokyo Medical and Dental University (now Institute of Science Tokyo) who participated in research in our labs as part of the summer research program.
We also extend our thanks to the federal, private and institutional agencies that fund our work – Canadian Institutes of Health Research, Natural Sciences and Engineering Research Council of Canada, Canada Foundation for Innovation, Align Technology Research Award Program, the Connaught Innovation Award Program and the University of Toronto Innovations and Partnerships Office.
References
- WHO. Global oral health status report: towards universal health coverage for oral health by 2030. World Health Organization. Geneva: 2022
- Jevdjevic M, Listl S. Global, Regional, and Country-Level Economic Impacts of Oral Conditions in 2019. J Dent Res 2025, 104: 17-21
- Hayes A, Azarpazhooh A, Dempster L, Ravaghi V, Quiñonez C. Time loss due to dental problems and treatment in the Canadian population: analysis of a nationwide cross-sectional survey. BMC Oral Health 2013, 13: 17
- Zhang OL, Niu JY, Yin IX, Yu OY, Mei ML, Chu CH. Bioactive Materials for Caries Management: A Literature Review. Dent J (Basel) 2023, 11
- de Carvalho LFDC, Zanatta RF. Editorial: Technological innovations for improved prevention and diagnosis of oral disease. Front Oral Health 2024, 5: 1481890
- Cui H, You Y, Cheng GW, Lan Z, Zou KL, Mai QY, Han YH, et al. Advanced materials and technologies for oral diseases. Sci Technol Adv Mater 2023, 24: 2156257
- Lederberg J, McCray AT. ‘Ome Sweet’ Omics – A genealogical treasury of words. The Scientist 2001, 15:8.
- Kilian M, Chapple IL, Hannig M, Marsh PD, Meuric V, Pedersen AM, Tonetti MS, et al. The oral microbiome – an update for oral healthcare professionals. Br Dent J 2016, 221: 657-666
- Santacroce L, Passarelli PC, Azzolino D, Bottalico L, Charitos IA, Cazzolla AP, Colella M, et al. Oral microbiota in human health and disease: A perspective. Exp Biol Med (Maywood) 2023, 248: 1288-1301
- Rajasekaran JJ, Krishnamurthy HK, Bosco J, Jayaraman V, Krishna K, Wang T, Bei K. Oral Microbiome: A Review of Its Impact on Oral and Systemic Health. Microorganisms 2024, 12
- Falsetta ML, Klein MI, Colonne PM, Scott-Anne K, Gregoire S, Pai CH, Gonzalez-Begne M, et al. Symbiotic relationship between Streptococcus mutans and Candida albicans synergizes virulence of plaque biofilms in vivo. Infect Immun 2014, 82: 1968-1981
- Loesche WJ. Role of Streptococcus mutans in human dental decay. Microbiol Rev 1986, 50: 353-380
- Burne RA. Oral streptococci… products of their environment. J Dent Res 1998, 77: 445-452
- Lingström P, van Houte J, Kashket S. Food starches and dental caries. Crit Rev Oral Biol Med 2000, 11: 366-380
- Hill C. Probiotics and pharmabiotics: alternative medicine or an evidence-based alternative? Bioeng Bugs 2010, 1: 79-84
- Food and Agriculture Organization of the United Nations, Organization WH. Guidelines for the evaluation of probiotics in food: Report of a Joint FAO/WHO Working Group on Drafting Guidelines for the Evaluation of Probiotics in Food (Food and Nutrition Paper No. 85). In: FAO/WHO ed. https://wwwfaoorg/3/a0512e/a0512epdfed. 2001
- Latif A, Shehzad A, Niazi S, Zahid A, Ashraf W, Iqbal MW, Rehman A, et al. Probiotics: mechanism of action, health benefits and their application in food industries. Front Microbiol 2023, 14: 1216674
- Meurman JH. Probiotics: do they have a role in oral medicine and dentistry? Eur J Oral Sci 2005, 113: 188-196
- Twetman S, Keller MK. Probiotics for caries prevention and control. Adv Dent Res 2012, 24: 98-102
- Dierksen KP, Moore CJ, Inglis M, Wescombe PA, Tagg JR. The effect of ingestion of milk supplemented with salivaricin A-producing Streptococcus salivarius on the bacteriocin-like inhibitory activity of streptococcal populations on the tongue. FEMS Microbiol Ecol 2007, 59: 584-591
- Burton JP, Drummond BK, Chilcott CN, Tagg JR, Thomson WM, Hale JD, Wescombe PA. Influence of the probiotic Streptococcus salivarius strain M18 on indices of dental health in children: a randomized double-blind, placebo-controlled trial. J Med Microbiol 2013, 62: 875-884
- Di Pierro F, Colombo M, Zanvit A, Risso P, Rottoli AS. Use of Streptococcus salivarius K12 in the prevention of streptococcal and viral pharyngotonsillitis in children. Drug Healthc Patient Saf 2014, 6: 15-20
- Carlsson J, Grahnén H, Jonsson G, Wikner S. Early establishment of Streptococcus salivarius in the mouth of infants. J Dent Res 1970, 49: 415-418
- Favier CF, Vaughan EE, De Vos WM, Akkermans AD. Molecular monitoring of succession of bacterial communities in human neonates. Appl Environ Microbiol 2002, 68: 219-226
- Wescombe PA, Hale JD, Heng NC, Tagg JR. Developing oral probiotics from Streptococcus salivarius. Future Microbiol 2012, 7: 1355-1371
- Health Canada. Natural health products. https://www.canada.ca/en/health-canada/services/drugs-health-products/natural-non-prescription.html. ed. 2025
- Tagg JR, Harold LK, Jain R, Hale JDF. Beneficial modulation of human health in the oral cavity and beyond using bacteriocin-like inhibitory substance-producing streptococcal probiotics. Front Microbiol 2023, 14: 1161155
- Ogawa A, Furukawa S, Fujita S, Mitobe J, Kawarai T, Narisawa N, Sekizuka T, et al. Inhibition of Streptococcus mutans biofilm formation by Streptococcus salivarius FruA. Appl Environ Microbiol 2011, 77: 1572-1580
- Gong SG, El-Shennawy S, Choudhary P, Dufour D, Lévesque CM. Antimicrobial activity of probiotic Streptococcus salivarius LAB813 on in vitro cariogenic biofilms. Arch Oral Biol 2023, 154: 105760
- Gong SG, Chan Y, Lévesque CM. Complete Genome Sequence of Megaplasmid-Bearing Streptococcus salivarius Strain LAB813, Isolated from the Dental Plaque of a Caries-Free Child. Microbiol Resour Announc 2019, 8
- Bowen WH. Rodent model in caries research. Odontology 2013, 101: 9-14
About the author

Dr. Gong is a scientist, educator, and dental clinician (orthodontist). Through her affiliation with the University of Toronto, she has contributed to studies in oral biology, orthodontic tooth movement and the development of probiotics for oral health.

Dr. Céline Lévesque is a professor and microbiologist at the University of Toronto. Her research focuses on oral bacteria, biofilm formation, and developing new approaches to prevent dental caries. She leads collaborative projects and mentors students in microbiome science, combining lab work with global partnerships to improve dental health.
