Collectively, periodontal disease and dental caries are among the most common diseases worldwide and are leading causes of tooth loss.1 They not only diminish quality of life and systemic health but also impose significant burdens on public health systems.2,3
For decades, clinicians have relied predominantly on visual assessment tools to detect and classify periodontal disease. While visual parameters and probing depths can indicate the presence of the active disease, they may not accurately predict risk. Similarly, the most reliable predictor of risk for caries is the presence of at least one carious lesion. Both diseases are complex and influenced by diet, age, oral hygiene, smoking, diabetes, lifestyle, and genetics. Ideally, it would be best to assess risk, before visual signs manifest.
For clinicians, how would access to specific genetic risk information change treatment and prevention strategies? Furthermore, how might this knowledge impact patient compliance and acceptance of care plans?
A new revolutionary assessment tool
AI Genetics, a Canadian company, has recently introduced OralRiskTM (distributed by Curion) (Fig. 1), a chairside salivary test. This test assesses genetic susceptibility and risk for developing periodontal disease and dental caries using a polygenic risk score.4 This score compares an individual’s risk against population baselines, derived from identified genetic variants associated with each condition through Genome-Wide Association Studies.5,6 Approximately 35 genetic markers for caries and 25 for periodontal disease have been identified, and AI technology, along with its vast mathematical and analytical capabilities, enables comprehensive evaluation of these markers, offering insights previously inaccessible.5
Fig. 1

Differentiating salivary tests
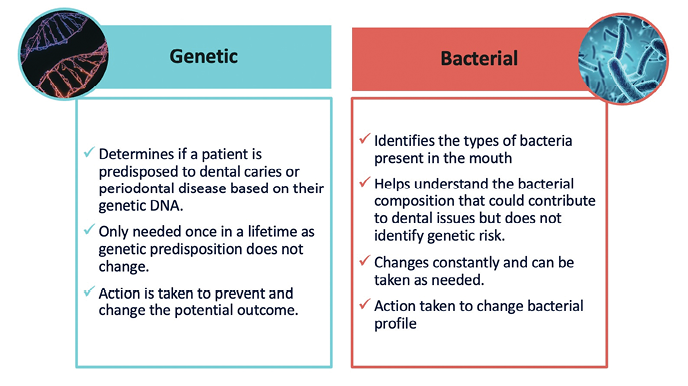
Salivary bacterial tests have been available for a long time. These traditional tests have been valuable tools for assessing risk by offering information on an individual’s pathogen profile and load.7 These tests can be taken often to assess the quantity and quality of pathogenic bacteria and help determine appropriate therapeutic measures to reduce the pathogenic burden. Unlike these tests, OralRiskTM genetic testing is a one-time assessment, as genetic risk is inherent and stable over time, contrasting with the fluctuating nature of bacterial profiles. It would be important to understand that bacterial tests do not assess a patient’s personal DNA or genetic predisposition (Fig. 2).
Fig. 2
The impact
Many patients overlook periodontal care or underestimate the importance of oral health maintenance. Leveraging technology like genetic testing can potentially increase patient acceptance of necessary treatments and foster more productive dialogues, encouraging proactive approaches to disease prevention rather than feeling resigned to their inevitability.
Target patients
Ideal candidates for genetic testing include those with familial, medical, or clinical histories that include high risks of periodontal disease, caries, diabetes, smoking, weight issues, dry mouth, stress, dietary concerns, and other associated lifestyle factors. The test would also be indicated before periodontal therapies, periodontal surgeries, implant cases, cosmetic or restorative work, and orthodontic treatment, for both knowledge and patient risk profile for success. It could also serve as a new patient service emphasizing personalized care.
Case report
One recent example of the impact of this technology came to the author from a 35-year-old female patient. As a teenager, her mother developed periodontal disease that led to the complete loss of her dentition. The impact this had on her ability to enjoy food, her social life, her mental health, and overall confidence was overwhelming. Witnessing her mother’s ordeal did not initially prompt the patient to consider her personal susceptibility. She was offered a genetic test, which she took and received the results as high risk. Remembering what she witnessed growing up, she immediately acted and spoke to the dental team to put together a proactive prevention plan. Her change in action and dedication underscores the potential of genetic insights to drive patient commitment to proactive health management.
The test process
The genetic test is simple and chairside, taking five minutes or less. The test kit includes a test vial, detailed instructions for use, registration information, and a prepaid return envelope to send with the specimen for analysis (Fig. 3). The test comes with a cheek swab device that is used by rubbing the swab along the gums or cheek for 10 seconds on each side to collect patient cells. The test is then placed inside the collection vial, sealed, shaken and placed into the prepaid envelope to Mount Sinai Hospital in Toronto for analysis. The office and the patient are notified by email when the results are available, and these can be accessed through their personal and clinical portals. The report received by the patient and the practice will show the patient’s genetic susceptibility on a graph depicted as low, moderate or high risk (Fig. 4).
Fig. 3
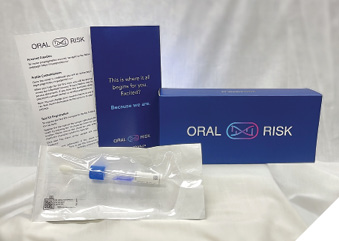
Fig. 4
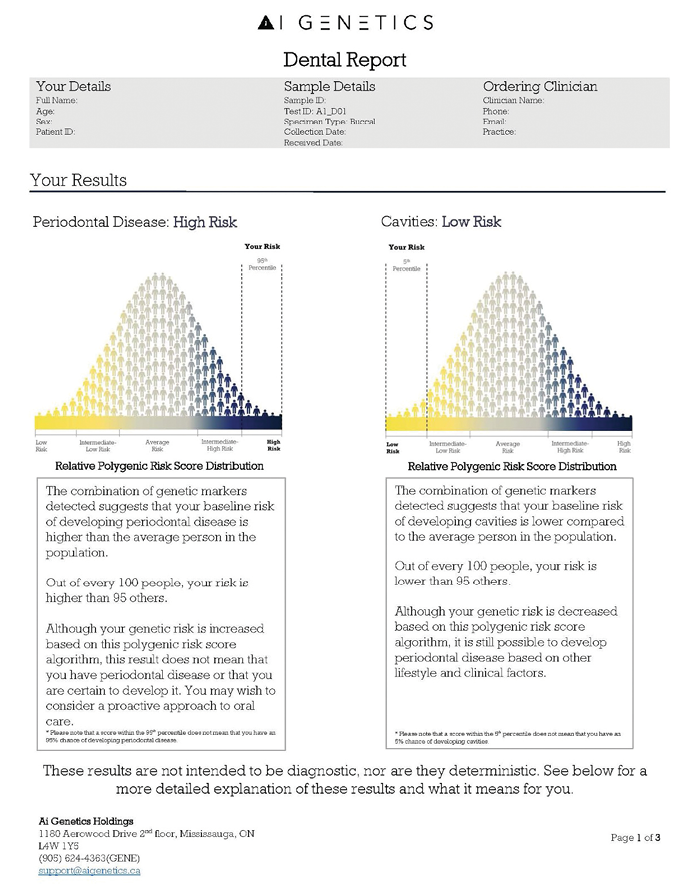
Conclusion
Genetic testing, supported by the analytical power of AI, presents a new frontier in dentistry that could forever change the approach to risk assessment and prevention strategies. Collaboratively, patients and oral health care providers can understand risks and intentionally minimize them to secure the most successful outcomes. 
Oral Health welcomes this original article.
References
- Baima G, Shin HS, Arrica M, Laforí A, Cordaro M, Romandini M. The co-occurrence of the two main oral diseases: periodontitis and dental caries. Clin Oral Investig. 2023;27(11):6483-6492. doi:10.1007/s00784-023-05253-2
- Featherstone JD. Caries prevention and reversal based on the caries balance. Pediatr Dent. 2006;28(2):128-198.
- Listl S, Galloway J, Mossey PA, Marcenes W. Global Economic Impact of Dental Diseases. J Dent Res. 2015;94(10):1355-1361. doi:10.1177/0022034515602879
- Munz M, Willenborg C, Richter GM, et al. A genome-wide association study identifies nucleotide variants at SIGLEC5 and DEFA1A3 as risk loci for periodontitis [published correction appears in Hum Mol Genet. 2018 Mar 1;27(5):941-942. doi: 10.1093/hmg/ddy015]. Hum Mol Genet. 2017;26(13):2577-2588. doi:10.1093/hmg/ddx151
- for periodontitis. Human Molecular Genetics, 2017, Vol. 26, No. 13
- Steven D.P. Moore, Solon Guzman , Priscillia Quan, April Kennedy, Mathew Gene, Joel Goodman. Study of a Relative Polygenic Risk Score Assay for Common Oral Health Conditions, Dental Journal Manuscript, November 2022
- Michalowicz BS, Diehl SR, Gunsolley JC, et al. Evidence of a substantial genetic basis for risk of adult periodontitis. J Periodontol. 2000;71(11):1699-1707. doi:10.1902/jop.2000.71.11.1699
- Javaid MA, Ahmed AS, et al. (2016) Saliva as a diagnostic tool for oral and systemic diseases. J Oral Biol Craniofacial Res. 2016 Jan-Apr; 6(1): 66–75
About the Author

Beth Parkes, RDH, BSc with nearly 20 years of clinical experience, is the Manager of Clinical Affairs for Curion, a writer, a board member for Dental Hygiene Quarterly and a cast member of The RDH View. Her overarching goal is to cultivate future leaders within the dental hygiene profession, while helping their patients obtain and maintain optimal oral health.