Autoimmune diseases occur when the body’s immune system dysregulates and mistakenly targets and attacks its cells.1 While the exact etiology remains unclear, it generally involves a genetic predisposition combined with an environmental trigger, such as bacterial or viral infections, hormonal changes, environmental toxins, certain medications, smoking, obesity, and gut dysbiosis.2,3 Even sun exposure can trigger certain autoimmune diseases.4 Women are disproportionately affected, accounting for nearly 80% of cases.4 Autoimmune diseases often present in clusters, meaning individuals with one autoimmune condition are more likely to develop another.2 The resulting dysregulation frequently necessitates pharmacological interventions that alter immune function, including anti-inflammatories, insulin, plasma exchanges, immunoglobulin therapy, corticosteroids, biologics, and immunosuppressants.
Periodontal disease is a chronic bacterial infection leading to chronic inflammation and immune system dysregulation. Acute inflammation serves as a protective response to eradicate antigens swiftly; however, chronic inflammation, as seen in periodontal disease, does not resolve and typically remains asymptomatic until the disease has significantly progressed. This chronic inflammatory process is central to the tissue destruction observed in periodontal disease.
Treating and maintaining periodontal health in patients with autoimmune conditions presents unique challenges. These patients face a trifecta of complications: primary immune dysregulation, medications that alter immune function, and periodontal disease itself, which involves chronic inflammation and further immune system dysregulation. For these reasons, it is essential to identify and meticulously address modifiable contributing factors.
This case report examines a pattern of periodontal disease in a patient with multiple sclerosis (MS), specifically as it relates to local contributing factors such as biofilm, calculus, and tooth concerns, particularly open contacts.
Case report
A 39-year-old female patient was referred by her general dentist to a periodontist due to significant changes in bone density visible on bitewing radiographs taken over two years. A comprehensive medical and dental history, periodontal examination, and a full mouth series of radiographs were conducted.
The patient’s medical history included multiple sclerosis (left side dominant), left foot drop requiring an Ankle-Foot Orthotic, osteoarthritis in the spine, a history of eating disorders, anxiety, and low iron levels. She was on an anti-anxiety medication and received ocrelizumab infusions every six months. Ocrelizumab, a monoclonal antibody targeting B cells involved in the nervous system attacks seen in MS, helps reduce relapses and slow disease progression. She also took various supplements, including Vitamin D, B, biotin, calcium, magnesium, and a probiotic. The patient had no history of smoking tobacco or cannabis and currently abstains from alcohol. Notably, she reported being in particularly good health during her twenties.
The patient’s last dental scaling occurred within the past two months, following a nine-month interval. Her family history revealed periodontal disease, with her father under the care of a periodontist. The patient expressed concern about shifting teeth and food impaction, stating, “The dentist says I am losing bone in a very specific pattern.” She also reported bleeding during brushing and flossing, clenching and grinding without a nightguard, and shifting lower anterior teeth. Her wisdom teeth had been extracted uneventfully, and no teeth had been lost due to periodontal disease. The patient described her diet as “somewhere in the middle” between healthy and unhealthy. Her salivation was normal in both consistency and quantity. An extraoral examination showed a history of lower lip piercing near tooth 43, which was removed twelve years ago. Intraoral examination revealed a history of a tongue piercing, removed fifteen years prior. The gingival tissues were red, spongy, and slightly enlarged, though stippling was present. Minimal gingival recession was observed, and papillae were slightly enlarged. The patient had a Class I bite with no history of orthodontics, minor crowding with rotations from 13 to 23 and 33 to 43, and a crossbite at tooth 32. She reported food impaction in the mesial areas of teeth 26, 36, and 46.
The periodontal examination noted light generalized supragingival calculus and moderate generalized subgingival calculus. The O’Leary plaque score was 17.8%, and the bleeding score was 47.6%. Purulent exudate was noted at tooth 26, and mobilities were present at teeth 24 and 25. The gingiva near tooth 43 was thin, likely due to the former piercing. Probed pocket depths revealed sixty sites measuring 4-6mm and nine sites greater than 6mm, all localized to teeth 26, 36, and 46, with depths ranging from 7-10mm. The periodontal diagnostic statement was Generalized Stage IV Grade C, unstable periodontitis. The prognosis for teeth 26, 36, and 46 was doubtful, while all others were classified as secure with ongoing supportive periodontal maintenance.
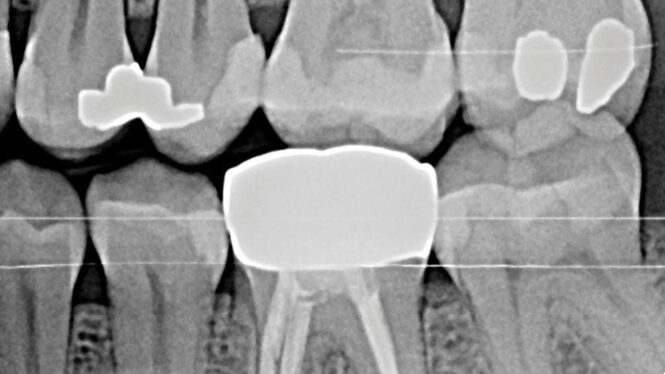
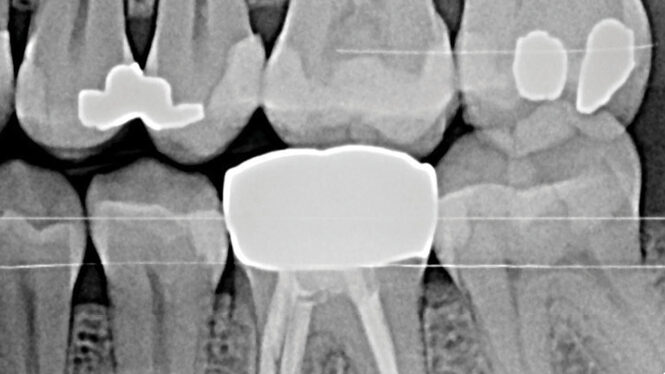
Radiographic examination, including a full mouth series of radiographs and two sets of bitewings provided by the referring dentist, confirmed rapid bone loss at teeth 26, 36, and 46 mesial. Diagnocat AI software was used to analyze the radiographic images, with findings certified by the periodontist. Recurrent caries were identified at teeth 17, 16, and 36, and open contacts were noted between teeth 25/26, 35/36, and 45/46 (Figs.1A and B, Figs. 2A and B).
Fig. 1A
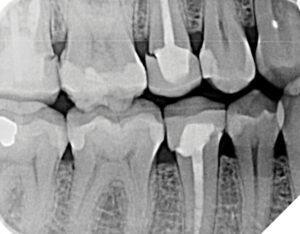
Fig. 1B
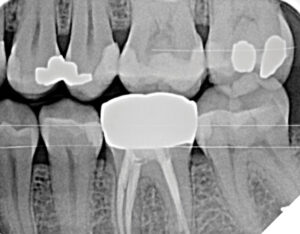
Fig. 2A
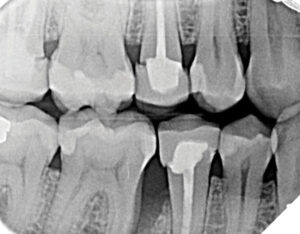
Fig. 2B
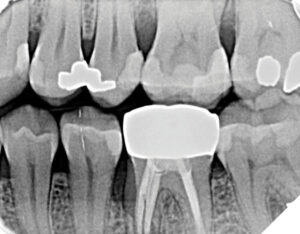
The periodontal treatment plan included non-surgical periodontal therapy comprising two scaling and root planing (SRP) appointments with local anesthetic, followed by antimicrobial therapy with 500 mg amoxicillin three times daily for ten days after SRP completion. Individually tailored oral hygiene instructions were provided, followed by an eight-week re-examination to reassess teeth 26, 36, and 46 for potential open flap surgery. The restorative treatment plan involved the referring dentist closing the open contacts at 26M, 36M, and 46M as soon as possible. Personalized oral hygiene instructions emphasized using a rechargeable toothbrush twice daily, daily flossing, and site-specific interproximal brushes.
At the eight-week re-exam, significant improvement was noted following non-surgical therapy, antimicrobial therapy, and restorative care to close the open contacts. Only six pockets measuring between 4-6mm (down from sixty at baseline) and one pocket greater than 6mm (down from nine at baseline) remained. The patient was scheduled for quarterly supportive periodontal maintenance and advised that more frequent care might be necessary during MS flare-ups, which could negatively impact her home care or immune function.
Autoimmune disease and periodontal disease: What we know
MS is an immune-mediate neurodegenerative disease affecting the brain and spinal cord.5 In MS, the immune system attacks the myelin sheath that protects nerve fibers, leading to communication problems between the brain and the body.6 Symptoms vary depending on which nerves are affected and the extent of damage, with common symptoms including muscle weakness, difficulty walking, vision problems, and fatigue.6 One hallmark of MS is elevated inflammation levels within the body due to immune system deterioration. MS patients are at a higher risk for gingivitis and periodontal disease due to this increased inflammation. The inflammation resulting from gingivitis and periodontal disease can exacerbate MS symptoms, creating a cycle of worsening oral and overall health. Several studies have demonstrated a link between periodontal disease and various autoimmune diseases, primarily through shared mechanisms of chronic inflammation and immune system dysfunction.7 In autoimmune patients, dysregulation of the immune system can also target periodontal tissues, leading to inflammation and the rapid destruction of the gingiva and supporting structures of the teeth.
Oral infections can trigger systemic inflammatory responses, potentially contributing to the development of systemic diseases. An imbalanced interaction between the periodontal microbiome and the host’s inflammatory response explains local tissue destruction in periodontitis.8 Bacteria from periodontal tissues can enter the bloodstream, initiate systemic inflammation, and cause cellular or molecular changes in the liver and bone marrow, potentially exacerbating other health conditions. This is particularly concerning for autoimmune patients, whose compromised immune systems make them more susceptible to such effects. Additionally, bacteria from the periodontal tissues can spread to other parts of the body, worsening inflammatory diseases.8
For these immunocompromised patients, it is crucial to address modifiable local factors through meticulous professional biofilm disruption, calculus removal, correction of restorative issues and targeted oral hygiene instruction. Antimicrobial therapy should be considered to assist in reducing pathogens after biofilm disruption. Providing antimicrobial therapy before biofilm disruption is ineffective, as antibiotics cannot effectively penetrate intact biofilm. Oral care providers must be particularly vigilant in assessing and correcting tooth factors that contribute to biofilm accumulation. In this case report, open contacts between teeth contributed to food impaction, biofilm accumulation, chronic inflammation, and subsequent bone loss. The patient’s immune system dysregulation likely exacerbated the immune response, leading to marked bone loss.
Additionally, fatigue, joint pain, and other systemic symptoms associated with autoimmune diseases can make effective oral hygiene challenging. These symptoms inhibit proper oral hygiene practices, allowing bacterial biofilm to thrive and contribute to periodontal disease. Therefore, individuals with MS must maintain regular dental visits and a stringent oral hygiene routine to mitigate these risks. MS patients with a diagnosis of periodontal disease may benefit from being under the care of a periodontist, as immune dysregulation can exacerbate the disease process.
Moreover, the interactions between MS medications and the oral environment must be considered. Common MS medications, including corticosteroids, muscle relaxants, anticonvulsants, antidepressants, and immunosuppressants, have side effects that may affect the oral mucosa and periodontium.9 Xerostomia, a common side effect, reduces salivary flow, increasing the risk of tooth decay, periodontal disease, and oral infections.9 Other side effects, such as gingival hyperplasia, ulcerative stomatitis, and candidiasis, also negatively affect the oral cavity and can exacerbate periodontal disease.9 Additionally, immunosuppressant medications can increase the risk of oral infections like gingivitis and periodontitis by weakening the body’s ability to eradicate pathogens.10
Discussion
Dental professionals understand the variations and complexities involved in treating each patient. In managing autoimmune patients, it is imperative to exercise rigorous diligence in identifying and mitigating all modifiable factors. Open contacts contributed to food impaction, challenging oral hygiene, and rapid bone destruction in this case. Consideration must be given to primary immune dysregulation, immune-altering pharmacological interventions, and their side effects. Disease symptoms such as fatigue and muscle weakness can affect the patient’s ability to perform effective self-care, requiring modifications to the treatment plan. Careful monitoring of the autoimmune disease state and periodontal condition is critical, as active disease processes can lead to a vicious cycle of inflammation and exacerbation of both diseases. Recommendations for each patient include eliminating contributory local factors, such as compromised restorations and tooth surface irregularities. A vital aspect of this strategy is definitive SRP, followed by antimicrobial treatment post-SRP once biofilm disruption is achieved. Individualized oral hygiene instruction should consider patient limitations and address local contributing factors such as rotations or crowding.
A quarterly supportive periodontal maintenance schedule should be advised, with increased frequency during flare-ups, to ensure optimal management and care. Referral to a periodontist should be strongly considered not only upon the diagnosis of periodontal disease or if the risk is deemed high due to factors such as smoking, diabetes, or genetic predisposition but also in cases where underlying autoimmune conditions are present, as these conditions warrant serious attention. 
Oral Health welcomes this original article.
References
- Kaur, G., Mohindra, K., & Singla, S. (2017). Autoimmunity – Basics and link with periodontal disease. Autoimmunity Reviews, 16(1), 64–71. https://doi.org/10.1016/j.autrev.2016.09.013
- Pieren, J., & Bowen, D. (2020). Darby and Walsh Dental Hygiene: Theory and Practice (Fifth Edition). Elsevier, Inc.
- Vojdani, A., Pollard, K. M., & Campbell, A. W. (2014). Environmental triggers and autoimmunity. Autoimmune Diseases, 2014, 798029. https://doi.org/10.1155/2014/798029
- National Institute of Environmental Health Sciences. (2024). Autoimmune diseases. US Department of Health and Human Services, National Institute of Environmental Health Sciences. Retrieved August 26, 2024, from https://www.niehs.nih.gov/health/topics/conditions/autoimmune
- Tang, Z., Cabezas, M., Liu, D., Barnett, M., Cai, W., Wang, C. (2021). LG-Net: Lesion Gate Network for Multiple Sclerosis Lesion Inpainting. In: de Bruijne, M., et al. Medical Image Computing and Computer Assisted Intervention – MICCAI 2021. MICCAI 2021. Lecture Notes in Computer Science, vol 12907. Springer, Cham. https://doi.org/10.1007/978-3-030-87234-2_62
- Lamb, Y. N. (2022). Ocrelizumab: A review in multiple sclerosis. Drugs, 82(3), 323–334. https://doi.org/10.1007/s40265-022-01672-9
- Nair, S., Faizuddin, M., & Dharmapalan, J. (2014). Role of autoimmune responses in periodontal disease. Autoimmune Diseases, 2014, 1–7. https://doi.org/10.1155/2014/596824
- Hajishengallis, G., & Chavakis, T. (2021). Local and systemic mechanisms linking periodontal disease and inflammatory comorbidities. Nature Reviews Immunology, 21(7), 426–440. https://doi.org/10.1038/s41577-020-00488-6
- Costa, C., Santiago, H., Pereira, S., Castro, A. R., & Soares, S. C. (2022). Oral health status and multiple sclerosis: Classic and non-classic manifestations—case report. Diseases, 10(3), 62. https://doi.org/10.3390/diseases10030062
- De Almeida, J. M., Matheus, H. R., Fiorin, L. G., Furquim, E. M. A., & Gusman, D. J. R. (2021). Influence of immunosuppression on the progression of experimental periodontitis and on healthy periodontal tissue: A rat in vivo study. Journal of Dental Research, Dental Clinics, Dental Prospects, 15(2), 94–99. https://doi.org/10.34172/joddd.2021.016
About the Authors

Colleen C. Porter is a Registered Dental Hygienist working in a periodontal specialty office and serving as part-time Professor at Niagara College Canada. She earned her Certificate in Dental Hygiene from Niagara College and her Bachelor of Education in Adult Education from Brock University. Most recently, she completed her Certificate in Local Anesthesia at Saskatchewan Polytechnic.

Christina R. Grimo is a Brock University graduate with a Bachelor of Science in Medical Sciences and has been accepted into the Brock University Master of Science in Applied Health Sciences, specializing in Quality Improvement Sciences.