Trigeminal neuralgia (TN) is defined as a chronic condition characterized by the presence of unilateral neuropathic facial pain, which is of severe nature with episodic pain distributed along the branches of the fifth cranial nerve and might result in physical and/or psychological disability. According to the International Classification of Headache Disorders, TN is a condition characterized by recurrent short shock-like painful episodes that usually last a few seconds to minutes. Even non-painful activities can trigger facial pain faster in onset and termination.1
The pathophysiology of TN is still unclear, with many theories attributing to abnormal firing in afferent neurons of gasserion ganglion. The most common causes of TN are idiopathic or secondary to neurovascular conflict of the trigeminal nerve (most commonly by the superior cerebellar artery) at the root entry zone, which may result in demyelination. The demyelinated axons are prone to ectopic action potential generation. There are other secondary causes which can cause TN, namely multiple sclerosis, demyelination disorders, neural tumours, etc.2,3
Although pharmacotherapy and conservative management are the initial preferred treatments, many patients become resistant to pharmacotherapy over the years, which warrants further invasive management. Both ablative and surgical options are done in refractory patients with varied results. The options include percutaneous glycerol rhizotomy, radiofrequency ablation of gasserion ganglion, balloon compression, gamma knife radiosurgery, and microvascular decompression (MVD).4 Radiofrequency ablation is a technique that allows selective lesioning of the divisions of the trigeminal nerve. This procedure was first performed in 1974 by using fluoroscopy imaging to guide the needle through the foramen ovale to Meckel’s cave and the trigeminal ganglion.
Diagnosis
The International Headache Society has defined criteria for diagnosing Classical TN. Neurovascular conflict is the most common cause of classical TN. Secondary causes of TN include multiple sclerosis, demyelination disorders, neural tumours, etc.5 The diagnostic criteria are given below in Table 1.
Table 1: International Headache Society diagnostic criteria for TN
| Classical | Paroxysmal |
| A. Paroxysmal attacks of pain lasting from a fraction of a second to 2 min, affecting one or more divisions of the trigeminal nerve, and fulfilling criteria B and C | A. Paroxysmal attacks of pain lasting from a fraction of a second to 2 min, with or without persistence of aching between paroxysms, affecting one or more divisions of the trigeminal nerve, and fulfilling criteria B and C |
| B. Pain has at least one of the following characteristics: 1. Intense, sharp, superficial, or stabbing 2. Precipitated from trigger zones or by trigger factors | B. Pain has at least one of the following characteristics: 1. Intense, sharp, superficial, or stabbing 2. Precipitated from trigger zones or by trigger factors |
| C. Attacks are stereotyped in the individual patient | C. Attacks are stereotyped in the individual patient |
| D. There is no clinically evident neurologic deficit | D. A causative lesion, other than vascular compression, has been demonstrated by special investigations and/or posterior fossa exploration |
| E. Not attributed to another disorder |
There are other classifications of TN, but the notable one which can be useful to clinicians is given in Table 2).
Table 2: Clinical classification of TN 6,7
| 1. TN1 – Idiopathic, Spontaneous facial pain that is predominantly episodic |
| 2. TN2 – Idiopathic, Spontaneous facial pain that is predominantly constant |
| 3. Trigeminal neuropathic pain resulting from unintentional injury to the trigeminal nerve due to trauma or surgery |
| 4. Trigeminal deafferentation pain resulting from intentional injury to the nerve by peripheral nerve ablation, gangliolysis, or rhizotomy to treat either TN or other related facial pain |
| 5. Symptomatic TN due to multiple sclerosis |
| 6. Post-herpetic TN following a cutaneous herpes zoster outbreak in the trigeminal distribution |
| 7. Atypical facial pain refers to facial pain secondary to a somatoform pain disorder and requires psychological testing for diagnostic confirmation. |
A complete neurological examination with multidisciplinary involvement is necessary to diagnose and treat patients with TN. The neurological examination is mostly normal, but patients should also be enquired about other signs and symptoms like numbness, tingling, blurring of vision, hearing loss, vertigo, imbalance, neurological deficit, and seizures. This could be the early symptom or sign in patients with cerebellopontine angle tumours or multiple sclerosis. Magnetic resonance imaging (MRI) scan of the brain is useful to diagnose patients with neurovascular conflict and exclude secondary causes like neural tumors, multiple sclerosis etc. MRI with CISS sequence (augmentation by a 3D gradient echo sequences such as fast flow steady-state precession or intravenous gadolinium–DTPA) can be used to improve visualization of the vascular compression around the trigeminal nerve root.8 Other causes of facial pain, like dental etiology, temporomandibular joint pain, chronic sinusitis, otitis media, atypical facial pain syndromes, and myofascial pain, should be excluded.
Patient Selection
Conservative management is the preferred line of treatment for most patients. Carbamazepine is the drug of choice, with doses starting from 100 mg up to 1200 mg. Second line medications include oxcarbazepine, gabapentin, pregabalin, baclofen, lamotrigine, topiramate and levetiracetam.9 Surgery (microvascular decompression) or interventional pain management (gasserion ganglion radiofrequency ablation) is considered when patients are refractory to at least three drugs.10 Patients refractory to medical management, elderly patients with comorbidities and those who are not candidates for surgery are indications of lesser invasive radiofrequency ablation of the trigeminal ganglion.
Case History
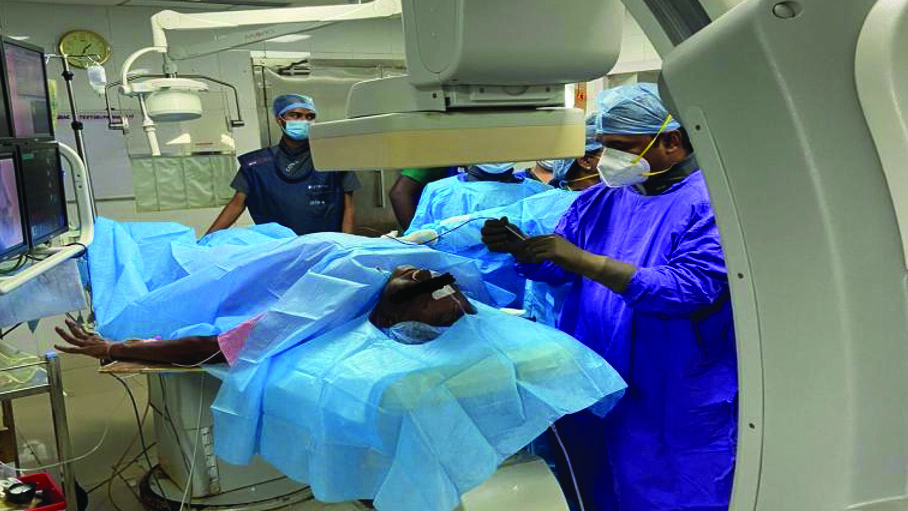
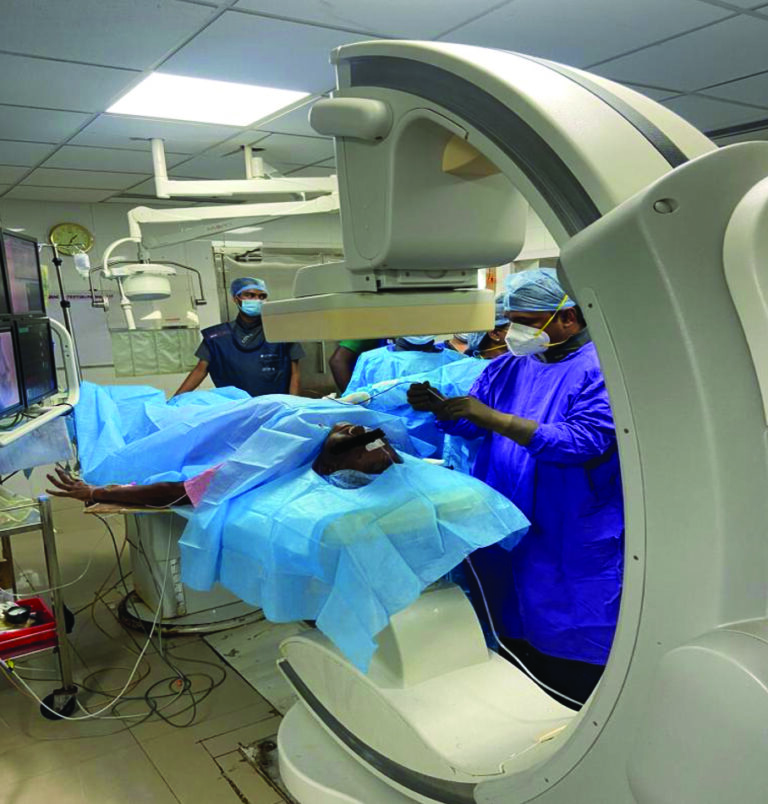
A 65-year-old male patient presented with left-sided facial pain, which was sharp, episodic and severe in nature for the past 5 years and was diagnosed as TN by the neurologist. He was on medical management for 5 years, and recently, he was found refractory to carbamazepine, gabapentin and baclofen. MRI showed a vascular loop of the superior cerebellar artery with neurovascular conflict of the trigeminal ganglion. The patient was unwilling to microvascular decompression and, hence, was referred to a pain clinic for further management. The pattern of pain distribution was along the maxillary and mandibular divisions. We proposed Radiofrequency ablation of the V2/V3 division of Gasserion ganglion under deep sedation, and the patient agreed to the same (Fig.1). The procedure was performed by an experienced pain physician (PK). Successful lesioning was done with the below-mentioned procedure steps, and the patient was discharged as a day care surgical procedure without any complications.
Fig. 1
Procedure
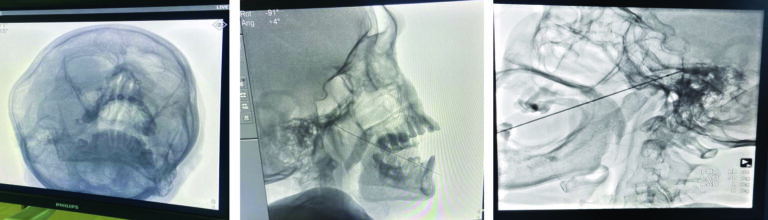
Preoperative preparation includes strict adherence to fasting guidelines. Patients should be instructed to communicate about the pattern of pain reception after sensory stimulation during the procedure. A prophylactic antibiotic is administered 1 hour before the procedure. An Anesthesiologist/ anesthesia assistant is requested for intravenous sedation with ASA standard monitoring, as radiofrequency ablation can cause patient discomfort during the procedure. With the patient in a supine position with the head extended, the C-arm is rotated to obtain an oblique submental view to visualize the foramen ovale. After infiltration of local anesthetic, the RF cannula (22 Gauge 5 mm or 10 mm active tip, Avanos, United States Inc.) is inserted 2–3 cm lateral to the angle of the mouth on the affected side to cannulate the foramen ovale. The trajectory follows a straight line directed toward the ipsilateral pupil, and it also passes 3 cm anterior to the external auditory meatus when seen from the lateral side as a visual marker. The cannula is advanced into the foramen ovale so that the fluoroscopic image shows the needle in a tunnel view (Fig. 2A), and the lateral fluoroscopic image in the foramen ovale is shown in (Fig. 2B). If there is a doubt, a digital palpation can be done inside the oral cavity to ensure that the buccal mucosa is not perforated. Once the cannula enters the foramen ovale, the depth of the cannula is assessed on the lateral fluoroscopic view. The cannula is advanced into foramen ovale till the tip of the cannula reaches 2 mm anterior to the junction of the clivus and the petrous ridge of the temporal bone. The stylet is removed from the cannula, and with gentle aspiration to make sure there is no CSF or blood, 0.5 mL of contrast dye (nonionic dye – iohexol) is injected to confirm that the needle has not penetrated the dura. The RF probe or electrode is inserted through the RF cannula for sensory and motor stimulation. Sensory stimulation at 50 Hz and 1 msec pulse duration at < 0.5 V (< 0.3V is desirable) should produce the symptoms. If the patient has paresthesia at > 0.5 V, the needle should be redirected to get the sensory symptoms at a desirable current (< 0.3 V). Motor stimulation is done at 2 Hz between 0.1 and 1.0 mA, and muscle contraction of the lower jaw may be seen, which indicates that the RF cannula is stimulating the mandibular division of the trigeminal ganglion. According to the patient’s symptoms, one or 2 lesions can be done 2 mm anterior to the clival junction for mandibular division and at the clival junction and petrous ridge for maxillary division (Fig. 2C) and 1 – 2 mm to the left of clival junction in the lateral fluoroscopic image for naso-ophthalmic division (which is usually not done routinely due to the fear of corneal ulcer and proximity of pituitary fossa)11. Usually, 1 or 2 RF lesioning at 600 – 700 C is done for 60 – 90 seconds. After the RF lesions, 40 mg of triamcinolone should be injected to prevent post-procedure neuritis. As RF lesioning is a painful procedure, deep sedation is usually required.
Fig. 2
Common Complications
Complications which are commonly encountered are bleeding, vessel puncture, and hematoma at the site of skin entry. Ablation of naso-ophthalmic division can cause diminished corneal reflex and corneal ulcer, dysesthesia, keratitis, facial numbness, infection, masseter weakness and paralysis. Rarely, aseptic meningitis, cavernous fistula, CSF leak can occur. Anesthesia dolorosa can occur if a higher temperature is used for thermocoagulation of the trigeminal ganglion (> 75 degrees Celsius). Transient paralysis of cranial nerves III, IV and VI has been reported.12-14
Efficacy of RF Ablation in Trigeminal Neuralgia – A Review of Literature
Percutaneous RF rhizotomy can result in significant pain relief as that can be obtained with a more invasive procedure. RF Ablation procedures destroy neurons or their axons of gasserion ganglion, thereby reducing the recruitment of ectopic neural activity. RF Ablation aims to produce mild to moderate hypoalgesia in the affected divisions, allowing for adequate pain control without causing significant sensory deficits. Initial pain relief following RF Ablation is relatively greater than 95% complete pain resolution.15 Pain control rates and pain relief duration consistently correlate with the degree of sensory loss. The long-term efficacy of RF ablation is comparatively lower than that of surgical technique;16 the only advantage of RF ablation over surgical technique is that it can be repeated in the same patient if required.
RF Ablation lesion size depends on multiple factors, including active tip diameter/gauge, tip length, tip temperature, and lesion time. The use of curved rather than straight electrodes allowed them to create more selective lesions to decrease rates of sensory complications.17 Real-time fluoroscopic imaging helped precisely guide, advance, and negotiate the needle tip through the foramen ovale to accurately reach the trigeminal cistern within the Meckel’s cave.18 Regarding the temperature, 68-70˚C was the optimal radiofrequency temperature for treating Idiopathic TN’s maxillary and mandibular division in certain studies.19,20 In certain other studies, the optimal temperature ranges from 60-65˚C for V1 and 72˚C and 75˚C for V2 and V2/V3 division.21
In all its forms, Percutaneous RF Ablation offers immediate pain relief with varying durability and a generally favourable side effect profile. Even though the procedure relies on landmarks to gain access to the foramen ovale, anatomic variation can make accessing the foramen ovale difficult and can increase the risk of complications. The complication has been mitigated by improving visualization and navigation to the foramen ovale. The real-time fluoroscopy-guided cannulation enables the safe passage of the needle through the foramen ovale and its accurate placement in the trigeminal cistern within the Meckel’s cave.
Although fluoroscopy has been the imaging modality of choice, newer studies have demonstrated the potential usefulness of computed tomography and neuronavigational systems, especially for patients with anatomic variants that may prevent the successful placement of needles in the target area. Cost and affordability are a limiting factor for these newer imaging studies. Due to the aforementioned reasons, we recommend RF ablation as the first-line operative procedure for severe and intractable TN, especially in elderly or medically compromised patients with comorbidities. However, treatment selection still remains a dilemma without strong evidence. In addition to incorporating these technical advances, future studies must overcome the complex patient presentation, procedural variation that limit interpretation of the existing literature. 
Oral Health welcomes this original article.
References
- Headache Classification Committee of the International Headache Society (IHS). The International classification of headache disorders. 3 edn. Cephalalgia, 2018: 1–211.
- Trigeminal neuralgia. Cruccu G, Di Stefano G, Truini A. N Engl J Med. 2020;383:754–762.
- Trigeminal neuralgia: current approaches and emerging interventions. Xu R, Xie ME, Jackson CM. J Pain Res. 2021;14:3437–3463.
- Liu JK, Apfelbaum RI. Treatment of trigeminal neuralgia. Neurosurg Clin N Am. 2004;15:319–334.
- Headache Classification Subcommittee of the International Headache Society. The international classification of headache disorders: 2nd edition. Cephalalgia. 2004;24 Suppl 1:9–160.
- Burchiel KJ. A new classification for facial pain. Neurosurgery. 2005;53(5):1164–1166.
- Eller JL, Raslan AM, Burchiel KJ. Trigeminal neuralgia: definition and classification. Neurosurg Focus. 2005;18(5):E3.
- Woolfall P, Coulthard A. Pictorail review: trigeminal nerve: anatomy and pathology. Br J Radiol. 2001;74(881):458–467.
- Cruccu G, Gronseth G, Alksne J, et al; American Academy of Neurology Society; European Federation of Neurological Society. AAN-EFNS guidelines on trigeminal neuralgia management. Eur J Neurol. 2008; 15(10):1013–1028.
- Cheshire WP. Trigeminal neuralgia: For one nerve a multitude of treatments. Expert Rev Neurother. 2007;7(11):1565–1579.
- Raj PP, Lou L, Erdine S, Staats PS, Waldman SD, eds. Radiographic Imaging for Regional Anesthesia and Pain Management. Philadelphia, PA: Churchill Livingstone; 2003.
- Percutaneous radiofrequency thermocoagulation in trigeminal neuralgia : analysis of early and late outcomes of 156 cases and 209 interventions. Gunduz HB, Cevik OM, Asilturk M, Gunes M, Uysal ML, Sofuoglu OE, Emel E. J Korean Neurosurg Soc. 2021;64:827–836.
- Radiofrequency percutaneous Gasserian ganglion lesions. Results in 140 patients with trigeminal pain. Onofrio BM. J Neurosurg. 1975;42:132–139.
- Percutaneous controlled radiofrequency trigeminal rhizotomy for the treatment of idiopathic trigeminal neuralgia: 25-year experience with 1,600 patients. Kanpolat Y, Savas A, Bekar A, Berk C. https://www.alisavas.com.tr/upload/Trigeminal-Neuralgia.pdf. Neurosurgery. 2001;48:524–532.
- Fraioli B, Esposito V, Guidetti B, et al. Treatment of trigeminal neuralgia by thermocoagulation, glycerolization, and percutaneous compression of the gasserian ganglion and/or retrogasserian rootlets: long-term results and therapeutic protocol. Neurosurgery 1989;24(2):239–45.
- Lopez BC, Hamlyn PJ, Zakrzewska JM. Systematic review of ablative neurosurgical techniques for the treatment of trigeminal neuralgia. Neurosurgery. 2004;54(4):973–982.
- Tew JM, Morgan CJ, Grande AW. Percutaneous rhizotomy in the treatment of intractable facial pain (trigeminal, glossopharyngeal, and vagal nerves). In: Schmidek HH, Sweet WH, editors. Operative neurosurgical techniques: indications, methods and results. 3 edition. Philadelphia: WB Saunders; 1996. p. 1409–18.
- Jeyaraj, P. Efficiency and Efficacy of Real-Time Fluoroscopic Image-Guided Percutaneous Gasserian Glycerol Rhizotomy (PGGR), for Intractable Cases of Trigeminal Neuralgia. J. Maxillofac. Oral Surg. 21, 1053–1064 (2022). https://doi.org/10.1007/s12663-021-01682-2
- Yao P, Hong T, Wang ZB, Ma JM, Zhu YQ, Li HX, Ding YY, Jiang CL and Pan SN: Treatment of bilateral idiopathic trigeminal neuralgia by radiofrequency thermocoagulation at different temperatures. Medicine (Baltimore). 95(e4274)2016.
- Zhao WX, Wang Q, He MW, Yang LQ, Wu BS and Ni JX: Radiofrequency thermocoagulation combined with pulsed radiofrequency helps relieve postoperative complications of trigeminal neuralgia. Genet Mol Res. 14:7616–7623. 2015.
- Guo Z, Wu B, Du C, Cheng M and Tian Y: Stereotactic approach combined with 3D CT reconstruction for difficult-to-access foramen ovale on radiofrequency thermocoagulation of the gasserian ganglion for trigeminal neuralgia. Pain Med. 17:1704–1716. 2016.
About the Author

Dr. Prem Kumar is a Professor of Anesthesia and Pain Medicine in the Department of Anaesthesia, Saveetha institute of medical and technical sciences. He is also the Fellowship Director of Regional Anesthesia and Pain medicine, specializing Regional anesthesia, Acute pain and Chronic pain management.

Dr. Dianitta Devapriya Veronica graduated from Kilpauk Medical College and Hospital, Chennai, India and is currently Assistant Professor of Otorhinolaryngology & Head and Neck Surgery, ACS Medical College & Hospital, Chennai, India.

Dr. Yuvaraj Kotteeswaran is a Staff Anesthesiologist and Interventional Pain Physician in the Department of Anesthesia and Pain Management at Thunder Bay Regional Health Sciences Centre and also an Assistant Professor at Northern Ontario School of Medicine, Thunder Bay. He is Medical Lead for Acute Pain Service.