Abstract
Hypothyroidism is one of the most prevalent diseases in the world and many patients go undiagnosed. The most common population with hypothyroidism is elderly women, but it can happen to anyone. It results when there is an inadequate production of thyroid hormone. Thyroid hormone has effects on all organs throughout the body, and when there is less hormone patients have a wide variety of symptoms. Symptoms vary from mild ones all the way to life threatening myxedema coma. It is important for all clinicians to be aware of the symptoms of hypothyroidism so that patients can be treated appropriately and dangerous situations can be prevented from arising. This paper will go into depth on what causes hypothyroidism, various manifestations of the disease, and the medical and dental management of it.
Epidemiology
Hypothyroidism is a common disease that results from the inadequate production of thyroid hormone or from the inadequate action of thyroid hormone in target tissues. Lack of this hormone has a multitude of effects including changes in metabolism, growth, and dysfunction of organ systems. Hypothyroidism is a very common disease with a higher prevalence in women and the elderly. National Health and Nutrition Examination Survey III data estimates that 3 in every 1000 (0.3%) Americans have overt hypothyroidism and 43 in every 1000 (4.3%) have subclinical hypothyroidism.1 A meta-analysis done in nine European countries estimates prevalence of undiagnosed hypothyroidism is estimated to be around 5-6%.2 A study in Leiden, Netherlands, found the prevalence of overt hypothyroidism to be 7% in a study of 558 subjects aged between 85 and 89.3 This high rate of hypothyroidism in the elderly population signifies that the prevalence of hypothyroidism increases significantly with age. A 20-year follow-up of the Whickham Survey in Great Britain found the mean incidence of spontaneous hypothyroidism in men was 0.6/1000 survivors per year and in women was 3.5/1000 survivors per year.4 Overall, hypothyroidism is up to 8-9 times more common in women with a peak incidence between the ages of 30 and 50 years.5 Due to its prevalence, it is important for dental practitioners to be well informed about the disease and how to best manage these patients in order to provide the utmost care.
Thyroid gland physiology
Anatomy
The thyroid is a small gland located under the larynx and it starts forming around day 20 in utero. It consists of two lobes connected by an isthmus that crosses the midline at the level of the first or second tracheal cartilaginous rings. The functional unit of the thyroid gland is called a follicle. There are hundreds of follicles in the gland, each consisting of epithelial follicular cells, also called thyrocytes, and colloid. Thyrocytes form a monolayered epithelium that surrounds the lumen filled with colloid, which serves as a reservoir for thyroid hormone.6 There are multiple other cells present in the thyroid, including neuroendocrine cells, fibroblasts and macrophages, but this paper will focus on the major cell type of the thyroid gland which is the thyrocyte. There are several extrinsic and intrinsic factors that are needed for normal development of the thyroid gland. The four main transcription factors that are present in precursors of thyrocytes that differentiate these cells from others in the pharynx include Hhex, Nkx2-1, Pax8, and Foxe1.7 If one of these transcription factors is absent, thyroid morphogenesis is severely impaired. The thyroid gland is completely developed by week 11-12 in utero and starts to secrete thyroid hormones around week 16.8 The two main hormones the thyroid secretes are triiodothyronine (T3) and tetraiodothyronine (T4).
Mechanism of hormone synthesis
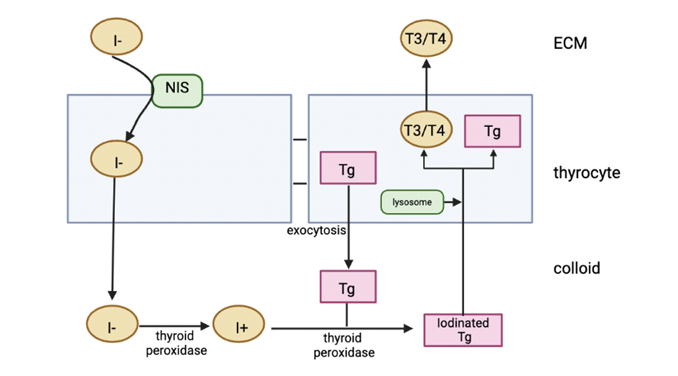
Synthesis of T3 and T4 is a multistep process that requires iodine. Adequate iodide intake is important in order to produce sufficient thyroid hormone. The minimum requirement of iodide is 75 micrograms per day and this is easily met in North America from iodized salt and dairy products. Iodide needs to be transported into the colloid for it to be incorporated into the thyroid hormone. Thyrocytes have tight junctions that form a strong barrier preventing diffusion through intercellular spaces; therefore, the iodide needs to be transported through a channel. Transport of iodide into thyrocytes is achieved by Na/I symporter (NIS) that are present on the basolateral plasma membrane. Then, iodide needs to be transported into the colloid through the apical plasma membrane which is facilitated by pendrin, ClC5, and anoctamin-1.9 The activity of these transporters is dependent on the actions of thyroid stimulating hormone (TSH), which will be discussed shortly. Once iodide is in the colloid, it must be oxidized by thyroid peroxidase into iodine for it to be utilized in the production of thyroid hormones.9 Once iodide is oxidized to iodine, it is ready to be used to create the thyroid hormone. Severe iodine deficiency, which prevents formation of thyroid hormone, is one of the most common causes of intellectual disability in the world.8
Thyroid hormone synthesis starts in the hypothalamus with the release of thyrotropin releasing hormone (TRH) which stimulates the anterior pituitary to release thyroid stimulating hormone (TSH). TSH goes and binds to GPCR on thyrocytes in the thyroid gland. This activates adenylate cyclase through Gs alpha protein which increases cAMP levels leading to activation of protein kinase A (PKA).9 PKA phosphorylates different proteins and modifies their function, allowing for the production of thyroid hormones. Thyroglobulin is a large glycoprotein made in thyrocytes and it serves as a template for thyroid hormone synthesis. TSH induces rapid simulation of exocytosis of thyroglobulin into the colloid. Once thyroglobulin is in the colloid, iodine is added to tyrosine residues by thyroid peroxidase and the iodinated tyrosine residues are then modified and joined to form T3 and T4. At this point the T3 and T4 are still attached to thyroglobulin. In order to secrete the hormones, the entire thyroglobulin molecule is endocytosed into the thyrocyte and joins with a lysosome, which has enzymes that cleave T3 and T4 from thyroglobulin.10 The thyroglobulin molecule is then recycled. T3 is the more active metabolite compared to T4. When there is less iodine available, more T3 is created compared to T4.11 T4 acts as a prohormone and most of it is converted to T3 by 50-monodeiodination in the liver, kidneys, and skeletal muscle.12 All of the steps in the formation of thyroid hormone occur due to the activation of GPCRs by TSH. When there is too much T3 and T4, there is a negative feedback loop and T3/T4 inhibit release of TRH. Once T3 and T4 are produced, they are released into the bloodstream and they exert their effects on all organs in the body (Fig. 1).
Fig. 1
Function of thyroid hormones
Thyroid hormones have many different functions throughout the body and affect tissues at different stages of development. Most of the actions of thyroid hormones are the result of potentiation of other signalling pathways.13 One important function is that the thyroid hormone stimulates the metabolic rate and controls oxygen consumption. It can cause an increase in the number and size of mitochondria and stimulates energy production by switching metabolism from the glycolytic pathway to more efficient oxidative phosphorylation.14 TSH has also been shown to act directly on adipocytes and trigger lipolysis.15 Therefore, absence of thyroid hormone leads to decreased energy, weight gain, and decreased tolerance to cold. This hormone is critical in growth and development as well. It promotes proliferation of cells and it helps virtually all organs to mature in the body.16 This includes growth of bones, skin, liver, pancreas, kidneys, and heart just to name a few. Thyroid hormone also plays a complex yet important role in neuronal development in the nervous system. Thyroid hormone helps to increase alertness, enhances memory, increases speed of nerve reflexes, and helps with retinal development.16 When there is a thyroid hormone deficiency, patients have significant neurologic deficits and growth deficiency.
Thyroid hormone has multiple effects on the cardiovascular system too. It increases heart rate, increases cardiac contractility, increases stroke volume, and reduces systemic vascular resistance.17 Since thyroid hormone is important in development of kidneys, it indirectly has an impact on renin production. Patients with hypothyroidism have been shown to have decreased plasma renin activity.17 These changes in the cardiovascular system in patients with decreased levels of thyroid hormone contribute to many cardiovascular problems, such as heart failure and high blood pressure. Overall, it is important to understand the functions of thyroid hormones in order to recognize the symptoms that one experiences when there is inadequate production of it.
Etiology
There can be several different causes of hypothyroidism. It can be split into primary hypothyroidism, which is due to problems in the thyroid gland itself, or central hypothyroidism, which is due to problems of the pituitary gland or hypothalamus. In primary hypothyroidism, there are increased levels of TSH in the blood but free T4 levels are reduced. In areas of adequate iodine, the most common cause of primary hypothyroidism is Hashimoto’s disease. This is an autoimmune disease where autoantibodies are directed against thyroglobulin and thyroid peroxidase.18 A multitude of genetic and environmental factors lead to autoantibodies developing; however, the exact mechanism is not known. Other causes of primary hypothyroidism include iodine deficiency, neck radiation, hemithyroidectomy, and radioiodine therapy.18 Radioiodine therapy is used to reduce thyroid function in patients with Grave’s disease, which is a disease with increased thyroid hormone synthesis, however 75% of these patients develop hypothyroidism.18 Also, excess iodide can sometimes lead to hypothyroidism as well in a phenomenon called the Wolff-Chaikoff effect. The thyroid gland has an intrinsic regulatory mechanism to prevent hyperthyroidism when there is excess iodine. Studies were done in which rats were given excess iodine, and it was determined that there was a reduction in thyroid hormone synthesis for 24 hours, termed the Wolff-Chaikoff effect. The exact mechanism of this process is not fully understood but involves the generation of inhibitory substances such as intrathyroidal iodolactones in the thyroid gland.19 In humans and rats, if iodine levels remain high, there is a subsequent reduction in the active transport of iodine into the thyroid gland which overcomes this phenomenon.19 If an individual is not able to escape from the Wolff-Chaikoff effect, increased iodine levels can lead to hypothyroidism. Furthermore, there are certain medications that make an individual more prone to hypothyroidism, including amiodarone, lithium, sunitinib, interferon, and certain medication for epilepsy such as carbamazepine and valproic acid.18 Subclinical hypothyroidism occurs when there is reduced thyroid function but the body is able to maintain a normal level thyroxine level by increasing production of TSH.
Central hypothyroidism is much less common and presents with decreased TSH and decreased thyroxine. It can be due to congenital defects in the hypothalamus or pituitary gland, leading to less production of TRH and TSH. Central hypothyroidism can also be acquired due to tumours or trauma to either the hypothalamus or the pituitary gland. Pituitary adenomas account for more than 50% of cases of central hypothyroidism.18 There are also certain medications linked with central hypothyroidism. Rexinoids, used in the treatment of certain malignancies and metabolic orders, have been found to cause clinically significant central hypothyroidism.20 These patients have to be supplemented with additional thyroid hormones. Dopamine agonists do not directly cause hypothyroidism but may exacerbate clinical hypothyroidism.20 One thing that is important to screen for is congenital hypothyroidism. Congenital hypothyroidism happens due to absent thyroid gland, ectopic location of thyroid gland, hypoplastic thyroid gland, or due to mutations in any of the steps in thyroid hormone biosynthesis.21 If this condition is not treated promptly, babies develop severe intellectual disabilities.21 The incidence of congenital hypothyroidism is estimated to be 1 in 3,500-4,000 births.22 Screening for congenital hypothyroidism is critical at birth in order to intervene early and prevent side effects of hypothyroidism.
Table 1: Causes of primary and central hypothyroidism
| Type of hypothyroidism | Causes |
| Primary hypothyroidism | – Hashimoto’s disease- Iodine deficiency – Neck radiation – Hemithyroidectomy – Radioiodine therapy – Wolff-Chaikoff effect – Some medications: amiodarone, lithium, sunitinib, interferon, carbamazepine, valproic acid |
| Central hypothyroidism | – Hypothalamus or pituitary gland congenital defect – Tumour – Trauma – Medications: rexinoids, dopamine agonists |
Clinical manifestations
Hypothyroidism has a wide array of symptoms, from some who are asymptomatic all the way to life-threatening symptoms such as myxedema coma. Patients with hypothyroidism have decreased metabolic rate, therefore they feel more tired and weak. Other symptoms include dry skin, brittle hair, voice distortion, decreased appetite, constipation, memory loss, increase in weight, and renal impairment.18 Hypothyroidism has a big impact on the cardiovascular system as well. It causes a decrease in heart rate, decreases stroke volume, increases vascular resistance, and elevates cholesterol levels.18
Myxedema coma is the most serious complication of hypothyroidism and the incidence rate is 0.22/1,000,000 per year.23 It is a rare condition but it is important to recognize in order to increase the chance of survival should it occur. The two cardinal features of myxedema coma are hypothermia and altered mental status. Other symptoms include respiratory failure, arrhythmias, hypotension, hypoglycemia, hyponatremia, and impaired immune response.23 The body is in a lot of distress and all the organ systems start shutting down. It occurs in longstanding, severe hypothyroidism or can be triggered by acute stressors such as infection, trauma, and surgery in patients with uncontrolled hypothyroidism.24 The dental office can be a source of stress which may potentially trigger myxedema coma. Some cases of myxedema coma happen when patients have been on thyroid hormone therapy but stop it for no apparent reason. Therefore, it is always critical to take a proper medical history in order to determine the level of control of the disease. The most common patient population to have myxedema coma are hospitalized elderly women, with 80% of cases being women who are >60 years of age.23 Although it is most common in the elderly, it can happen in younger patients as well. Myxedema coma has a 30% mortality rate; therefore, treatment should begin immediately while waiting for laboratory confirmation.24
Oral manifestations
Since patients visit the dentist regularly, often more than their family physician, certain diseases including hypothyroidism may be detected first by dentists. Therefore, it is important for dentists to know the oral manifestations of disease in order to avoid potential complications and guide patients to appropriate care. Patients with congenital hypothyroidism have vertical facial growth, small jaws, delayed eruption of teeth, short roots, changes in enamel and malocclusion. They also have macroglossia, thick lips, geographic tongue, and enlargement of salivary glands.22 The internal aspect of the ramus does not resorb normally, causing insufficient space for the eruption of mandibular second molars leading to crowding and impaction.25 Untreated hypothyroidism in adults contributes to poor periodontal health.25 Overall, these symptoms are not very specific to hypothyroidism, however they reinforce that systemic diseases do have an impact on the oral cavity so it is important to consider when providing dental treatment.
Diagnosis
As mentioned previously, hypothyroidism has a wide variety of symptoms and they are nonspecific. Since symptoms are nonspecific and are often attributed to aging, there is a high rate of undiagnosed hypothyroidism. If the disease is suspected, the only way to confirm is through laboratory testing. Overt hypothyroidism is diagnosed when the TSH level is > 10 mIU/L.26 Hashimoto’s disease can be diagnosed by the presence of antibodies against thyroid peroxidase and thyroglobulin.27 Overall, if hypothyroidism is suspected, patients should be encouraged to get testing in order to come up with an accurate diagnosis and prevent complications.
Medical management
Hypothyroidism is managed by providing a synthetic form of thyroxine called levothyroxine. This drug has a long half life so it only has to be taken once daily. It is recommended when patients have overt hypothyroidism (> 10 mIU/L), however the threshold is lower for pregnant and younger patients.28 Most pregnant women who are on levothyroxine require a higher dose in order to maintain euthyroidism during pregnancy.29 When a patient is anti-TPO positive, international guidelines recommend levothyroxine when TSH is > 4 mIU/L.28 The average dose of levothyroxine is approximately 1.6 mcg/kg/day for adults and steady TSH concentrations are not achieved for a minimum of 6 weeks.28 This means that the hypothyroidism is not under control for quite some time once patients start taking medication. Patients with cardiac conditions or elderly patients should start with a lower dose of levothyroxine, between 12.5 and 50 mcg, and then slowly titrate up.30 Thyroid function should initially be tested every 6 to 8 weeks. Then, once TSH levels reach reference ranges, annual exams are sufficient. If the TSH levels stay high despite administration of > 1.9 mcg/kg/day of levothyroxine, the patient has refractory hypothyroidism and further examination of other causes is indicated.31 Levothyroxine should be taken on an empty stomach because food impairs absorption, with a minimum of 30 minutes before breakfast.28 Levothyroxine is one of the most prescribed drugs in the world, so it is important for all clinicians to be aware of it.
Dental management
Dental management of hypothyroidism first starts by taking a detailed history of the condition. It is important to determine the level of control since this dictates if treatment can proceed. If the patient has hypothyroidism and is not taking medication or recently started taking levothyroxine, elective treatment should be deferred. This is because it takes 6-8 weeks for TSH levels to stabilize. If emergency treatment is needed, patients should be referred to a hospital dental clinic. If the control is not known, a medical letter should be sent to the patient’s family physician or endocrinologist. Patients that are compliant with their medication and have controlled hypothyroidism are good to receive dental treatment and no modifications are necessary. It is important to know the signs and symptoms of hypothyroidism since so many people are undiagnosed. If it is suspected, the patient should be referred to their family physician for laboratory testing.
If myxedema coma is suspected in a dental clinic, then 911 should be called immediately and patients are admitted to the intensive care unit. The patient requires careful management due to multiple issues presenting simultaneously. First step is to have prompt initiation of thyroid hormone therapy, even before having the thyroid hormone results back.32 Intravenous (IV) hydrocortisone is recommended to be administered prior to IV levothyroxine to prevent adrenal crises. The dose of hydrocortisone should be 100 mg IV initiating dose (total 200-400 mg daily) which can be weaned down based on cortisol levels as patient stabilizes. The initial recommended dose of IV levothyroxine is 200-400 mcg and subsequent dosing is based on 1.6 mcg/kg/day reduced to 75% when given as IV.32 Hypothermia is managed with blankets and increasing the room temperature, while balancing negative side effects such as peripheral vasodilation which can lead to hypotension and shock.32 There are many other complications of myxedema coma that have to be considered in the ICU including hyponatremia and hypoglycemia. It is best to prevent this from happening in the first place by taking a detailed medical history and making sure hypothyroidism is controlled.
Conclusion
Overall, hypothyroidism is a very common disease and dentists are sure to encounter patients that have it on a regular basis. If hypothyroidism is suspected, dentists should refer the patient to their physician for laboratory testing. Most patients with hypothyroidism are well controlled and are taking levothyroxine. If the level of control is unclear, it is best to send a medical letter. Patients with uncontrolled hypothyroidism should not receive dental treatment outside of a hospital. Some patients may get angry when dental care is refused, but it is important to reinforce to them that it is for their best interest and well-being. Being vigilant will prevent emergencies such as myxedema coma from occurring in the dental chair and will allow the dentist to provide safe treatment to all patients. 
Oral Health welcomes this original article.
References
- Almandoz, J. P., & Gharib, H. (2012). Hypothyroidism: etiology, diagnosis, and management. Medical Clinics of North America, 96(2), 203-221. https://doi.org/10.1016/j.mcna.2012.01.005
- Garmendia Madariaga, A., Santos Palacios, S., Guillén-Grima, F., & Galofré, J. C. (2014). The incidence and prevalence of thyroid dysfunction in Europe: a meta-analysis. The Journal of clinical endocrinology and metabolism, 99(3), 923–931. https://doi.org/10.1210/jc.2013-2409
- Vanderpump M. P. (2011). The epidemiology of thyroid disease. British medical bulletin, 99, 39–51. https://doi.org/10.1093/bmb/ldr030
- Vanderpump, M. P. J., Tunbrldge, W. M. G., French, J. M., Appleton, D., Bates, D., Clark, F., Evans, J. G., Hasan, D. M., Rodgers, H., Tunbridge, F., & Young, E. T. (1995). The incidence of thyroid disorders in the community: a twenty-year follow-up of the Whickham Survey. Clinical Endocrinology, 43(1), 55–68. https://doi.org/10.1111/j.1365-2265.1995.tb01894.x
- Chiovato, L., Magri, F., & Carlé, A. (2019). Hypothyroidism in context: where we’ve been and where we’re going. Advances in therapy, 36(Suppl 2), 47–58. https://doi.org/10.1007/s12325-019-01080-8
- Nilsson, M., & Fagman, H. (2017). Development of the thyroid gland. Development, 144(12), 2123–2140. https://doi.org/10.1242/dev.145615
- De Felice, M., & Di Lauro, R. (2011). Minireview: Intrinsic and Extrinsic Factors in Thyroid Gland Development: An Update. Endocrinology, 152(8), 2948–2956. https://doi.org/10.1210/en.2011-0204
- Obregon, M. J., Calvo, R. M., Escobar Del Rey, F., & Morreale de Escobar, G. (2007). Ontogenesis of thyroid function and interactions with maternal function. Endocrine development, 10, 86–98. https://doi.org/10.1159/000106821
- Carvalho, D. P., & Dupuy, C. (2017). Thyroid hormone biosynthesis and release. Molecular and Cellular Endocrinology, 458, 6-15. https://doi.org/10.1016/j.mce.2017.01.038
- Shahid, M. A., & Sharma, S. (2023). Physiology, Thyroid Hormone. Nih.gov; StatPearls Publishing. https://www.ncbi.nlm.nih.gov/books/NBK500006/
- Markou, K., Georgopoulos, N., Kyriazopoulou, V., & Vagenakis, A. G. (2001). Iodine-Induced Hypothyroidism. Thyroid, 11(5), 501–510. https://doi.org/10.1089/105072501300176462
- Farhadi, S. A. S., & Dizaye, K. F. (2019). Aliskiren, Fosinopril, and Their Outcome on Renin-Angiotensin-Aldosterone System (RAAS) in Rats with Thyroid Dysfunction. International journal of endocrinology, 2019, 5960563. https://doi.org/10.1155/2019/5960563
- Brent G. A. (2012). Mechanisms of thyroid hormone action. The Journal of clinical investigation, 122(9), 3035–3043. https://doi-org.myaccess.library.utoronto.ca/10.1172/JCI60047
- Noli, L., Khorsandi, S. E., Pyle, A., Giritharan, G., Fogarty, N., Capalbo, A., Devito, L., Jovanovic, V. M., Khurana, P., Rosa, H., Kolundzic, N., Cvoro, A., Niakan, K. K., Malik, A., Foulk, R., Heaton, N., Ardawi, M. S., Chinnery, P. F., Ogilvie, C., Khalaf, Y., Ilic, D. (2020). Effects of thyroid hormone on mitochondria and metabolism of human preimplantation embryos. Stem cells (Dayton, Ohio), 38(3), 369–381. https://doi-org.myaccess.library.utoronto.ca/10.1002/stem.3129
- Teixeira, P. D. F. D. S., Dos Santos, P. B., & Pazos-Moura, C. C. (2020). The role of thyroid hormone in metabolism and metabolic syndrome. Therapeutic advances in endocrinology and metabolism, 11. https://doi.org/10.1177/204201882091786
- Pascual, A. & Aranda, A. (2013). Thyroid hormone receptors, cell growth and differentiation. Biochimica et Biophysica Acta, 1830(7), 3908-3916. https://doi.org/10.1016/j.bbagen.2012.03.012
- Barreto-Chaves M. L. M., Carrillo-Sepúlveda, M. A., Carneiro-Ramos, M. S., Gomes, D. A., & Diniz, G. P. (2010). The crosstalk between thyroid hormones and the renin-angiotensin system. Vascular Pharmacology, 52(3-4), 166-170. https://doi.org/10.1016/j.vph.2009.10.009
- Zamwar, U. M., & Muneshwar, K. N. (2023). Epidemiology, Types, Causes, Clinical Presentation, Diagnosis, and Treatment of Hypothyroidism. Cureus, 15(9), e46241. https://doi.org/10.7759/cureus.46241
- Sohn, S. Y., Inoue, K., Rhee, C. M., & Leung, A. M. (2024). Risks of Iodine Excess. Endocrine Reviews. https://doi.org/10.1210/endrev/bnae019
- Haugen, B. R. (2009). Drugs that suppress TSH or cause central hypothyroidism. Best Practice & Research Clinical Endocrinology & Metabolism, 23(6), 793-800. https://doi.org/10.1016/j.beem.2009.08.003
- Abduljabbar, M. & Afifi, A. (2012). Congenital hypothyroidism. Journal of Pediatric Endocrinology and Metabolism, 25(1-2), 13-29. https://doi-org.myaccess.library.utoronto.ca/10.1515/jpem.2011.408
- Miulescu, R. E. D., & Guja, C. (2019). Oral pathology in overt thyroid diseases. The Publishing House of the Romanian Academy, 21(2), 125-6.
- Wartofsky, L., & Klubo-Gwiezdzinska, J. (2019). Myxedema coma. The thyroid and its diseases: a comprehensive guide for the clinician, 281-292.
- Elkattawy, S., Dhanoa, P., Kotys, J., Fichadiya, H., & Eckman, A. (2021). Myxedema Coma: Case Report and Literature Review. Cureus, 13(5), e15277. https://doi.org/10.7759/cureus.15277
- Al-Yasiry, A. & Al-Jammali, M. (2020). Oral Manifestation for Patients with Thyroid Dysfunction and it’s Management in Dental Clinic -A Review. International Journal of Drug Research and Dental Science, 2(1) 23-26. https://doi.org/10.36437/ijdrd.2020.2.1.O
- Gottwald-Hostalek, U., & Schulte, B. (2022). Low awareness and under-diagnosis of hypothyroidism. Current medical research and opinion, 38(1), 59–64. https://doi.org/10.1080/03007995.2021.1997258
- Vargas-Uricoechea, H. (2023). Molecular Mechanisms in Autoimmune Thyroid Disease. Cells, 12(6), 918. https://doi.org/10.3390/cells12060918
- Hughes, K., & Eastman, C. (2021). Thyroid disease: Long-term management of hyperthyroidism and hypothyroidism. AJGP, 50, 1-2. doi: 10.31128/AJGP-09-20-5653
- Pearce E. N. (2022). Management of Hypothyroidism and Hypothyroxinemia During Pregnancy. Endocrine practice : official journal of the American College of Endocrinology and the American Association of Clinical Endocrinologists, 28(7), 711–718. https://doi-org.myaccess.library.utoronto.ca/10.1016/j.eprac.2022.05.004
- Wilson, S. A., Stem, L. A., & Bruehlman, R. D. (2021). Hypothyroidism: Diagnosis and Treatment. American family physician, 103(10), 605–613.
- Liu, H., Li, W., Zhang, W., Sun, S., & Chen, C. (2023). Levothyroxine: Conventional and Novel Drug Delivery Formulations. Endocrine reviews, 44(3), 393–416. https://doi-org.myaccess.library.utoronto.ca/10.1210/endrev/bnac030
- Elshimy, G., Chippa, V., & Correa, R. (2023). Myxedema. In StatPearls. StatPearls Publishing.
About the author

Dr. Deepak Sran graduated from the Faculty of Dentistry at the University of Toronto in 2025 and is currently doing a general practice residency at Princess Margaret Hospital.

Dr. Aviv Ouanounou is an associate professor of Pharmacology & Preventive Dentistry at the Faculty of Dentistry, U of Toronto. He is often invited to deliver lectures locally, nationally and internationally. He is the recipient of the 2014-2015 Dr. Bruce Hord Master Teacher Award for excellence in teaching and the 2018-2019 National W.W. Wood Teaching Award for Excellence in Dental Education. Dr. Ouanounou maintains a private practice in Toronto and is the corresponding author for this article. Reach him at aviv.ouanounou@dentistry.utoronto.ca.