
Abstract
Menopause is a universal physiologic transition, driven by the decline in ovarian follicular activity and sex-steroid production. While its systemic implications are widely recognized, the oral and craniofacial sequelae, and its association with sleep-disordered breathing, remain under-addressed in both routine medical and dental care environments. This article synthesizes literature data and findings, and outlines what dental teams need to know: the scope of menopausal change in the dental patient population; the mechanisms linking estrogen and progesterone decline, to periodontal disease, salivary dysfunction, burning mouth syndrome (BMS), temporomandibular disorders (TMDs), and implant outcomes; the rising risk of Sleep Disordered Breathing (SDB), which includes snoring, Upper Airway Resistance Syndrome (UARS) and Obstructive Sleep Apnea (OSA) after menopause; and the psychosocial context that shapes symptoms, behaviors, and adherence to therapy. The role for dental professionals is also discussed, facilitating awareness and education, screening, interprofessional collaboration, referral, and treatment options including oral appliance therapy, and long-term support. By using structured checklists, flowcharts, and communication scripts, your dental team can be adequately prepared to integrate menopause-aware protocols into recall visits that can improve quality of life, reduce oral disease burden, and contribute to better systemic health outcomes for millions of patients.1-3
Key takeaways
- A substantial share of adult dental patients are peri- or postmenopausal; many will not volunteer symptoms unless asked.1
- Estrogen deficiency is associated with accelerated periodontal breakdown, xerostomia, BMS, TMDs, and altered bone metabolism relevant to implant outcomes.2-5
- The risk and severity of SDB rise after menopause due to hormonal, anatomic, and neuromuscular factors; disease presentation of SDB is often atypical in women.6-9
- Psychosocial symptoms (anxiety, low mood, brain fog) and sleep disruption can reduce self-care, increase parafunction, and impair adherence.10-12
- Regarding Sleep Disordered Breathing, adequately trained Dentists can create awareness and education, screen, refer for medical diagnosis, and coordinate with physicians and sleep specialists to provide oral appliance therapy when indicated. Once oral appliance therapy is provided to manage SDB, regular follow-up and reporting to the physician is required.13,30
- Simple chairside tools—targeted history questions, validated questionnaires, fluoride/saliva strategies, and brief scripts—support implementation of peri- and postmenopausal patient care in the dental office.
Menopause is the permanent cessation of menstruation resulting from the loss of ovarian follicular activity, confirmed retrospectively after 12 consecutive months of amenorrhea (the absence of menstruation) in the absence of other causes.1 In North America the mean age is about 51 years, though the window spans the early 40s to the late 50s and varies by genetics, race/ethnicity, smoking status, and medical history.1
Perimenopause: the transition period leading up to menopause, can be years of fluctuating estradiol and progesterone with irregular cycles, vasomotor symptoms, sleep disturbance, and on occasion, oral symptoms such as mucosal sensitivity and gingival inflammation.1-3
Menopause: permanent cessation of menstruation, defined as the point in time following 12 consecutive months without a period.1
Postmenopause: the years after menopause, characterized by chronically low estrogen, rising risks for xerostomia, burning mouth syndrome (BMS), osteoporosis, cardiometabolic disease, progression of periodontitis, bone loss, development of anxiety, depression, and Sleep Disordered Breathing (SDB).2-6
Related article: More women break menopause taboos — and why dentistry must pay attention
Pathophysiology relevant to dentistry involves reduction of certain hormone levels. Estradiol supports collagen turnover, mucosal thickness, vascular perfusion, and host immune modulation. Decline in estrogen is linked to heightened gingival inflammation under similar plaque loads, so even when hygiene habits remain unchanged, an increase in gingival inflammation is observed. It is also linked to greater crestal bone loss, altered salivary quantity/quality, taste changes, and dysesthesia (e.g., BMS).2,5 Progesterone interacts with ventilatory control and upper-airway neuromuscular tone; reductions may contribute to airway collapsibility during sleep (Fig. 1).6,7
Fig. 1
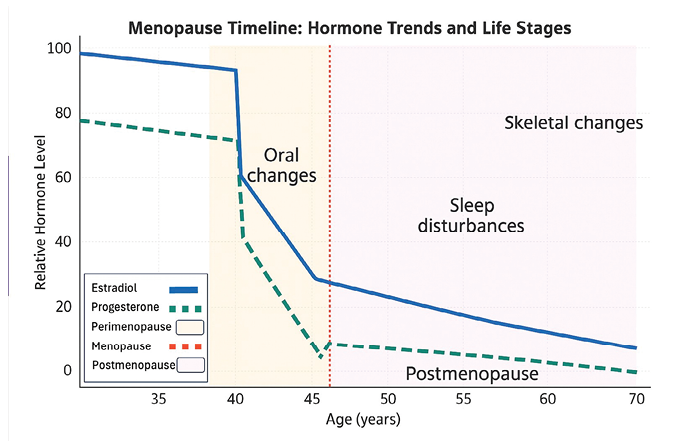
Percentage of the dental patient population affected
Women constitute roughly half of adult dental patients in a general dental practice. With life expectancy above 80 in many countries, over one-third of a typical woman’s life is lived post menopause.1 Perimenopause commonly begins in the 40s, so dental teams caring for midlife adults will see a substantial cohort with hormonally influenced oral and sleep concerns at any given time. Across North America alone, more than a million women transition to menopause each year, creating a large and growing care need within dentistry.1,3 Considering the frequency with which patients attend for regular and follow-up dental visits, dentistry is ideally positioned to efficiently and routinely screen and refer patients in a timely manner (See Table 1).
Table 1: Proportion of menopause dental patients
| Age range | % Female population | Estimated % of dental patient pool | Common oral symptoms | Common sleep symptoms |
| 45-49 | 7% | 5-10% | Early Xerostomia, Mild Periodontal Changes | Mild Insomnia, Occasional night Sweats |
| 50-54 | 8% | 8-12% | Peak Xerostomia, Increased gingival Inflammation | Frequent night sweats, difficulty maintaining sleep |
| 55-59 | 8% | 8-10% | Burning mouth, attachment loss, Sleep Changes | Insomnia possible SDB onset |
| 60-64 | 7% | 6-9% | Tooth mobility, bone density changes | SDB symptoms more common, fragmented sleep |
| 65+ | 12% | 10-15% | Prosthetic changes, implant osseointegration issues | SDB prevalence higher, comorbidities higher |
Practical implications: dental practices that proactively ask about life-stage and sleep health will uncover unaddressed symptoms and risks more reliably than practices that wait for patients to volunteer concerns. Because oral signs (e.g., unexplained sensitivity, burning, worsening periodontitis under stable plaque control) may precede a formal medical conversation about menopause, the dental setting plays an important sentinel role,2,4,5 helping to direct patients in this stage of life to have a conversation with their physician, helping to reduce unnecessary suffering while the issue slowly progresses with symptoms of increased intensity.
Current awareness and diagnostic gaps
Despite prevalence, menopause-related oral and sleep issues are often under-recognized in dentistry. Barriers include patient stigma and uncertainty about relevance to the mouth; limited formal training on menopause in dental curricula; and absence of standardized screening prompts in dental and medical histories.1,8 Regarding SDB, women with OSA or UARS may present with insomnia, fatigue, headaches, or mood changes rather than the classic loud snoring and sleepiness profile typically found in men suffering with SDB. These differences in presentation contributes to delayed referral, and mis- or underdiagnosis.6,7,10 Similarly, oral dryness, taste change, and BMS may be attributed to stress alone, without consideration of menopause related, hormonal etiology. Closing this gap requires “normalizing” the topic to facilitate conversation, creating simple chairside prompts, and building clear referral pathways to medical diagnosis and the required medical/dental care.10-13
Menopause and oral health
Periodontal health
Estrogen modulates inflammatory responses and bone remodeling. In estrogen-deficient states, customary plaque loads, that have resulted in healthy gingival tissues pre menopause, produce heightened gingival inflammation, more bleeding on probing, and faster clinical attachment loss.2,4 Postmenopausal women, especially with osteoporosis, demonstrate greater alveolar bone resorption and may experience more rapid progression in the absence of targeted management, which includes risk factor control (meticulous biofilm disruption, smoking cessation), adjunctive fluoride, anti-inflammatory rinses as indicated, and tighter periodontal maintenance intervals.14-15
Salivary function, xerostomia and caries
Reduced salivary flow and salivary compositional changes are common complaints during midlife. Several studies demonstrate that menopause leads to measurable declines in salivary flow, both unstimulated and stimulated, although dry mouth symptoms may not always be overtly perceived.17 More recent reviews emphasize that reduced salivary flow is common post-menopause and predisposes to oral issues like xerostomia, caries, and periodontal disease.18 A significant inverse correlation has been demonstrated between salivary estradiol and xerostomia scores, alongside elevated salivary calcium.19
Xerostomia increases caries risk, mucosal irritation, and denture intolerance.5 Assessment should include medication review (polypharmacy is common), ensuring good hydration habits, and regular screening for candidiasis. Management includes saliva-sparing strategies, such as humidification, sugar-free gum/xylitol, saliva substitutes, topical fluoride (varnish/at-home gels), chlorhexidine as indicated, and antifungal therapy if superinfection is present.15-16 Hormone Replacement Therapy (HRT) has been shown to restore salivary estradiol and significantly reduce symptoms of dry mouth.20,21
A structured prevention plan includes diet counseling with focus on meal timing and non-cariogenic snacks; prescription-strength topical fluorides (varnish at recall and 5,000 ppm home use); casein phosphopeptide–amorphous calcium phosphate (CPP-ACP)22 or arginine-containing pastes;23 and customized trays for neutral sodium fluoride gel when rampant caries risk is present.15-16 Erosive tooth wear may accelerate with nocturnal mouth breathing or reflux, both more common when sleep is fragmented. Screening questions for reflux and referral to primary care are warranted when dental signs are present.14-16
Burning Mouth Syndrome (BMS)
BMS is a neuropathic pain disorder characterized by burning, tingling, or scalding sensations with a normal-appearing mucosa. It is more frequent in perimenopausal and postmenopausal women and may be influenced by neurohormonal changes.5 It is important to exclude secondary causes (candidiasis, nutritional deficiencies, dry mouth, allergy). Management is multimodal: validation and education, parafunctional awareness, salivary support, topical clonazepam or capsaicin when appropriate, and collaboration with pain specialists for persistent cases.16,24
Temporomandibular Disorders (TMDs)
Estrogen receptors in the TMJ and masticatory muscles suggest hormonal influences on joint homeostasis. Some studies report increased joint pain or degenerative changes with estrogen decline, though findings are heterogeneous.3,4 Practical care includes conservative therapy: education, habit awareness, physical therapy, occlusal appliances as appropriate, and stress reduction. Rule out inflammatory arthritides when red flags (fever, swelling, morning stiffness) are present.20,24
Implants and bone metabolism
Altered bone turnover in hypoestrogenic states and osteoporosis medications (e.g., antiresorptives) intersect with implant planning. While most postmenopausal patients are candidates, risk stratification matters: evaluate bone density history, medication exposure, and periodontal status. Emphasize impeccable plaque control, staged loading where indicated, and close monitoring of peri-implant tissues.15,25
Table 2: Oral conditions and management strategies in menopause
| Condition | Mechanism | Clinical signs | Management strategies |
| Periodontal Disease | Estrogen Deficiency leads to increased inflammation and bone loss | Attachment loss, bleeding on probing, bone loss | Meticulous oral hygiene, frequent maintenance, adjunctive therapies, coordination with physician |
| Xerostomia | Reduced salivary gland function due to hormonal decline or medications | Dry mouth, altered taste, difficulty swallowing | Saliva substitutes, hydration, sugar free gum, sialagogues |
| Burning Mouth Syndrome | Neuropathic changes and mucosal atrophy | Burning, tingling oral mucosa without lesions | Topical clonazepam, alpha-lipoic acid, CBT, patient reassurance, multidisciplinary referral |
| Carries / Erosion | Hyposalivation and diet changes and medication side effects | Increased caries incidence, smooth surface lesions | Caries risk assessment, fluoride varnish/gel, dietary counselling, remineralization agents |
| TMD | Hormonal effects o Joint cartilage and pain modulation | Pain, clicking, limited jaw opening | Occlusal appliances, physiotherapy, anti-inflammatory medications |
| Implant Osseointegration Issues | Altered bone metabolism and density loss | Poor primary stability, delayed integration | Pre-op bone assessment, possible grafting, extended healing times |
Menopause and sleep
Multiple cohort studies demonstrate disturbed sleep and a higher SDB prevalence and severity in postmenopausal women compared with premenopausal peers; independent of age and BMI.6-9 Mechanisms include sleep fragmentation, reduced upper-airway dilator muscle responsiveness, increase in loop gain (defined as an instability in ventilatory control), and changes in fat distribution leading to increased parapharyngeal fat pads.6,7,9 Comorbid insomnia, restless legs, and vasomotor symptoms can further fragment sleep and magnify daytime somnolence.10-12
Presentation often differs from men: women may report non-restorative sleep, difficulty falling or staying asleep, morning headaches, concentration problems, fatigue and mood changes rather than witnessed apneas, disruptive snoring and sleepiness. Because these symptoms overlap with perimenopausal complaints, SDB risk is easily missed without targeted screening.7,10 Dental professionals can deploy brief, self-administered questionnaires such as the STOP-Bang questionnaire,26,27 inquire about snoring, witnessed apneas, nocturia, morning headaches, and excessive sleepiness or fatigue, and route patients for diagnostic testing when appropriate.13 The Epworth Sleepiness Scale questionnaire, which is commonly used by sleep physicians to evaluate the level of sleepiness is also a useful self-administered questionnaire, but it does not provide an assessment for the risk of SDB and should not be relied on for this purpose.28
The STOP-Bang questionnaire is a useful self-administered tool to help assess the risk of sleep apnea and has been validated for use with both males and females.26,27 However, studies suggest that it should be adapted for women by adjusting the interpretation of scores, particularly for women with intermediate scores (3 or 4), and potentially by considering a lower BMI cutoff point.29 While the original STOP-Bang questionnaire performs well in identifying sleep apnea across various populations, including women, a lower threshold for women might be necessary due to the questionnaire’s inherent bias towards male characteristics.
Untreated SDB in postmenopausal women is associated with elevated risks of hypertension, atrial fibrillation, insulin resistance, dyslipidemia, and cognitive decline, with additive effects when poor sleep overlaps with vasomotor symptoms.6-10 Effective therapy, such as continuous positive airway pressure (CPAP), oral appliance therapy (OAT), weight optimization, and positional therapy, improve blood pressure, daytime alertness, and quality of life, and in some patients, may also reduce cardiometabolic risk markers.13,30,31
Hormone Replacement Therapy (HRT) may alleviate vasomotor symptoms and improve sleep continuity for eligible patients under medical care, but it is not a SDB treatment; a sleep study to obtain an accurate evaluation and diagnosis remains necessary when symptoms or risk flags are present or persist post HRT.1,3 For patients testing positive for SDB despite undergoing HRT, CPAP or OAT may then be appropriate.13,30
Treatment is individualized. For patients with SDB (OSA or UARS), who are CPAP-intolerant or prefer alternatives, custom oral appliance therapy (OAT) is evidence-based and recommended by joint AASM–AADSM guidelines, with medical oversight and follow-up sleep testing to document efficacy.13,30 Although CPAP is recommended as first line therapy for SDB, the literature documents that CPAP adherence, which when using a commonly cited benchmark of four hours use/night, five nights/ week, is approximately 50% at six months, and 17% at five years,31,32,33,34,35 leaving many diagnosed patients unmanaged for their SDB. According to the AASM-AADSM joint guidelines, OAT is considered a “Standard of Care” to manage all severities of SDB if a patient cannot tolerate CPAP, or if they prefer an oral appliance to CPAP.13,30 All dental personnel including the dentist and entire team can play a role regarding increasing awareness, screening, referring, managing (under the direction of a physician) and follow-up for patients afflicted with SDB. Adequate training is required in order to properly fulfill this initiative along with selecting the most appropriate appliance for a particular patient, counseling on side effects (tooth movement, bite changes), using titration and follow-up protocols, and coordinating with sleep physicians for ongoing management and comorbidity control (See Fig. 2).13,30,35
Fig. 2
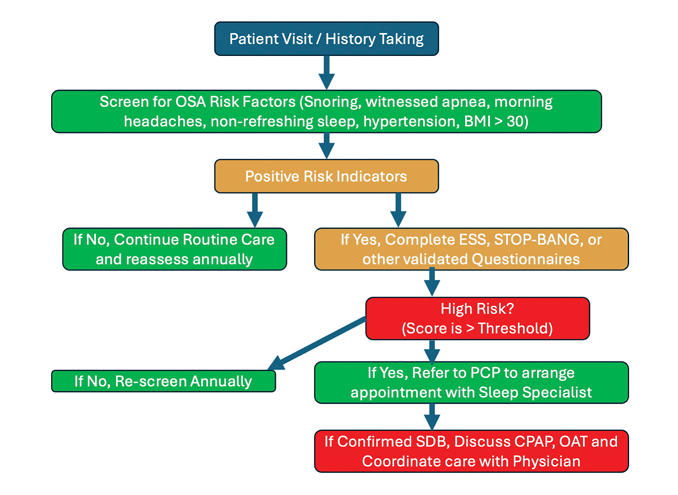
Psychosocial & mental health impacts
The menopausal transition is associated with increased rates of anxiety, depressed mood, irritability, and cognitive complaints (e.g., ‘brain fog’), which are intensified by poor sleep quality.10-12 Psychosocial stress can exacerbate parafunction (clenching/bruxism), trigger or amplify orofacial pain, and reduce adherence to home care and maintenance appointments. Stigma around discussing menopause or snoring can delay care-seeking. Dental teams can mitigate these effects by normalizing the topic, screening sensitively, and linking patients to supportive resources. Brief, self-administered questionnaire such as the PHQ-2 is a validated screening instrument for major depression, especially in older populations, where it has shown excellent sensitivity (100%) and acceptable specificity (77%). It offers a succinct, self-administered first-step screening for depression and anxiety symptoms, flagging patients who may benefit from medical or behavioral referrals.36,37,38
Table 3: Menopause psychosocial impacts for dental professionals
| Psychosocial / mental health impact | Clinical notes for dental professionals |
| Mood Changes – Depression / Anxiety | Observe patient during visits; screen for mood symptoms using validated questionnaires |
| Cognitive Effects – Memory & Concentration | Be aware of forgetfulness with home care instructions; provide written & verbal guidance |
| Sleep Disturbances – Insomnia / SDB | Identify bruxism, xerostomia, airway issues that may link to poor sleep; refer for medical assessment |
| Decreased Self-Esteem – Body Image Concerns | Supportive, empathetic communication |
| Social Withdrawal – Relationship Strain | Encourage open discussion about dental anxiety or social impact |
| Fatigue & Low Motivation – Reduced Daily | Recognize fatigues impact on oral hygiene compliance, offer simplified home care |
| Increased Stress Sensitivity – Burnout Risk | Discuss stress management and self-care, refer to mental health support when appropriate |
Screening
Embed menopause-aware prompts into medical histories: stage (peri/post), vasomotor symptoms, sleep quality, snoring, witnessed apneas, nocturia, morning headaches, daytime sleepiness/fatigue, mood changes, xerostomia, taste changes, burning sensations, TMD symptoms. Use targeted indices such as Bleeding on Probing (BOP), Periodontal Disease (PD), Clinical Attachment Level (CAL), radiographic crestal levels, and caries risk assessments with attention to salivary flow. Consider brief Sleep Disordered Breathing questionnaires and document risk flags that trigger a referral.13,27,28,30,36,39 Access to the useful screening tools should be facilitated and incorporated into the screening process. (See Table 4)
Table 4: Menopause impacts and dental professional roles
| Domain | Menopause impact | Dental professional role |
| Oral Health | Dry mouth, caries, burning mouth, periodontal changes | Create awareness and educate on link between menopause, oral health, and SDB; normalize conversations, screen, refer, preventive care, manage xerostomia, burning mouth, caries risk. |
| Sleep | SDB risk increases with menopause | Create awareness & educate, (Integrate menopause-related questions into health history, ESS, STOP-BANG, PHQ-2 etc.), screen, referral for medical assessment, oral appliance therapy |
| Psychosocial | Mood disorders, quality of life decline | Create awareness and educate, empathetic communication, refer, provide support |
Table 5: Risk factors and implications for dental practice
| Risk Factor | Relevance to menopause | Dental practice implication |
| Hormonal Changes | Decline in estrogen impacts bone density, soft tissues | Monitor periodontal status, bone loss risk |
| Sleep Disturbances | Insomnia, SDB more prevalent | Screen using STOP-BANG, refer for MD Sleep Evaluation |
| Medication Use | SSRIs, bisphosphonates, HRT, common | Evaluate xerostomia, MRONJ risk, oral mucosal health |
| Psychological Stress | Anxiety, depression linked to menopause | Provide empathetic support, oral parafunction monitoring |
Documentation and risk management
Document menopause stage (self-reported), key symptoms, screening scores, and counseling/referrals provided. Track periodontal indices, caries activity, salivary status, and OAT outcomes over time. Quality improvement can include auditing how often sleep risk is screened, how many referrals are completed, and outcomes of those referrals. Provide patients with written summaries and after-visit instructions; these support adherence and reduce medico-legal risk by demonstrating informed counseling and shared decision-making.13,30,39
Table 6: Screening tools relevant to menopausal patients
| Screening tool | Purpose | Dental application |
| Epworth Sleepiness Scale (ESS) | Self-administered first-step screening for daytime sleepiness | Integrate into patient intake for at-risk groups |
| STOP-BANG Questionnaire | Self-administered first-step screening for SDB | Integrate into patient intake for at-risk groups |
| PHQ-2 Questionnaire | Self-administered first-step screening for depression and anxiety | Integrate into patient intake for at-risk groups |
| Mallampati Classification | Assesses Airway crowding | Visual assessment during oral exam |
| Friedman Tongue Position | Evaluates tongue size/position | Helps in SDB screening and referral decisions |
Awareness & patient education
Normalize menopause discussions. Position it as a routine life stage with oral and sleep implications. Provide take-home resources that connect oral findings to systemic context (e.g., why dry mouth raises caries risk, why non-refreshing sleep warrants medical evaluation). Use visuals and analogies to make mechanisms tangible, and ensure written, easy-to-understand instructions are accessible for patient handouts.1,11
Treatment & interprofessional collaboration
Oral symptom care includes salivary support, fluoride regimens, anti-inflammatory rinses, antifungal therapy when indicated, conservative TMD management, and rigorous periodontal maintenance. For sleep-disordered breathing, coordinate with primary care and sleep medicine for testing; provide OAT when appropriate with medical oversight and objective efficacy verification. Communicate bidirectionally with primary care physician (PCP), sleep physician, gynecologist, and behavioral health when SDB, mood or insomnia complicate care.13,30
Fig. 3
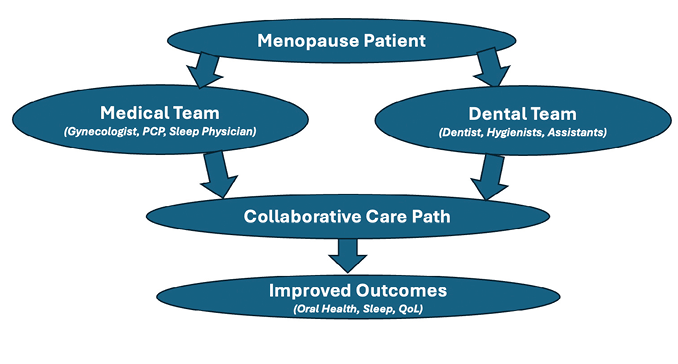
Ongoing support
Schedule closer recalls during the transition for at-risk patients. Monitor periodontal parameters, caries activity, mucosal comfort, implant status, and OAT side effects. Reinforce home-care routines, revisit sleep symptoms, and update referrals as status changes. Document outcomes and thoroughly explain care plans.13,15,25,30,39
Table 7: Menopause chairside screening checklist
| Screening tool | Rationale | Tool / method | Referral trigger |
| Age & Menstrual History | Identify peri/postmenopausal status | Patient Interview, health record | No menses > 12 months or classic symptoms |
| Menopausal Symptoms Questionnaire | Capture symptom severity and impact | Validated menopause symptom scales | Moderate / severe symptoms affecting function |
| Oral Exam | Identify oral manifestations linked to menopause | Full oral exam, periodontal charting | Periodontal disease, xerostomia, BMS, bone loss |
| Sleep History | Identify, Snoring, UARS, sleep apnea, insomnia risk | ESS, STOP-BANG or similar tools | High ESS, STOP-BANG score, or OSA suspicion |
| Medical History | Identify co-morbidities and medications affecting oral health | Health history form, medication list review | Complex medical issues, polypharmacy |
| Psychosocial Assessment | Identify mood, cognitive and social factors influencing care | PHQ-4, patient conversation | Severe mood disturbance, memory issues |
Table 8: Menopause myth and fact
| Menopause Myth | Menopause Myth |
| Menopause doesn’t affect the mouth | Estrogen decline alters inflammation, bone remodeling, and salivary function, increasing risk for periodontal disease, caries, and dysesthesia |
| Snoring is normal in midlife | Persistent snoring can signal SDB, which carries cardiometabolic and cognitive risks and warrants evaluation |
| If CPAP is hard, nothing else will help | Custom oral appliances are guideline-supported alternatives for many patients and can be combined with positional strategies and weight management to optimize outcomes |
Patient communication strategies
Sensitive, efficient conversations increase disclosure and adherence. Example openers:
- “Many women notice changes in dry mouth, taste, or gums around midlife. Have you noticed anything like that?”
- “Snoring can change over time. Has anyone mentioned snoring or pauses in your breathing during sleep?”
- “Non-refreshing sleep and morning headaches sometimes relate to airway issues—we can screen for those and coordinate testing if needed.”
Motivational interviewing tips: Ask open questions, affirm efforts, reflect ambivalence, and summarize a plan (“OARS”). Provide one or two written actions per visit to avoid overload.
Normalize referral: Part of comprehensive dental care is making sure that sleep and hormones aren’t working against your mouth. Offer follow-up touchpoints and celebrate small wins.
Table 9: Key resources for dental professionals
| Resource | Use in dental practice |
| North American Menopause Society (NAMS) | Up-to-date information on menopause management and guidelines1 |
| American Academy of Dental Sleep Medicine (AADSM) | Evidence based protocols for oral appliance therapy and OSA management13 |
| American Academy of Sleep Medicine (AASM) | Diagnostic and Treatment Guidelines30 |
| Evidence Based Dentistry resources | Salivary dysfunction, periodontal risk, and TMD management14-25 |
| National Institute of Aging | Patient-friendly education materials for menopause and health |
| Journal Articles/CE Courses | Continuing education for clinicians on menopause-related oral health |
Menopause: Summary of clinical red flags
- Rapid periodontal attachment loss despite stable biofilm control.2
- New or worsening xerostomia, dysgeusia, or mucosal burning.5
- Non-refreshing sleep, morning headaches, or bed-partner reports of snoring/pauses.6-8
- Daytime sleepiness or fatigue affecting work/safety; near-miss driving events.7
- TMD pain or limited opening that escalates with stress or sleep loss.20,24
- Recurrent candidiasis or angular cheilitis in a xerostomic patient.15,16
- Osteoporosis diagnosis, antiresorptive therapy, or history of fragility fracture prior to implant planning.25
Summary & conclusion
Menopause is both common and consequential for oral and airway health. Dental teams see menopausal patients regularly, and are well positioned to spot early signs, frame supportive conversations, refer when appropriate, reduce symptom burden, and participate in care. Ask explicitly about life-stage: many patients will not connect oral or sleep symptoms with menopause. A small set of structured questions, risk-focused examinations, preventive measures, and clear referral pathways can transform outcomes and improve quality of life dramatically. Build a referral network (primary care, gynecology, sleep medicine, behavioral health) and keep the loop closed with brief reporting letters (SOAP Notes). Making menopause-awareness a standing element of recall protocols advances patient-centered dentistry and strengthens interprofessional ties—with tangible benefits for oral, sleep, and overall health. 
Oral Health welcomes this original article.
Disclaimer: The author has no conflict of interest to disclose regarding any companies manufacturing/distributing products or services mentioned.
References
- North American Menopause Society. Menopause Practice: A Clinician’s Guide. 4th ed. Pepper Pike (OH): The North American Menopause Society; 2010.
- Goyal L, Goyal T, Gupta ND. Osteoporosis and periodontitis in postmenopausal women: a systematic review. J Midlife Health. 2017 Oct-Dec;8(4):151-158. doi:10.4103/jmh.JMH_55_17.
- El Khoudary SR, Greendale GA, Crawford SL, Avis NE, Brooks MM, Thurston RC, et al. The menopause transition and women’s health at midlife: a progress report from the Study of Women’s Health Across the Nation (SWAN). Menopause. 2019 Oct;26(10):1213-1227. doi:10.1097/GME.0000000000001424.
- Passos-Soares JS, Vianna MI, Gomes-Filho IS, Cruz SS, Barreto ML, Adan LF, et al. Association between osteoporosis treatment and severe periodontitis in postmenopausal women. Menopause. 2017 Jul;24(7):789-795. doi:10.1097/GME.0000000000000839.
- Silvestre FJ, Silvestre-Rangil J, López-Jornet P. Burning mouth syndrome: a review and update. Rev Neurol. 2015 May 16;60(10):457-463.
- Zaw M, Hein L, Martinez AC, et al. Gender differences in sleep-disordered breathing: a review of literature. Curr Pulmonol Rep. 2021;10:121-128. doi:10.1007/s13665-021-00273-3.
- Shahar E, Whitney CW, Redline S, Lee ET, Newman AB, Nieto FJ, et al. Sleep-disordered breathing and cardiovascular disease: cross-sectional results of the Sleep Heart Health Study. Am J Respir Crit Care Med. 2001 Jan;163(1):19-25. doi:10.1164/ajrccm.163.1.2001008.
- Bixler EO, Vgontzas AN, Lin HM, Ten Have T, Rein J, Vela-Bueno A, et al. Prevalence of sleep-disordered breathing in women: effects of gender. Am J Respir Crit Care Med. 2001 Mar;163(3 Pt 1):608-613. doi:10.1164/ajrccm.163.3.9911064.
- Antonaglia V, et al. Estrogen withdrawal, sleep fragmentation, and ventilatory instability in menopause. Sleep Med Rev. 2025;69:101763. doi:10.1016/j.smrv.2025.101763.
- Bromberger JT, Kravitz HM, Chang YF, Cyranowski JM, Brown C, Matthews KA. Major depression during and after the menopausal transition: Study of Women’s Health Across the Nation (SWAN). Psychol Med. 2011 Sep;41(9):1879-1888. doi:10.1017/S0033291711000207.
- Gao CC, Kapoor E, Lipford MC, Miller VM, Schroeder DR, Mara KC, et al. Association of vasomotor symptoms and sleep apnea risk in midlife women. Menopause. 2018 Apr;25(4):391-398. doi:10.1097/GME.0000000000001023.
- Weber MT, Rubin LH, Schroeder R, Steffenella T, Maki PM. Cognitive profiles in perimenopause: hormonal and menopausal symptom correlates. Climacteric. 2021 Aug;24(4):401-407. doi:10.1080/13697137.2020.1858479.
- Ramar K, Dort LC, Katz SG, Lettieri CJ, Harrod CG, Thomas SM, et al.; American Academy of Sleep Medicine; American Academy of Dental Sleep Medicine. Clinical practice guideline for the treatment of obstructive sleep apnea with oral appliance therapy: an American Academy of Sleep Medicine and American Academy of Dental Sleep Medicine joint guideline. J Clin Sleep Med. 2015 Jul 15;11(7):773-827. doi:10.5664/jcsm.4960.
- Chapple IL, Van der Weijden F, Doerfer C, Herrera D, Shapira L, Polak D, et al. Periodontal health and systemic conditions: consensus report of the joint EFP/AAP workshop. J Periodontol. 2013 Apr;84(4 Suppl):S164-S169. doi:10.1902/jop.2013.1340018.
- Scully C, Felix DH. Oral medicine—update for the dental practitioner: dry mouth and disorders of salivation. Br Dent J. 2005 Sep 10;199(7):423-427. doi:10.1038/sj.bdj.4812740.
- Villa A, Connell CL, Abati S. Diagnosis and management of burning mouth syndrome: a review and update. Oral Dis. 2018 Jul;24(6):879-890. doi:10.1111/odi.12785.
- Minicucci EM, Pires RB, Vieira RA, Miot HA, Sposto MR. Assessing the impact of menopause on salivary flow and xerostomia. Gerodontology. 2013 Jun;30(2):120-123. doi:10.1111/j.1741-2358.2012.00653.x.
- Labunet F, Popa C, Hâncu AI, et al. Oral health, salivary changes, and xerostomia in postmenopausal women: a scoping review. Medicina (Kaunas). 2025;61(5):837. doi:10.3390/medicina6100837.
- Soundarya N, Rao A, Hegde MN, et al. Salivary menopausal markers and oral health status. J Midlife Health. 2022;13(2):118-125. doi:10.4103/jmh.jmh_17_22.
- Ohrbach R, Sharma S. Behavioral therapy for temporomandibular disorders. Front Oral Maxillofac Med. 2021;3:37. doi:10.21037/fomm-21-16.
- Wang H, Han J, Yang Q, Dong Y, Wang Y. Effects of hormone replacement therapy on salivary estradiol and xerostomia in postmenopausal women. BMC Oral Health. 2021 Feb 26;21(1):157. doi:10.1186/s12903-021-01470-y.
- Baccolini V, da Silva LP, Teixeira L, de Sousa RT, Manarte-Monteiro P. The role of casein phosphopeptide-amorphous calcium phosphate (CPP-ACP) in white spot lesion remineralization: a systematic review. J Funct Biomater. 2025;16(8):272. doi:10.3390/jfb16080272.
- Goyal V, Damle S, Puranik MP, Nuvvula S, Kakanur M, Marwah N, et al. Arginine: a new paradigm in preventive oral care. Int J Clin Pediatr Dent. 2023 Sep-Oct;16(5):698-706. doi:10.5005/jp-journals-10005-2717.
- Schiffman E, Ohrbach R, Truelove E, Look JO, Anderson GC, Goulet JP, et al. Diagnostic criteria for temporomandibular disorders (DC/TMD) for clinical and research applications: recommendations of the International RDC/TMD Consortium Network and Orofacial Pain Special Interest Group. J Oral Facial Pain Headache. 2014 Winter;28(1):6-27. doi:10.11607/jop.1151.
- Bornstein MM, Cionca N, Mombelli A. Systemic conditions and treatments as risks for implant therapy. Periodontol 2000. 2019 Oct;81(1):7-24. doi:10.1111/prd.12291.
- Pivetta B, Chen L, Nagappa M, Saripella A, Waseem R, Englesakis M, et al. Use and performance of the STOP-Bang questionnaire for obstructive sleep apnea screening across geographic regions: a systematic review and meta-analysis. JAMA Netw Open. 2021 Mar 1;4(3):e211009. doi:10.1001/jamanetworkopen.2021.1009.
- Nagappa M, Liao P, Wong J, Auckley D, Ramachandran SK, Memtsoudis S, et al. Validation of the STOP-Bang questionnaire as a screening tool for obstructive sleep apnea among different populations: a systematic review and meta-analysis. PLoS One. 2015 Dec 14;10(12):e0143697. doi:10.1371/journal.pone.0143697.
- Scharf MT. Reliability and efficacy of the Epworth Sleepiness Scale: is there still a place for it? Nat Sci Sleep. 2022 Dec 13;14:2151-2156. doi:10.2147/NSS.S385407.
- Peña Orbea C, Gonzalez-Isaza P, Borsari G, Suárez-Iglesias D, Maturana-Ramirez A, Saaresranta T, et al. Predictive ability and reliability of the STOP-Bang questionnaire in screening for obstructive sleep apnea in midlife women. Maturitas. 2020 Aug;135:1-5. doi:10.1016/j.maturitas.2020.02.004.
- Epstein LJ, Kristo D, Strollo PJ Jr, Friedman N, Malhotra A, Patil SP, et al. Clinical guideline for the evaluation, management and long-term care of obstructive sleep apnea in adults. J Clin Sleep Med. 2009 Jun 15;5(3):263-276.
- Sutherland K, Phillips CL, Cistulli PA. Efficacy versus effectiveness in the treatment of obstructive sleep apnea: CPAP and oral appliances. J Dent Sleep Med. 2015;2(4):175-181. doi:10.15331/jdsm.5072.
- Chai-Coetzer CL, Luo YM, Antic NA, Zhang XL, Chen BY, He QY, et al. Predictors of long-term adherence to continuous positive airway pressure therapy in patients with obstructive sleep apnea and cardiovascular disease in the SAVE study. Sleep. 2013 Dec;36(12):1929-1937. doi:10.5665/sleep.3232.
- Bartlett D, Wong K, Richards D, Moy E, Espie CA, Cistulli PA, et al. Increasing adherence to obstructive sleep apnea treatment with a group social cognitive therapy intervention: a randomized trial. Sleep. 2013 Nov;36(11):1647-1654. doi:10.5665/sleep.3114.
- Catcheside PG. Predictors of continuous positive airway pressure adherence. F1000 Med Rep. 2010;2:70. doi:10.3410/M2-70.
- Phillips CL, Grunstein RR, Darendeliler MA, Mihailidou AS, Srinivasan VK, Yee BJ, et al. Health outcomes of continuous positive airway pressure versus oral appliance treatment for obstructive sleep apnea: a randomized controlled trial. Am J Respir Crit Care Med. 2013 Apr 15;187(8):879-887. doi:10.1164/rccm.201212-2223OC.
- Errazuriz P, Crisostomo N, Gonzalez J, Aguilera P, Gomez J, Plaza M, et al. Validation of the PHQ-2 among Spanish-speaking Latin American immigrants. Int J Environ Res Public Health. 2022 Nov 4;19(21):13975. doi:10.3390/ijerph192113975.
- American Psychological Association. Patient Health Questionnaire (PHQ) overview. Washington (DC): American Psychological Association; 2025 [cited 2025 Aug 1]. Available from: https://www.apa.org/pi/about/publications/caregivers/practice-settings/assessment/tools/patient-health.
- Li C, Friedman B, Conwell Y, Fiscella K. Validity of the Patient Health Questionnaire 2 (PHQ-2) in identifying major depression in older people. J Am Geriatr Soc. 2007 Apr;55(4):596-602. doi:10.1111/j.1532-5415.2007.01103.x.
- Miller WR, Rollnick S. Motivational interviewing in health care: helping patients change behavior. New York: Guilford Press; 2002.
About the author

John Viviano, DDS, D, ABDSM obtained his credentials from the University of Toronto in 1983. His clinic is limited to managing sleep disordered breathing and bruxism. He is a Credentialed Diplomate of the American Board of Dental Sleep Medicine and has lectured internationally, conducted original research, and authored original articles on the management of sleep-disordered breathing. His clinic was the first Canadian facility accredited by the American Academy of Dental Sleep Medicine, and he is Clinical Director of the Sleep Disorders Dentistry Research and Learning Centre.
