Management of alveolar defects is a common challenge in dental practice, particularly when preparing for implants or prosthetic restorations. A strong bone and soft tissue foundation is crucial to achieving predictable esthetic and functional results.1
Ridge augmentation can be achieved through block bone grafting or guided bone regeneration (GBR), utilizing various graft materials, including autogenous (patient’s own), allogeneic (cadaveric), xenogeneic (animal-derived), or alloplastic (synthetic) bone options. While autogenous bone has traditionally been the gold standard due to its ability to support bone growth, it requires a second surgical site, increasing patient discomfort.2 Guided bone regeneration (GBR) procedures address this by employing barrier membranes to direct bone growth and prevent soft tissue invasion. They are often used with in conjunction with the graft materials such as allogeneic bone, but allogeneic bone predominantly provides only a scaffold for bone regeneration.3
Incorporating regenerative medicine techniques can further enhance the body’s natural healing process. In one approach using Plasma Rich Growth Factors (PRGF), the patient’s own platelets and growth factors are concentrated to accelerate bone healing.4 By harnessing the body’s own regenerative abilities, PRGF can be used alongside various grafting materials to improve the effectiveness of ridge augmentation and reduce healing time.
History of PRGF
The use of platelet concentrates in medicine has its roots stemming from the 1970s and 1980s, emerging from hematology research and transfusion medicine.5 In 1987, Ferrari et al. reported the first clinical use of platelet-rich plasma (PRP) during open-heart surgery to minimize bleeding, laying the groundwork for future therapeutic applications.6 By the 1990s, PRP found its way into oral surgery, demonstrating its ability to accelerate bone regeneration, particularly in oral and maxillofacial surgery procedures.7 PRP is commonly prepared using rapid centrifugation at high G-forces to concentrate leukocytes and platelets. However, this aggressive approach can disrupt cellular integrity, resulting in greater variability in growth factor release and potentially triggering a stronger inflammatory response.8 In contrast, PRGF is obtained through slower centrifugation which isolates platelets and growth factors without leukocytes, resulting in a more controlled release of growth factors with minimal inflammatory response.4 Endoret® PRGF, developed by BTI Biotechnology Institute, is a standardized preparation protocol characterized by its leukocyte-free composition and controlled activation, leading to predictable biological outcomes and enhanced tissue regeneration.9 This technique allows surgeons to separate a patients blood into three main fractions (Fig. 1). The first fraction (F1) is the Plasma Rich in Fibrin (Superficial layer) composed of primarily plasma with low platelet and growth factor concentrations but a high fibrin concentration that can facilitate soft tissue healing and can also act as barrier membranes. The second fraction (F2), Plasma Rich in Growth Factors (Intermediate layer), contains a higher concentration of platelets and a significant level of growth factors. The F1 and F2 layers are separated from the red blood cell fraction by the third fraction which is the buffycoat layer rich in leukocytes which is not incorporated into the graft to reduce inflammation.
Fig. 1: PRGF fractions of blood after slow centrifugation. The three main fractions obtained after slow centrifugation include from top to bottom: Fraction 1 (F1) – Plasma Rich in Fibrin (Superficial layer) composed of primarily plasma with low platelet and growth factor concentrations but a high fibrin concentration that can act as a biological barrier membrane for guided bone regeneration (GBR) procedures 2. Fraction 2 (F2) – Plasma Rich in Growth Factors (Intermediate layer) contains a higher concentration of platelets and a significant level of growth factors to promote bone healing. F1 and F2 layers are separated from red blood cell fraction by a buffycoat layer rich in rich in leukocytes.

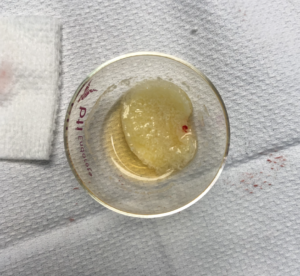
PRGF can be delivered in three distinct forms, each suited to specific clinical applications in regenerative medicine and surgery. The first is an injectable liquid form, typically administered into soft tissues or joints (Fig. 2A). The second is a gel-like fibrin clot formed upon heat-activation of PRGF. This semi-solid clot acts as a natural scaffold rich in platelets and growth factors, making it ideal for wound healing, extraction sites, and soft tissue regeneration (Fig. 2B). The third form, known as “sticky bone,” is created by combining PRGF with bone graft particulate to form a moldable, cohesive matrix. This structure improves graft handling and placement, as the PRGF-derived fibrin binds the particulate bone together, ensuring stability and ease of use during surgical procedures (Fig. 2C).
Fig. 2A
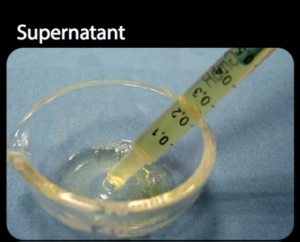
Fig. 2B

Fig. 2C
Fountain View PRGF Procedure Protocol
Since its introduction in our practice in 2019, PRGF bone grafting has significantly transformed our approach to dentoalveolar reconstruction, enhancing both regenerative outcomes and patient recovery through its bioactive and autologous properties. There has been a treatment shift towards guided bone regeneration procedures with the use of titanium mesh and PRGF, and away from autogenous grafting iliac crest. The reconstructive cases utilizing this technique have produced predictable and reproducible clinical results time and time again. The established treatment protocol includes (Fig. 3):
- Obtaining a Cone Beam (CT) of the alveolar defect site.
- Utilizing the CBCT data to print an anatomical (stereolithographic) model of the defect site and its surrounding hard tissues.
- Bending of titanium mesh to the stereolithographic model pre-operatively in order to contour the alveolar ridge to the desired anatomy for future implant placement.
- Performing the guided bone regeneration grafting procedure using the pre-bent titanium mesh as a crib to contain the combined allogeneic bone and PRGF (sticky bone) bone graft.11 Superficial to the titanium mesh, an F1 membrane is onlayed to prevent graft dehiscence (Fig. 4).
Fig. 3A
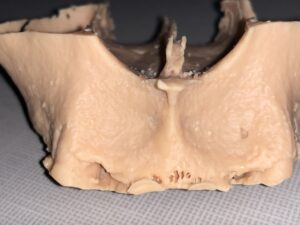
Fig. 3B

Fig. 3C
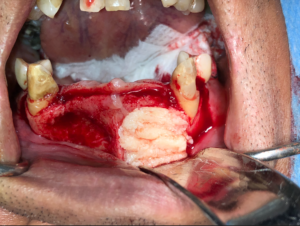
Fig. 3D
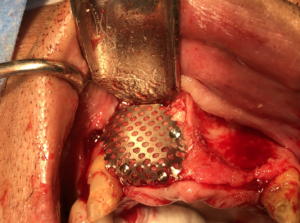
Fig. 4A
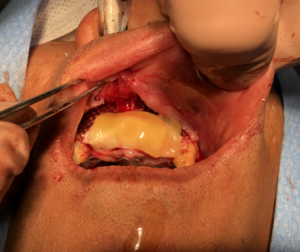
Fig. 4B
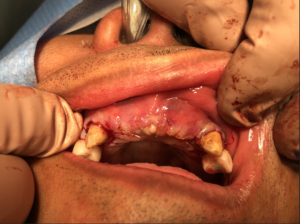
Case study 1
A 24-year-old female was referred to clinic for replacement of a congenitally absent tooth 12 with a dental implant. She was under the care of her orthodontist and restorative dentist who had maintained the restorative space in the region of the 12. Tooth 13 was extracted at young age due to its ectopic eruption. Teeth 22 and 23 were planned for future crown build ups and were not part of the treatment plan. The patient’s medical history was non-contributory with the exception of a documented allergy to oxycodone.
Clinically the patient demonstrated a high smile line with 3-4mm of gingival display on full smile. The vertical height of the alveolar crest was well maintained; however, the bucco-lingual dimension, particularly at the apical portion of the alveolus, was markedly concave. Clinically a pre-op CBCT demonstrated a narrow alveolus approximating 3.5 mm at its crest with tapering at the midcrestal level to 2.9mm. Virtual planning of the case with the restorative dentist suggested prosthetic challenges in restoring the implant in this position due to the anatomy and the buccal flaring alveolus. As such, preprosthetic grafting was discussed with the patient including autogenous block grafting (ramus graft) as well as guided bone regeneration procedures with and without the use of PRGF to correct these concerns.
Informed consent was obtained to proceed with a PRGF guided bone regeneration procedure. Pre-operative planning followed the clinical protocol as described above, with pre-bending of the titanium mesh. This was facilitated by the 3D printing of the stereolithographic model and preoperative bending (adaptation) of the titanium mesh
The titanium mesh was sterilized prior to the procedure. In the perioperative setting, blood was drawn from the patient and centrifuged (at low speed) following the Endoret® protocol allowing for isolation of F1 and F2 segments. The F2 segment was mixed with 1.0 cc of particulate allogeneic bone and activated to form “sticky bone”.
Intraoperatively, the preparation of the surgical site included a wide sulcular and crestal incision to avoid vertical releases and potential gingival scaring. The extent of dissection required to support the bone graft was optimized by utilizing the pre-bent mesh as a frame of reference.
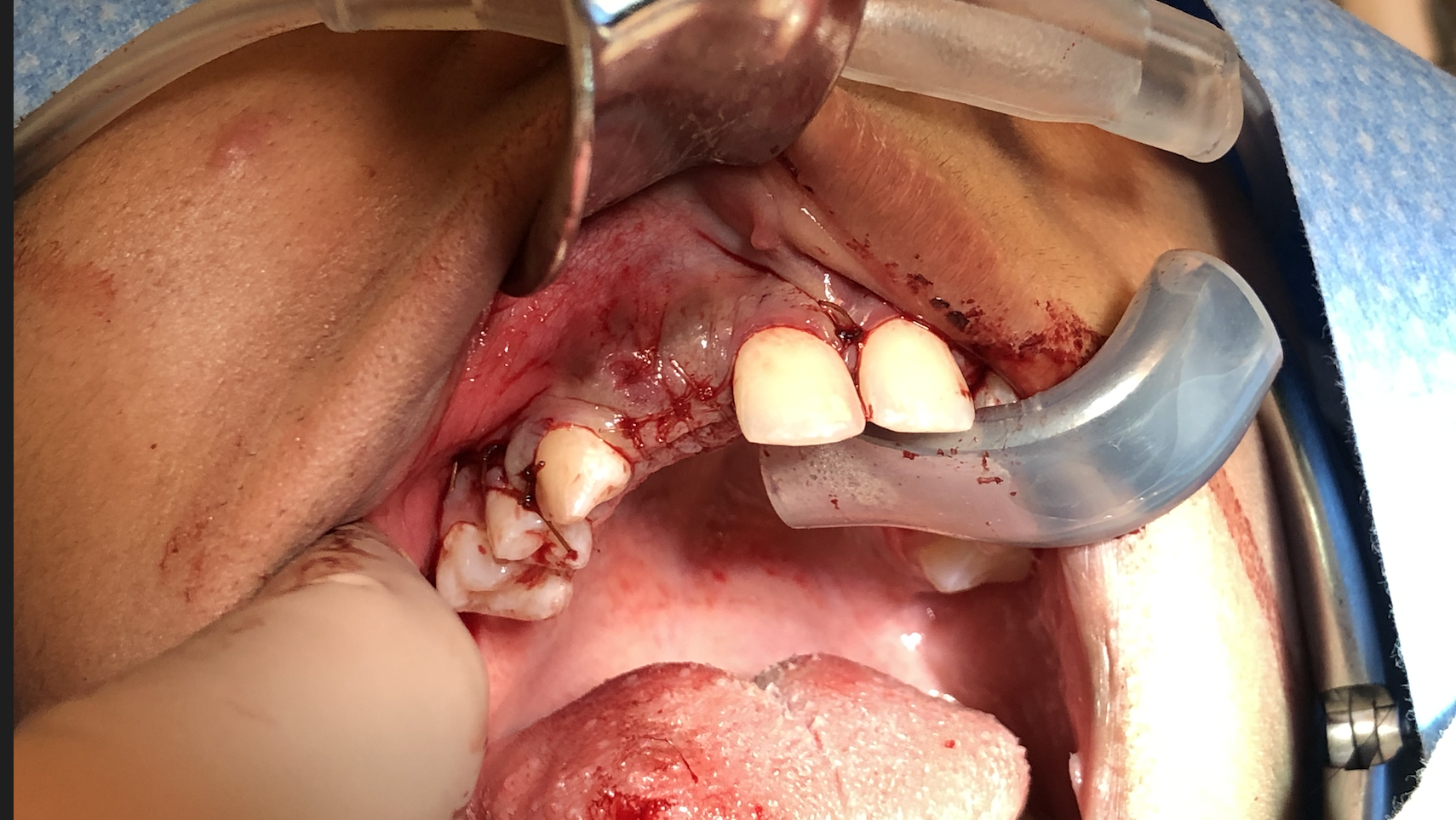
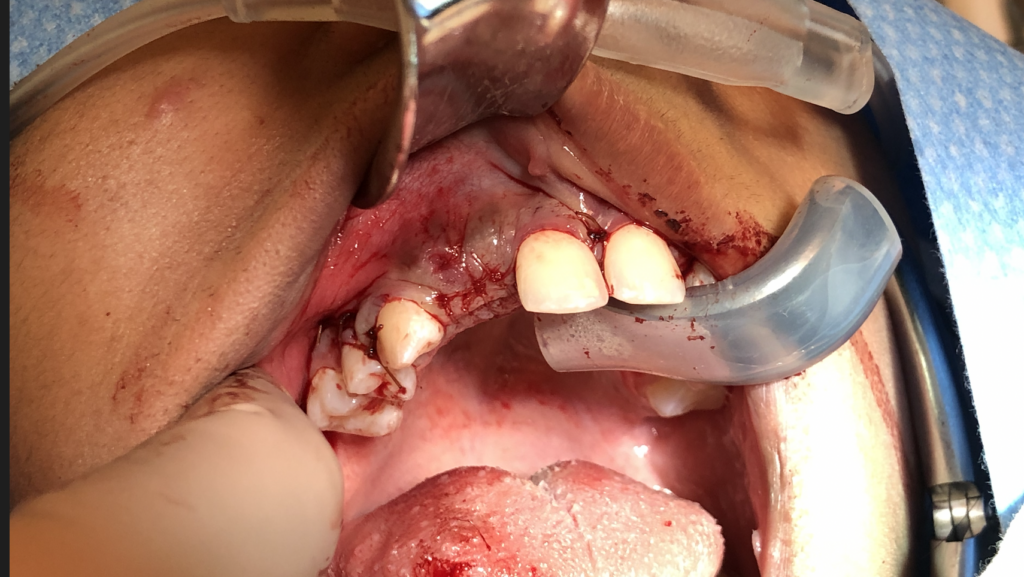
At the preference of the surgeon, the labial cortex was perforated to facilitate neovascularization of the sticky bone. The sticky bone was then onlayed to the buccal cortex and sandwiched between the buccal cortex and titanium mesh. Excess graft material was removed, and the titanium mesh was secured with a single 8mm micro fixation screw through the alveolar crest. The F1 membrane was then layered on top of the titanium mesh and the site was primarily closed (Fig. 5).
Fig. 5A
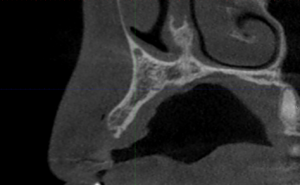
Fig. 5B
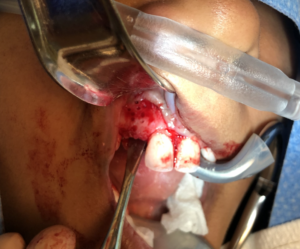
Fig. 5C
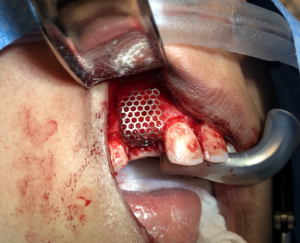
Fig. 5D
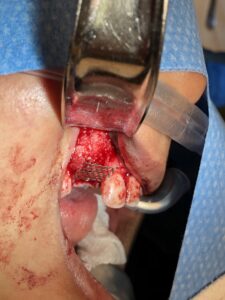
Fig. 5E
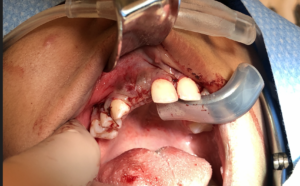
The surgical site was assessed at 6 months for healing. Clinical evaluation suggested favourable healing with a significantly thickened ridge contour, without any dehiscence of the titanium mesh. Radiographic examination of the area demonstrated integration of the bone graft that was well adapted to the previous buccal cortex. The thickness of the alveolus was increased to 5.8mm of uniform thickness of bone from the alveolar crest to its apex.
Clinically at the time of implant placement, removal of the titanium mesh revealed well vascularized, type 2 bone with a homogenous thickness from the alveolar crest to apex of the alveolus. This facilitated placement of a 3.3mm diameter implant at the 12 site with adequate buccal and palatal bone support (Fig. 6).
Fig. 6A
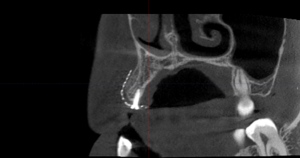
Fig. 6B
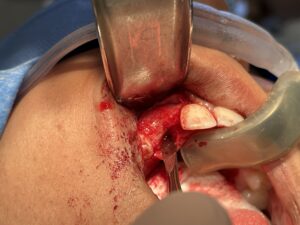
Fig. 6C
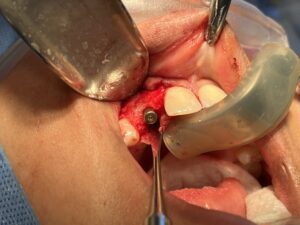
Fig. 6D
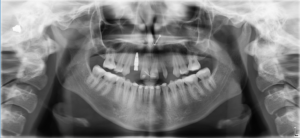
Case study 2
A 67-year-old female was referred to the clinic for management of a failed 13-22 bridge. The patient had consulted with other dental specialists and had multiple treatment plans proposed however declined to proceed as all treatment options proposed autogenous iliac crest bone grafting. The patient cited that her reasoning in declining the recommended treatment options were due to concerns of post-operative morbidity of the hip as she was a competitive golfer. The patient’s medical history included stable rheumatoid arthritis, and she was not taking any medications. However, she did attribute some of her bridge failure to impaired manual dexterity impairing her from being able to floss under her bridge. As such, the patient and her restorative dentist requested single implant crowns to facilitate easier access for future hygiene measures.
Clinically the patient demonstrated a high smile line, with good alveolar height. The anterior alveolus was significantly deficient in width. Previously obtained CBCT imaging demonstrated severe buccal-lingual atrophy of the alveolus rendering it uniformly inadequate to support dental implants from the 14-22 sites (Fig. 7). All grafting options were reviewed with the patient and informed consent was obtained to proceed with the extraction of the remaining 13 and 22 with concurrent guided bone regeneration bone grafting with the use of PRGF sticky bone.
Fig. 7A
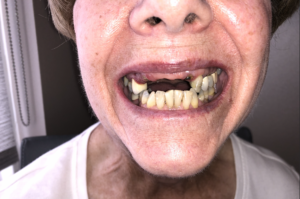
Fig. 7B
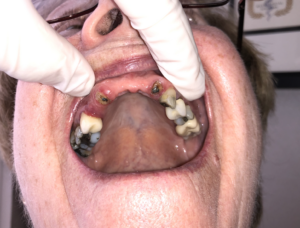
Fig. 7C
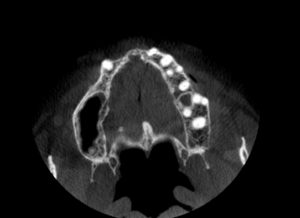
Fig. 7D
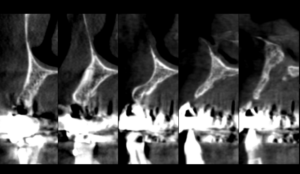
Following the PRGF bone grafting protocol at our clinic, a stereolithographic model was printed to facilitate bending of the titanium mesh pre-operatively. Intra-operatively teeth 13, and 22 were extracted with concurrent grafting utilizing 4cc of allogeneic particulate bone baked with the F2 forming sticky bone. The sticky bone was sandwiched between the alveolus and titanium mesh that was secured with 2 microfixation screws. The titanium mesh was then draped with an F1 fibrin membrane to provide a biological barrier as well as facilitate soft tissue healing and decrease the risk of mesh exposure post-operatively.
The graft was allowed to heal for 6 months at which time it was evaluated with a cone beam CT. A uniform increase in the width of the anterior maxilla as observed, with excellent graft adaptation to the native bone and good volume of graft consolidation (minimal bone graft loss below the mesh). Furthermore, the pre-operative planning phase for guided implant placement, revealed that the implant-implant and implant-tooth spacing allowed for only 5 implants to replace the 6 missing teeth. As such the arch was to be replaced with single implant crowns to replace teeth 13, 12, 11, 21 and 22.
Intra-operative removal of the titanium mesh revealed a well vascularized, uniformly thick alveolus with a type 2 bone quality for implant placement. This facilitated placement of 5 dental implants all within alveolar bone achieving primary stability of 35ncm or greater. The dental implants were given 4 months to osseointegrate at which time the bone was implants were restored with single crowns (Fig. 9).
Fig. 8A
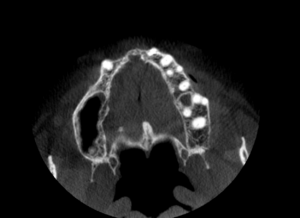
Fig. 8B
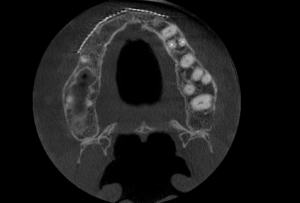
Fig. 9A
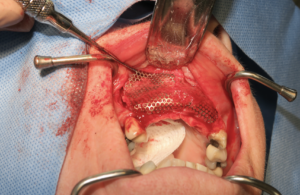
Fig. 9B
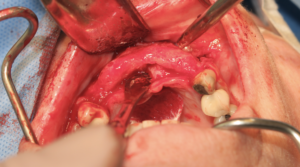
Fig. 9C
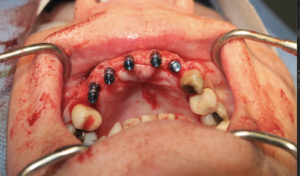
Fig. 10A
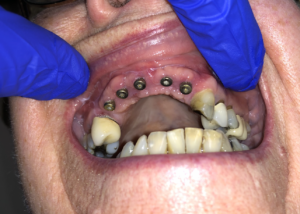
Fig. 10B
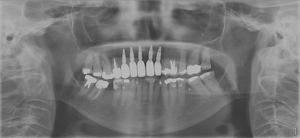
Conclusion
PRGF has demonstrated significant advantages in guided bone regeneration procedures, particularly when used with titanium mesh. It has been shown to accelerate bone healing, enhance angiogenesis, decrease inflammation and improve soft tissue healing. While PRGF-based GBR techniques offer promising, minimally invasive alternatives to autogenous bone grafts with high success rates, the technique cannot be used in all clinical scenarios.4 Contraindications for PRGF grafting include blood disorders such as severe thrombocytopenia and patients with active malignancies. Furthermore, although our clinical outcomes in GBR procedures have improved with the addition of PRGF, the literature reports mixed findings on its efficacy. While various researchers found improved wound healing.4,9,13 Other researchers found no significant difference.10,12 This is not to discredit the technique but points out the challenges in performing clinical cases with adequate numbers and controls. With increased cases and better designed studies, the significance of the therapeutic benefits of the use of PRGF may be further established.14 Despite this, the consistent clinical success of this technique observed in our practice offers valuable insights that may inform technique refinement and improve patient selection. Our clinical results strongly support that PRGF has clear benefits in guided bone regeneration procedures, particularly when combined with titanium mesh. 
Oral Health welcomes this original article.
References
- Buser, D.; Dula, K.; Belser, U. C.; Hirt, H. P.; Berthold, H. Long-Term Stability of Osseointegrated Implants in Augmented Bone: A 5-Year Prospective Study in Partially Edentulous Patients. Int. J. Periodontics Restorative Dent. 2002, 22 (2), 109–117.
- Urban, I. A.; Jovanovic, S. A.; Lozada, J. L.; Nagy, K. Vertical Ridge Augmentation with Titanium-Reinforced, Dense-PTFE Membranes and a Combination of Particulate Autogenous Bone and Anorganic Bovine Bone-Derived Mineral: A Prospective Case Series in 19 Patients. Int. J. Oral Maxillofac. Implants 2009, 24 (3), 502–510.
- Dahlin, C.; Linde, A.; Gottlow, J.; Nyman, S. Healing of Bone Defects by Guided Tissue Regeneration. Plast. Reconstr. Surg. 1988, 81 (5), 672–676.
- Anitua, E. Plasma Rich in Growth Factors: Preliminary Results of Use in the Preparation of Future Sites for Implants. Int J Oral Maxillofac Implants 1999, 14.
- Whitman, D. H.; Berry, R. L.; Green, D. M. Platelet Gel: An Autologous Alternative to FibFrin Glue with Applications in Oral and Maxillofacial Surgery. J. Oral Maxillofac. Surg. 1997, 55 (11), 1294–1299.
- Ferrari, M.; Zia, S.; Valbonesi, M.; Henriquet, F.; Pisano, A.; Spagnolo, S.; Sandrelli, L. A New Technique for Hemodilution, Preparation of Autologous Platelet-Rich Plasma and Intraoperative Blood Salvage in Cardiac Surgery. Int. J. Artif. Organs 1987, 10 (1), 47–50.
- Marx, R. E. Platelet-Rich Plasma: Evidence to Support Its Use. J. Oral Maxillofac. Surg. 2004, 62 (4), 489–496.
- Marx, R. E. Platelet-Rich Plasma (PRP): What Is PRP and What Is Not PRP? Implant Dent. 2001, 10 (4), 225–228.
- Anitua, E.; Sánchez, M.; Orive, G.; Andía, I. The Potential Impact of the Preparation Rich in Growth Factors (PRGF) in Different Medical Fields. Biomaterials 2007, 28 (31), 4551–4560.
- Lafzi, A., Faramarzi, M., Shirmohammadi, A., Behrozian, A., Kashefimehr, A., and Khashabi, E. Subepithelial connective tissue graft with and without the use of plasma rich in growth factors for treating root exposure. J. Periodontal Res. 2012, 42, 196.
- Urban, I. A., Nagursky, H., Lozada, J. L., and Nagy, K. Horizontal ridge augmentation with a collagen membrane and a combination of particulated autogenous bone and anorganic bovine bone–derived mineral: A prospective case series in 25 patients. Int J Periodontics Restorative Dent. 2013, 33, 299–307.
- Farina, R., Bressan, E., Taut, A., Cucchi, A., and Trombelli, L. Plasma rich in growth factors in human extraction sockets: A radiographic and histomorphometric study on early bone deposition. Clin. Oral Implants Res. 2012, 24, 1360–1368.
- Cumming, A. Does the use of plasma rich in growth factors (PRGF) produce benefits for patients with chronic periodontal disease in non-surgical treatment? BDJ Team 2021, 8, 16–17.
- Rocchietta, I.; Fontana, F.; Simion, M. Clinical Outcomes of Vertical Bone Augmentation to Enable Dental Implant Placement: A Systematic Review. J. Clin. Periodontol. 2008, 35, 203–215.
About the authors

Ben A. Kertesz, Department of Chemistry and Chemical Biology, McMaster University, Hamilton, Ontario.

Dr. David Psutka, Staff Surgeon, Fountain View Oral Surgery, Mount Sinai Hospital, and Trillium Health Partners.

Dr. Peter Ta, Staff Surgeon, Fountain View Oral Surgery, Sunnybrook Health Sciences Centre, Trillium Health Partners.