Abstract
Monitoring peri-implant soft tissue stability has long been hindered by the subjectivity and limitations of traditional methods such as probing, photography, and radiography. Layered intraoral scanning offers a reproducible digital method to detect subtle changes in soft-tissue morphology over time. By superimposing baseline and recall scans, clinicians can identify early shifts in papilla height, mucosal margins, or buccal contours with sub-millimeter precision, strengthening diagnostic confidence and patient communication by visually demonstrating longitudinal changes. As scanning technologies advance and integrate with subsurface imaging, layered intraoral scans are poised to become a cornerstone of evidence-based soft tissue monitoring in dentistry.
Dental implants demonstrate high levels of long-term survival, with systematic reviews reporting survival rates exceeding 90% over observation periods of at least 5 years.1 Yet the presence of an osseointegrated fixture is not the sole marker of treatment success. What matters most in the years following restoration is the behavior of the peri-implant soft tissues.2,3 Minor changes, such as a millimeter of midfacial recession, slight papillary shrinkage, or subtle collapse of buccal contour, can erode esthetics, expose restorative margins, and create plaque-retentive niches that predispose to peri-implant inflammation.3,4
The tools traditionally used to track these tissues are limited. Periodontal probing, though ubiquitous, is notoriously technique-sensitive: variations in probe angulation, insertion force, and tissue tone introduce measurement error and inter-/intra-operator variability.5 Clinical photographs capture visual impressions but suffer from inconsistencies in lighting, angulation, perspective, and soft-tissue position, complicating reliable longitudinal comparison.6 Radiographs yield excellent insight into bone levels, but cannot depict the soft tissue surface morphology perceived by patients.7 These modalities deliver snapshots rather than reproducible longitudinal records. By the time changes become conspicuous, opportunities for minimally invasive intervention may already be narrowed.3
This gap motivated us to adopt layered intraoral scanning as a method to visualize and quantify soft tissue change over time.
The history of digital dentistry
Over the last four decades, digital dentistry has undergone a profound transformation, progressing from early computer-aided design and manufacturing (CAD/CAM) systems to advanced three-dimensional (3D) imaging technologies like digital radiography and cone-beam computed tomography (CBCT).8 Initially, CAD/CAM systems revolutionized restorative dentistry by enabling the rapid and precise fabrication of crowns and bridges.8 The subsequent rise in digital imaging technologies provided stronger and more detailed 3D visualizations of oral structures, enhancing diagnostic accuracy and treatment planning. Eventually, the integration of 3D printing into dental practice replaced traditional impressions and manual fabrication with highly customizable and precise restorations.8
The problem with traditional monitoring
Many clinicians have faced the uneasy task of relying on memory when comparing tissues across visits. A patient sits down for a recall examination and the clinician wonders: Was the papilla this short before? Has the margin migrated apically? Or is this recollection inaccurate?
Patient X, who received an implant at site #11 in 2020, illustrates how traditional records can reinforce this uncertainty. Over multiple recalls, the documentation seemed stable, yet each modality left unanswered questions about subtle soft-tissue change.
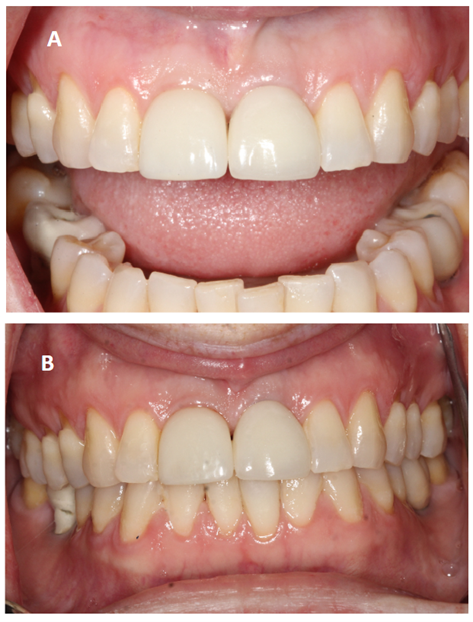
Clinical photographs, though seemingly objective, often fail to provide reliable answers. Subtle changes in lip position, soft-tissue retraction, lighting, or head angle can create the impression of tissue gain or loss when none has occurred.6 In this patient, the clinical images taken in 2021 and 2025 appear similar (Figure 1). However, variations in framing and light reflection make it nearly impossible to determine whether true soft-tissue alteration occurred over the four-year interval. What should be a precise record becomes a subjective impression.6
Fig. 1
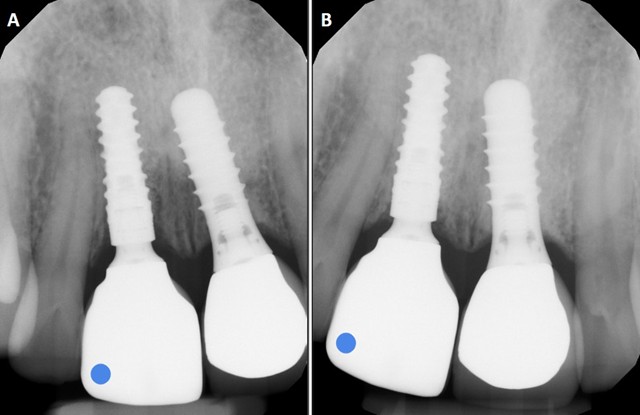
Radiographs, by contrast, give clear and reproducible insight into implant osseointegration and crestal bone stability.7 In the same patient, periapical films from 2021 and 2025 (Figure 2) confirm stable bone levels at #11. However, while radiographs reassure the clinician that the fixture is integrated, they are blind to surface tissue contours that determine long-term esthetics.7 Patients do not typically notice crestal bone levels; they perceive small shifts in visible tissues such as the mucosal margin or contour. These esthetic changes often begin subtly and may go unnoticed at first, only becoming evident after years of gradual progression.3 Conventional records, whether photographic or radiographic, provide little help in identifying these early stages.6,7
Fig. 2
Meanwhile periodontal probing depth, though entrenched in practice, is highly technique-sensitive: probe angulation, insertion force, and tissue tone all introduce variability, and studies have shown that reproducibility often fluctuates by half a millimeter or more, even within the same operator.5 These margins of error commonly range from 0.3 to 0.8 mm and are greater than the magnitude of many clinically significant tissue shifts.5
This ambiguity not only frustrates diagnosis but also complicates communication. It is difficult to justify intervention when the only evidence is a clinician’s impression that “something looks different.” Patients understandably hesitate to accept additional treatment on such a subjective basis. Without an objective, reproducible way to demonstrate change, clinicians risk losing both diagnostic confidence and patient trust.
The concept of layering intraoral scans
Intraoral scanners (IOS) were first introduced to replace conventional impressions in restorative dentistry.9 Over the last decade, however, their clinical role has broadened considerably, and one of the most impactful applications extends beyond prosthetic fabrication: longitudinal soft-tissue monitoring. By capturing a baseline scan before final restoration delivery and superimposing it with recall scans at later visits, even subtle changes in peri-implant morphology can be detected. The process relies on surface registration, using stable anatomical landmarks such as adjacent teeth or palatal rugae to ensure that comparisons reflect true biological change rather than alignment error.10
Digital workflows in implant dentistry now increasingly incorporate IOS data not only for planning but also for follow-up. Several reports have highlighted that intraoral scanning can provide profilometric measurements of mucosal margin stability and volumetric changes in peri-implant soft tissue, with reproducibility sufficient to track shifts in the range of tenths of a millimeter.10,11 A recent narrative review highlighted intraoral scanning among the main digital modalities for diagnosing, planning, and monitoring peri-implant soft tissue conditions.8
At Chrysalis Dental Centres, we employ the DEXIS IS 3800WⓇ (DEXIS, Hatfield, PA, USA) in combination with exocadⓇ software (exocad GmbH, Darmstadt, Germany) to perform these overlays (Figure 3). This scanner offers high resolution and wireless flexibility, but the principle of the method is what matters: any modern IOS capable of accurate surface capture, paired with alignment software, can reproduce this workflow with similar effectiveness.12
Fig. 3
How we built a reliable protocol
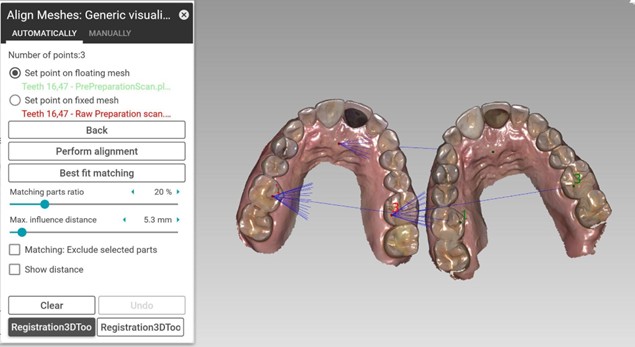
Turning the concept of scan layering into a practical clinical tool required a protocol that could be reproduced consistently across visits. We begin with a baseline scan taken before the final restoration is delivered. We carefully manage moisture control and soft-tissue retraction so that the contours are recorded without distortion. At each visit, we follow a standardized scanning path, moving sequentially from buccal to occlusal to palatal; this minimizes stitching errors and ensures that subsequent datasets can be compared with maximum accuracy.10
At recall appointments, we capture a new scan under the same controlled conditions and import it into exocadⓇ software. We trim both the baseline and recall datasets to remove extraneous regions such as the floor of the mouth or redundant palatal tissue. Alignment is then performed using stable, immobile landmarks such as enamel surfaces or palatal rugae, while excluding gingival tissues, which are too mobile to serve as reliable reference points (Figure 4).10
Fig. 4
Once we register the scans, the software generates cross-sectional slices and colour-coded deviation maps, allowing precise visualization of both the location and magnitude of tissue change. We carefully check residual error on reference structures; if deviations exceed ≈0.1 mm, we consider the registration unreliable and repeat the scan.11 We archive only alignments within tolerance as part of the patient’s longitudinal record.
In practice, this workflow adds only a few minutes to a recall visit. Hygienists or assistants can manage scanning and preliminary alignment, while clinicians interpret results. The modest time investment yields a significant gain in reproducibility and objectivity, transforming subjective observation into quantifiable evidence.
What the overlays reveal
Layered intraoral scans make it possible to identify soft-tissue changes at a stage when they are still subtle and potentially reversible. Papillary shrinkage, which may compromise esthetics and function if left unchecked, can often be detected before it becomes obvious to either the clinician or the patient.4,14 Midfacial drift of the mucosal margin can be measured to provide early warning of recession that would be imperceptible with conventional methods.13 In a controlled clinical trial, three-dimensional intraoral scanning was used to quantify peri-implant soft-tissue dimensional changes over time, demonstrating that even small shifts could be reliably detected across different implant-abutment systems.13 Moreover, clinicians can track buccal contour flattening longitudinally (notably in thin biotypes or grafted sites), observe creeping attachment, and verify post-augmentation volumetric stability using objective measurements.4,14
The alignment software enhances visualization by producing color-coded deviation maps. By setting thresholds that render deviations under ±0.1 mm as neutral (consistent with scanner accuracy literature), clinicians can filter out background noise and focus on biologically meaningful change.10
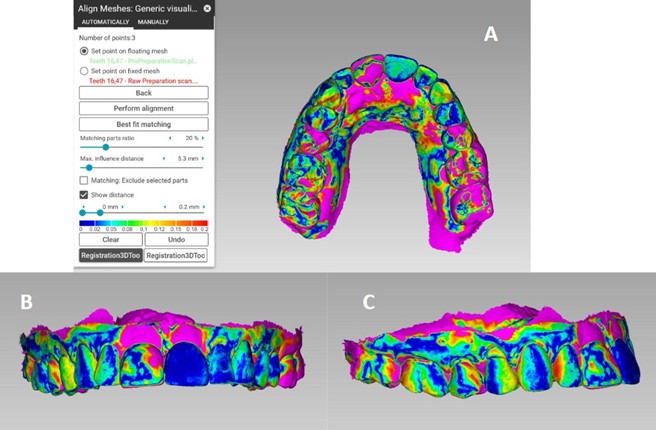
In Patient X, the overlays revealed distinct patterns of tissue remodeling. From the occlusal perspective (Figure 5A), diffuse buccolingual reduction is visible across the maxillary arch, with pink regions corresponding to ≈0.2 mm loss. The frontal view (Figure 5B) highlights localized change in papillary-facial region adjacent to implant #11, where papillary height and facial contour show early resorption. The lateral view (Figure 5C) confirms these findings, illustrating facial flattening of the peri-implant mucosa when compared to baseline.
Fig. 5
Beyond surface deviation maps, cross-sectional slicing of superimposed scans provides a quantitative way to measure tissue remodeling in millimeters. By cutting through the midfacial aspect of an implant restoration, the software allows clinicians to pinpoint whether changes are occurring vertically at the mucosal zenith, horizontally across the facial contour, or both. This approach translates color-coded deviation into values, reinforcing diagnostic confidence and providing clear evidence for patients.
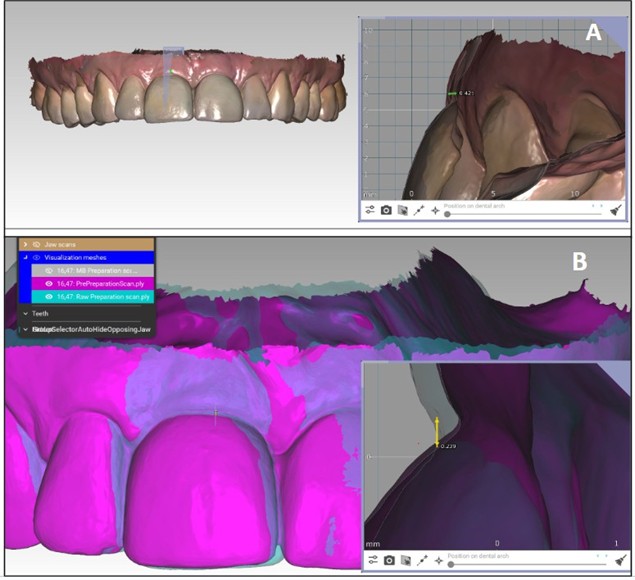
In Patient X, two cross-sections through implant #11 demonstrate complementary patterns of change. Figure 6A shows a vertical slice at the midfacial zenith using native (non-colour) surface textures; the superimposed 2025 recall surface sits ≈0.42 mm apical to the 2021 baseline, indicating vertical mucosal recession. Figure 6B displays a facial slice with colour overlays (pink = 2025; purple = 2021); the 2025 outline lies lingual to the 2021 baseline by ≈0.24 mm, consistent with a buccolingual reduction. Together, these cross-sections confirm both vertical and horizontal soft-tissue remodeling that would have been imperceptible without digital superimposition.
Fig. 6
When patients see what we see
One of the most transformative aspects of scan layering is its ability to turn abstract clinical findings into visual evidence that patients can easily grasp. Describing “tissue loss” or presenting numbers in a chart often fails to resonate, but showing patients their own scans layered over time makes change tangible. Whether displayed as a color map or in a simple overlay, patients can immediately recognize where tissues have shifted and by how much. This clarity helps them appreciate why maintenance and early intervention matter.15
Evidence supports this impact: a systematic review of digital patient education tools in dentistry found that visual aids such as intraoral scans significantly improve comprehension and increase willingness to accept recommended care.15 Patient-reported outcomes with digital workflows are also favorable; for example, intraoral scanning reduces working time and improves overall patient experience compared with conventional impressions.16 Supporting this, a prospective randomized controlled trial in adult prosthodontic patients (n = 75) showed that a software-based visual patient decision aid produced significantly more favorable patient ratings across all decision-quality items compared with usual verbal consultation (all p ≤ 0.032), the highest overall satisfaction (p = 0.015), and the shortest consultation time.17
In our own setting, scan layering provides a similar effect for implants: patients who might hesitate at a clinician’s verbal description become far more receptive when they see their own tissue contour flattening or margin migration over time. The conversation shifts from abstract explanation to shared observation, grounding treatment decisions in images that feel both personal and undeniable.
Beyond implants: Expanding applications
Although our initial motivation was peri-implant monitoring, the value of scan layering reaches far beyond implant dentistry. In periodontology, intraoral overlays provide an objective way to document gingival recession and to verify creeping attachment following grafting procedures, offering visual confirmation of stability or relapse.14 In orthodontics, subtle arch form changes, minor rotations, or retainer displacement can be tracked over time, giving clinicians a precise tool for monitoring post-treatment stability and relapse.12,18 Prosthodontists, meanwhile, can apply the same approach to assess occlusal wear, monitor marginal adaptation of crowns and bridges, or evaluate shifts in vertical dimension across years of function.10,19
Oral surgeons have also begun to explore the use of overlays for volumetric analysis of soft- and hard-tissue grafts, providing quantitative evidence of augmentation success or resorption that would otherwise rely on subjective judgment.10,13 Moreover, while IOS layering cannot currently diagnose subsurface bone loss, it makes time-based soft-tissue outcomes visible. Because
soft-tissue change reflects both biologic remodeling and prosthetic/surgical variables, longitudinal overlays complement CBCT and clinical probing by translating complex healing into measurable, patient-specific visuals.
Lessons learned along the way
Adopting this protocol was not without challenges. Early attempts at automatic alignment often misregistered when movable gingiva was included, producing false deviations. Saliva contamination introduced artifacts along the gingival margin and patient movement occasionally fragmented scans, leading to noisy datasets.
To address these issues systematically, the protocol incorporated routine dry-field isolation, greater emphasis on retraction for stable tissue capture, and precise definition of alignment regions to exclude mobile tissues. If noise persisted, we repeated only the relevant segment rather than the entire arch. These refinements transformed an initially cumbersome process into a streamlined workflow.
Looking ahead: Subsurface imaging
While current IOS technology is limited to capturing surface morphology, the future of monitoring may lie beneath the tissues. Optical coherence tomography (OCT), already well established in medicine and comprehensively reviewed for dental applications, provides
non-ionizing, micrometer-scale cross-sectional imaging of both soft and hard tissues.20-23 Unlike radiographs, which infer bone and are blind to mucosa, OCT has shown potential to detect early mineral changes in enamel and bone, as well as to directly visualize sulcular structures (gingival epithelium, connective tissue, and junctional layers) in vivo.20-23
The implications are profound. Integrated with IOS, OCT could generate a single dataset that captures both external contours and internal tissue architecture longitudinally. In such a system, the clinician would not be limited to surface overlays or two-dimensional radiographs but could instead monitor esthetic contours, mucosal thickness, and crestal bone remodeling simultaneously, non-invasively, and with greater precision. At that point, the need for routine radiographs in certain monitoring contexts may diminish, as a single scan would provide a comprehensive, layered record of oral health.
Conclusion
Dentistry has long relied on probing, photography, and radiography to monitor soft-tissue change. These remain valuable, but their subjectivity and limitations restrict early detection. Layered intraoral scans represent a paradigm shift: transforming serial impressions into analyzable digital records that detect subtle change early, communicate evidence clearly, and guide conservative intervention.
In our clinic, adoption of scan layering has moved follow-up care from memory-based assessment to evidence-based monitoring. What once seemed static now reveals dynamic change and what once felt reactive now becomes proactive. As imaging and software technologies (e.g.,
OCT integration) evolve, IOS has the potential to become the backbone of comprehensive soft-tissue surveillance in dentistry.
References
- Pjetursson BE, Thoma D, Jung R, Zwahlen M, Zembic A. A systematic review of the survival and complication rates of implant-supported fixed dental prostheses after ≥5 years. Clin Oral Implants Res. 2012;23(Suppl 6):22-38.
- Berglundh T, Armitage G, Araujo MG, et al. Peri-implant diseases and conditions: 2017 World Workshop consensus report. J Clin Periodontol. 2018;45(Suppl 20):S286-S291.
- Thoma DS, Naenni N, Figuero E, et al. Effects of soft-tissue augmentation on peri-implant stability: systematic review and meta-analysis. Clin Oral Implants Res. 2018;29(Suppl 15):32-49.
- Cosyn J, Eghbali A, De Bruyn H, et al. Importance of gingival biotype in esthetic dentistry: review. J Periodontol. 2011;82(4):519-528.
- Listgarten MA. Periodontal probing: what does it mean? J Clin Periodontol. 1980;7(3):165-176.
- Ahmad I. Digital dental photography: an overview. Br Dent J. 2009;207(4):193-199.
- Brägger U. Radiographic parameters: biological significance and clinical use. Periodontol 2000. 2005;39:73-90.
- Gawali N, Shah PP, Gowdar IM, Bhavsar KA, Giri D, Laddha R. The evolution of digital dentistry: a comprehensive review. J Pharm Bioallied Sci. 2024;16:123-131.
- Logozzo S, Zanetti EM, Franceschini G, Kilpelä A, Mäkynen A. 3D intraoral scanners for restorative dentistry. Opt Lasers Eng. 2014;54:203-221.
- Haddadi Y, Bahrami G, Isidor F, Wenzel A. Factors affecting accuracy of intraoral digital impressions: systematic review. Clin Oral Investig. 2022;26(1):111-125.
- Mangano FG, Veronesi G. Digital versus conventional impressions in dentistry: A systematic review and meta-analysis. J Esthet Restor Dent. 2018;30(2):85-91. doi:10.1111/jerd.12393
- Grünheid T, McCarthy SD, Larson BE. Clinical use of a direct chairside oral scanner: accuracy, time, and patient acceptance. Am J Orthod Dentofacial Orthop. 2014;146(5):673-682.
- Derakhshani AJ, et al. 3D analysis of soft-tissue dimensional changes after dental implant placement: clinical trial. Int J Implant Dent. 2024;10:66.
- Chen ST, Buser D. Esthetic outcomes following immediate and early implant placement in the anterior maxilla: systematic review. Int J Oral Maxillofac Implants. 2014;29(Suppl):186-215.
- Schwendicke F, Krois J, Gomez J. Patient-centered dentistry: digital tools for improved patient communication and education. J Dent Res. 2020;99(6):685-692. doi:10.1177/0022034520911110
- Siqueira R, et al. Intraoral scanning reduces procedure time and improves patient-reported outcomes vs conventional impressions: systematic review. Clin Oral Investig. 2021;25:6517-6542.
- Brandt S, Lauer H-C, Güth J-F, Bühling S, Sayahpour B, Romanos G, Winter A. Impact of two different patient decision aids in prosthodontic consultations: a prospective randomized controlled study. Clin Oral Investig. 2023;27(12):7271-7280. doi:10.1007/s00784-023-05375-7.
- Kravitz ND, Groth C, Jones PE, Graham JW, Redmond WR. Intraoral digital scanners for orthodontics: efficiency and effectiveness. Am J Orthod Dentofacial Orthop. 2014;145(4):509-519.
- Vohra F, Alrahlah A, Zafar MS, et al. Monitoring occlusal wear using intraoral digital scanning: systematic review. J Prosthodont. 2020;29(7):593-601.
- Al-Adili A, Mirzaali MJ, Eggermont F, Forouzanfar T, Kleverlaan CJ, Wismeijer D. Clinical applications of optical coherence tomography in dentistry: review. Dentomaxillofac Radiol. 2021;50(8):20210053.
- Popescu DP, Flueraru C, Mao Y, Chang S, Sowa MG. Optical coherence tomography: fundamentals and biomedical applications. J Biomed Opt. 2011;16(12):121907.
- Colston BW Jr, Everett MJ, Da Silva LB, Otis LL, Stroeve P, Nathel H. Imaging oral hard- and soft-tissue with OCT. Appl Opt. 1998;37(19):3582-3585.
- Sharma P, Yadav R, Wadhwa J, et al. Optical coherence tomography in dentistry: current applications and future perspectives. Photodiagnosis Photodyn Ther. 2020;31:101832. doi:10.1016/j.pdpdt.2020.101832.
About the authors

Nives Braunstein earned a BSc (Hons) in Life Sciences from Queen’s University in June 2025 and works on the Research & Development team at Chrysalis Dental Centres in Toronto. For this project, she coordinated the full workflow, from intraoral scan acquisition and layered analysis to data interpretation and manuscript preparation, in collaboration with the surgical, prosthodontic, and dental laboratory teams. She will be pursuing dentistry, with interests in surgical excellence, patient-centred evidence-based workflows, longitudinal follow-up that guides care, and applied facial anatomy.

Maya is currently pursuing her Bachelor of Health Sciences degree at Queen’s University. She is dedicated to advancing treatment outcomes in clinical environments that prioritize evidence-based practices and patient-centered care. As a summer research assistant at Chrysalis Dental Centres, Maya directly utilized intraoral scanners for data acquisition and assisted with manuscript preparation, revisions, and finalization. She is passionate about interdisciplinary collaboration within healthcare, striving to improve care delivery through innovative therapeutic approaches.

Dr. Freund received his medical degree from McMaster University and received his dental and specialist training from the University of Toronto. He practices as an Oral and Maxillofacial surgeon at the Chrysalis Dental Centres in Toronto with special interests in immediately loaded implant rehabilitation, neuromuscular dysfunction of the head and neck and minimally invasive facial rejuvenation.

Dr. Steven Bongard obtained his DDS degree from the University of Toronto Faculty of Dentistry in 1986. He is the founder and clinical head of Chrysalis Dental Centre in Toronto, a dental implant only facility focused on “same day” dental implant solutions. Dr. Bongard has extensive experience in implant placement, bone grafting procedures, as well as the prosthetic component of implant restoration.

Dr. Alex Bongard has extensive surgical and restorative experience in both full arch and single tooth implant dentistry practicing at Chrysalis Dental Centre since May 2022. He is passionate about changing the world of implantology by embracing simplicity and immediacy with a patient centered approach to overcome barriers to treatment.

Dr. John P. Zarb earned his DDS at the University of Detroit Mercy in 1995, followed by graduate prosthodontic training at the University of Toronto, earning his MSc in 2002. Dr. Zarb is a staff prosthodontist at Mt Sinai Hospital in Toronto, and a Clinical Assistant Professor at the University of Rochester, Eastman Institute for Oral Health.