
An ecology-first approach that supports nitrate-reducing commensals, improves salivary pH, and enhances nitric oxide bioavailability, without relying on broad-spectrum antimicrobial suppression.
A practical shift from “kill the biofilm” to “support oral function”
For decades, everyday dentistry has leaned heavily on suppression, antimicrobial rinses, antibacterial pastes, and repeated attempts to “control” oral biofilm by reducing bacterial load. Those tools still have a role. But the modern oral-systemic conversation is increasingly shaped by a more nuanced idea: the mouth is not simply a site to disinfect. It is a living ecosystem where the composition and function of the oral microbiome can influence both local inflammatory tone and host physiology.
Prebiotic nitrate lozenges and chewing gum represent a different kind of intervention. They are designed not to sterilize the mouth, but to support a beneficial biochemical capability carried out by commensal oral bacteria: converting nitrate (NO3-) to nitrite (NO2-), a key step in the nitrate-nitrite-nitric oxide (NO) pathway. That pathway begins in the oral cavity and continues through the gastrointestinal tract and circulation. When it works well, it can support nitric oxide signaling, vascular function, and host homeostasis, while also shifting oral chemistry in ways that can be more compatible with health.
Humans are biologically “limited” here and that’s the point
A useful way to understand nitrate biology is to acknowledge a simple constraint: humans do not complete nitrate reduction effectively without microbial help. The enterosalivary nitrate-nitrite-NO pathway depends on oral bacteria as the first step. In this model, the oral microbiome is not merely something to manage, it is a functional partner that enables physiology the host cannot fully execute alone.
Humans have a built-in limitation here: we don’t directly “complete” nitrate biology on our own. Instead, we rely on a symbiotic backup system, the enterosalivary nitrate-nitrite-NO pathway, where oral bacteria reduce dietary nitrate (NO3-) to nitrite (NO2-), enabling downstream nitric oxide signaling and related intermediates that support homeostasis. This is a useful way to understand why a microbiome-supportive strategy matters: if the oral microbiome is doing essential biochemical work for the host, then broad antimicrobial suppression can unintentionally disrupt a beneficial function, not just “bad bacteria.”
Clinical observations reinforce that point. Antiseptic mouthwashes have been reported to reduce oral nitrate reduction and lower nitrite availability in controlled studies, which can blunt aspects of nitrate-related physiology. In other words, an antimicrobial-first routine may compromise the very symbiosis that links oral microbial function to nitric oxide bioavailability and broader systemic resilience.
Chairside reframe
Don’t erase the pathway you’re trying to support.
If nitrate-reducing commensals are a functional part of host NO biology, routine indiscriminate antimicrobial exposure can reduce nitrate to nitrite conversion and nitrite availability. Use antiseptics deliberately, when indicated, rather than as a default, chronic strategy.
What is “prebiotic nitrate” in dentistry?
Prebiotic nitrate therapy uses inorganic nitrate (NO3-), the same nutrient found in leafy greens and beets, as a selective substrate for nitrate-reducing commensal bacteria. In dental terms, “prebiotic” means the intent is to favor beneficial microbial function rather than suppress bacteria broadly.
This approach is best framed as:
- Site-specific: the oral cavity is the intended site of the key conversion step, NO3- to NO2-.
- Function-forward: the goal is to support a microbial capability tied to host physiology, nitric oxide biology, and oral chemistry, including pH trends.
- Adjunctive: it complements, not replaces, mechanical plaque disruption, periodontal therapy, fluoride strategies, and risk-based prevention.
Many formulations are designed to be used topically and repeatedly, often paired with supportive cofactors. From a practical standpoint, the defining feature for dentistry is the same: making the oral conversion step more likely to occur consistently in real patients.
Fig. 1
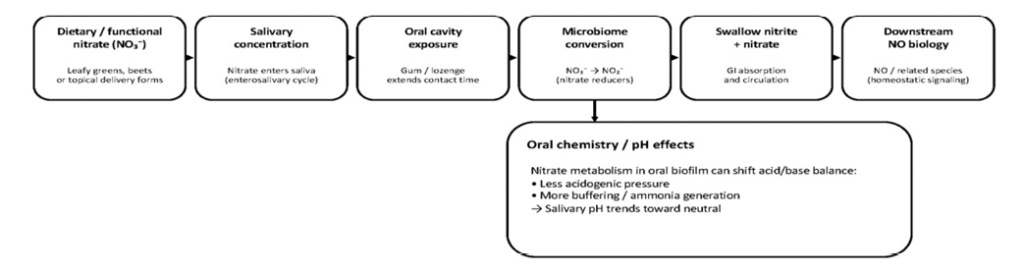
Why delivery format matters; “hang-time” is the intervention
If oral bacteria are the gatekeeper, then contact time in the mouth becomes a controllable clinical variable. A core hypothesis presented by Dr. Shawn J. Green in a Nature Biofilm is that oral and systemic outcomes may improve by extending the residence, “hang-time,” of prebiotic nitrate formulations in the oral cavity, particularly in individuals with reduced salivary flow or cardiometabolic patterns associated with dysbiosis and low NO bioavailability.¹
This is where slow-release chewing gum and lozenges become clinically relevant delivery systems:
- Gum can extend oral exposure through chewing time and increased salivary flow, reinforcing repeated exposure in daily routines.
- Lozenges can extend exposure through slow dissolution, serving patients who prefer not to chew or want longer contact without mastication.
Within the MyFitStrip patented formulation ecosystem, prebiotic nitrate has been developed in both gum and lozenge formats specifically to support this hang-time principle. In a journal setting, these are best described as delivery examples, not product claims, and paired with clear disclosure.
Mechanism, nitrate-nitrite-NO biology and oral pH
The clinical appeal of this pathway is that it links a familiar preventive concept, supporting oral conditions compatible with health, with a broader systemic signaling pathway.
1) Oral microbiome conversion, NO3- to NO2-
Nitrate reduction is not just chemistry; it is microbial function. When nitrate-reducing bacteria are present and active, nitrite becomes available for downstream NO-related intermediates. When that community is disrupted, by dysbiosis, low salivary flow, or repeated antiseptic suppression, the pathway can underperform.
2) Oral chemistry and pH trends
Beyond NO signaling, nitrate metabolism can influence oral chemistry in ways that are relevant to daily dentistry. Mechanistic work suggests that nitrate exposure can shift biofilm metabolism away from strongly acidogenic conditions, supporting a salivary environment that trends more neutral. For patients, this is an intuitive concept: the goal is not simply “fewer bacteria,” but a healthier, less inflammatory and less acidogenic oral environment.
3) Local tissues and systemic signaling
NO-related signaling is tied to vascular tone, endothelial function, and inflammatory regulation. Dentistry does not need to overstate systemic claims to acknowledge a clinically meaningful point: the mouth can influence a systemic pathway through microbiome-enabled chemistry.
What the emerging evidence suggests
The broader direction of evidence supports continued clinical investigation.
- Biofilm and mechanistic studies support the plausibility that nitrate can act as a selective substrate that favors nitrate-reducing commensals and shifts microbial community behavior.
- Early human observations and recently completed controlled studies using prebiotic nitrate delivery formats, including chewing gum, point toward improvements in oral inflammatory tone and oral chemistry consistent with the mechanistic model.
- Taken together, the most defensible conclusion for a dental readership is that this pathway is coherent, clinically actionable from a delivery standpoint, and worthy of broader, well-designed study, particularly as an adjunctive tool.
Clinical relevance, where this may fit in practice
Prebiotic nitrate delivery is best positioned as a risk-based adjunct in patients where microbial function, salivary chemistry, and inflammatory tone are central challenges.
Potential practice-fit scenarios include:
- Patients with recurring gingival inflammation despite reasonable home care
- High-frequency snacking patterns or other caries-risk profiles where pH and oral chemistry are persistent drivers
- Xerostomia or low-flow states where salivary dynamics are compromised
- Patients using antiseptic mouthwash chronically as a default habit rather than as a targeted, time-limited intervention
The goal is not to replace plaque control, but to support a microbiome-enabled pathway that may improve the oral environment in a way patients can realistically sustain.
Practical implementation, simple and sustainable
Chewing gum
- Use at consistent daily anchors; for example, after meals, after brushing or before bed.
- Encourage meaningful chewing time rather than quick use.
- Avoid immediately following with antiseptic mouthwash.
Lozenges
- Allow slow dissolution to extend oral contact time.
- Use daily, after eating, drinking, or brushing, or before sleeping.
- Consider for patients who prefer non-chewing formats or need longer contact without mastication.
The primary implementation principle is consistency: repeated, practical exposures are more aligned with microbiome function than occasional “hero dosing.”
Conclusion
Prebiotic nitrate lozenges and gum represent a promising, practice-friendly way to support an oral microbial function that the host relies on for nitrate-nitrite-NO biology. The clinical message is straightforward: if commensal bacteria help run important chemistry that influences both oral conditions and nitric oxide bioavailability, then dentistry should consider when stronger antimicrobials are truly needed, and when supporting beneficial microbial function may be the smarter long-term lever for oral and systemic health.7,8
As the profession continues to move toward prevention models grounded in biofilm behavior, salivary chemistry, and host-microbe symbiosis, microbiome-supportive nitrate delivery formats designed for oral hang-time deserve serious clinical attention and rigorous study. 
Oral Health welcomes this original article.
Disclosure: The author(s) have a financial relationship with MyFitStrip and/or are involved in the development of prebiotic nitrate lozenge and chewing gum formulations discussed as examples of delivery formats. This article is educational and does not reproduce unpublished clinical outcome data, which will be reported separately through peer-reviewed publication.
References
- Green J, Green SJ. Topical prebiotic nitrate: can extending the “hang-time” in the mouth improve oral-vascular health outcomes? npj Biofilms and Microbiomes. 2024. doi:10.1038/s41522-024-00527-3.
- Lundberg JO, Carlström M, Weitzberg E. Metabolic effects of dietary nitrate in health and disease. Cell Metabolism. 2018.
- Hezel MP, Weitzberg E. The oral microbiome and nitric oxide homeostasis. Oral Diseases. 2015.
- Rosier BT, et al. Nitrate as a potential prebiotic for the oral microbiome. Scientific Reports. 2020.
- Doel JJ, et al. Protective effect of salivary nitrate and microbial nitrate reductase activity against caries. European Journal of Oral Sciences. 2004.
- Li L, et al. Salivary nitrate and oral acidity: implications for oral biofilm behavior. Oral Microbiology and Immunology. 2007.
- Methods and compositions for alleviating respiratory dysfunction SJ Green – US Patent 11,844,811, 2023
- Green, SJ. Letter by Green RegardingArticle “Small Amounts of Inorganic Nitrate or Beetroot Provide Substantial Protection From Salt-Induced Increases in Blood Pressure. J Hypertension, doi:10.1161/https:// www.ahajournals.org/doi/abs/10.1161/ HYPERTENSIONAHA.119.13189
About the author

William R. Sotka, MA, is Director and Founding Member of MyFitStrip LLC and Global Operations Director at Hypertension Diagnostics Research. His work supports clinical adoption of nitric oxide science in oral and vascular health through saliva nitrate, nitrite, and pH interpretation and prebiotic nitrate lozenge and gum formats that extend oral nitrate hang time. He has co-authored peer-reviewed publications and supported the University of Maryland School of Dentistry trial NCT06029283.
