In the complex ecosystem of oral cavity, a microscopic battle unfolds daily, shaping the delicate balance between health and disease. At the forefront of this is the formation of dental plaque and bacterial biofilm, which plays a fundamental role in the development of one of the most prevalent oral conditions known as periodontitis.1 This multifaceted and dynamic process involves a myriad of microbial interactions and host responses, creating an intricate interplay that, when disrupted, can result in the destruction of periodontal tissues. The oral cavity harbors a diverse and dynamic microbial community, collectively referred to as the oral microbiome.2 This ecosystem plays a pivotal role in maintaining oral health and the delicate equilibrium between commensal and pathogenic microorganisms is crucial for preventing the onset of oral diseases, with the balance tipping towards disease when this equilibrium is disrupted.3
Dental plaque, a biofilm formed by the adhesion of microorganisms to the tooth surface, is the precursor to periodontal disease. The process of plaque formation begins with the reversible attachment of initial colonizers to the acquired pellicle, a thin layer of salivary proteins that coats the tooth surfaces.4,5 Initial colonizers, primarily streptococci, adhere to the acquired pellicle through interactions with specific adhesins on their cell surfaces.5 This reversible attachment is followed by coaggregation between different microbial species, creating a scaffold for the subsequent colonization of additional microorganisms.4,5 As microbial communities mature, they undergo a process of succession, with early colonizers creating a favorable environment for the recruitment of late colonizers.5 This process involves the development of complex microbial interactions and the secretion of extracellular polymeric substances (EPS) that form a protective matrix around the biofilm.4
Once established, dental plaque transforms into a formidable structure known as bacterial biofilm.5 This three-dimensional, highly-organized community of microorganisms presents a challenge to the host’s immune system and provides a sanctuary for the proliferation of pathogenic bacteria.4 Within the biofilm, bacteria communicate through a process known as quorum sensing, enabling them to coordinate their activities and respond collectively to environmental changes.5 This synchronized communication enhances the virulence of pathogenic bacteria, allowing them to modulate gene expression, resist host defenses, and adapt to the dynamic conditions within the oral cavity.5 The intricate interplay between dental plaque, bacterial biofilm, and host factors sets the stage for the development of periodontal disease.4
The initial stage of periodontal disease is gingivitis, marked by inflammation of the gingival tissues in response to the presence of dental plaque.6 Although gingivitis is reversible with effective oral hygiene measures, the progression to periodontitis occurs when the inflammatory process extends beyond the gingiva to involve the deeper periodontal tissues.7 Periodontitis is characterized by inflammation of the supporting structures of the teeth, leading to tissue destruction, tooth mobility, and, ultimately, tooth loss.
The host’s immune response, designed to protect against microbial invaders, paradoxically contributes to the pathogenesis of periodontal disease.7 The release of inflammatory mediators, such as cytokines and prostaglandins, amplifies the local inflammatory response, leading to collateral damage to the surrounding tissues.6,7 Moreover, the chronic nature of periodontitis perpetuates an ongoing cycle of inflammation and tissue destruction.
The Challenge of Dental Biofilm and Plaque Disruption and Removal
The multifaceted nature of periodontal disease demands a comprehensive therapeutic approach that targets both microbial and host factors. Current strategies focus on disrupting biofilm formation, modulating the host’s immune response, and promoting the restoration of a balanced oral microbiome.8 The battle against plaque and bacterial biofilm in the oral cavity is an ongoing struggle for individuals and oral health professionals alike. While the importance of effective plaque removal is widely acknowledged, the inherent challenges in disrupting biofilm formation and plaque removal pose significant obstacles.9
Adherence and tenacity of biofilm: A primary challenge in plaque and biofilm removal lies in the remarkable tenacity of these microbial communities on tooth surfaces.10 Bacterial adhesion is facilitated by specific adhesins that interact with components of the acquired pellicle,9 making the initial attachment of microorganisms reversible but subsequently leading to irreversible adhesion and biofilm maturation.11 The complex extracellular polymeric substances further cement the biofilm structure, creating a protective fortress that shields bacteria from mechanical and chemical interventions.11
Microbial coaggregation and succession: The process of microbial coaggregation within the biofilm creates an environment conducive to the growth of diverse microorganisms.9 The formation of specific microenvironments and niches supports the establishment of both commensal and pathogenic bacteria, complicating the task of selective removal.10,11 The succession of microorganisms within the biofilm adds another layer of complexity, making it challenging to target specific species responsible for disease progression.
EPS as a protective shield: Extracellular polymeric substances, comprising polysaccharides, proteins, and nucleic acids, contribute to the robustness of the biofilm.10,11 These substances create a physical barrier that hinders the penetration of antimicrobial agents and mechanical forces.9 The resistance of biofilm to conventional toothbrushing and flossing emphasizes the need for innovative strategies that can effectively penetrate and disrupt the protective matrix.
The challenge of anatomy and access to plaque removal: The diverse and intricate anatomy of the oral cavity presents challenges in reaching and effectively cleaning all tooth surfaces. The complexity of tooth morphology, including interproximal spaces, occlusal surfaces, and areas adjacent to restorations, makes it difficult to ensure comprehensive plaque removal.11 Inadequate access to certain areas may result in persistent biofilm accumulation and contribute to the development of periodontal disease.9,10 The tight spaces between teeth pose a significant challenge for effective plaque removal and traditional toothbrushing may struggle to reach these interproximal areas, leading to the accumulation of plaque and biofilm.9 While dental floss and interdental brushes can aid in cleaning between teeth, patient compliance and technique variability remain concerns. Occlusal surfaces, fissures, and areas surrounding dental restorations provide additional hiding places for biofilm. The irregularities and crevices in these areas make complete plaque removal difficult, increasing the risk of bacterial colonization.11 In the case of restorations, the presence of marginal gaps and rough surfaces can further complicate biofilm removal and contribute to periodontal disease initiation and progression leading to irreversible loss of supporting tissues of teeth.
HYBENX® Technology
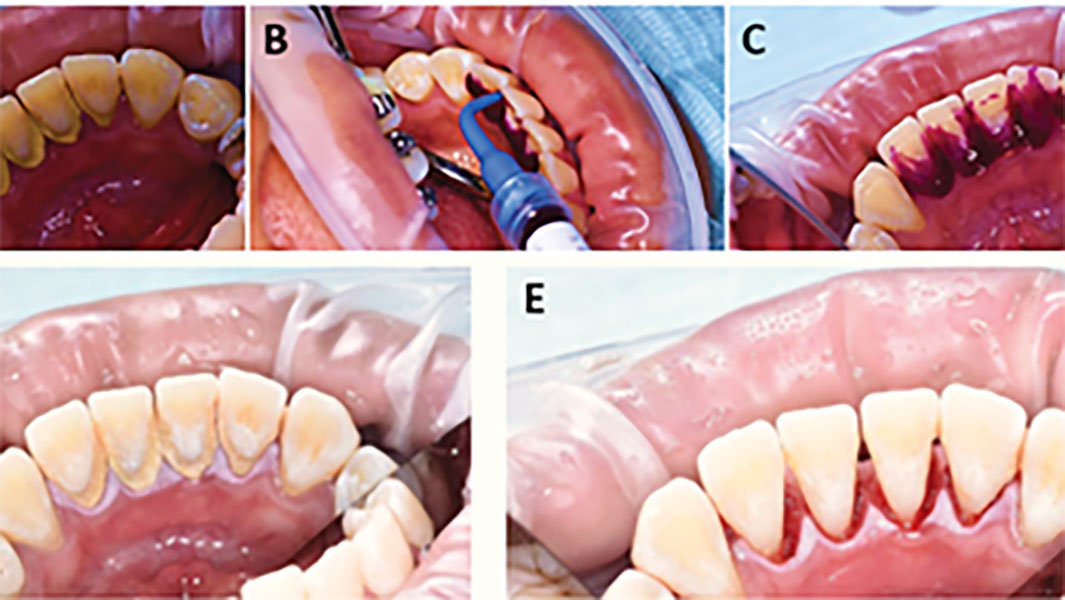
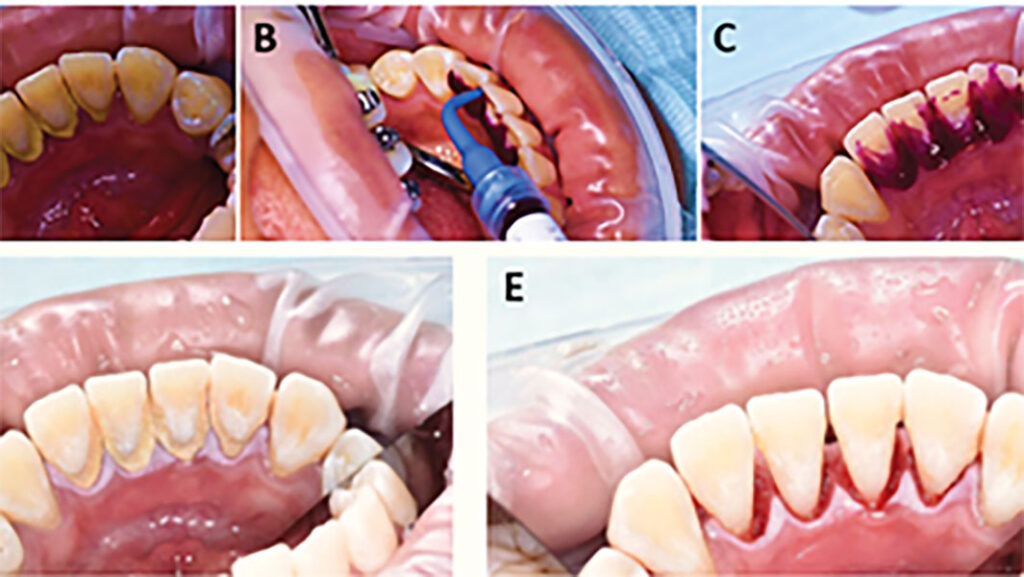
HYBENX® products operate through a unique chemical process, functioning as antimicrobial tissue cleansers or tissue conditioning focal rinse agents. These products actively detach pathological matter from surfaces during application by employing a superficial denaturing action, facilitated by a proprietary chemical process. This denaturing activity aids the mechanical rinsing action of the product, effectively removing targeted materials. Application of HYBENX® products is designed to complement standard dental treatment methods. Formulated as dense, moderately viscous liquids, they are typically applied using a small irrigation syringe with a dental applicator tip (Fig. 1). The product is left in place for 10-30 seconds, depending on the situation, and then rinsed away with water and evacuated (Figs. 2, 3).
Fig. 1
Fig. 2
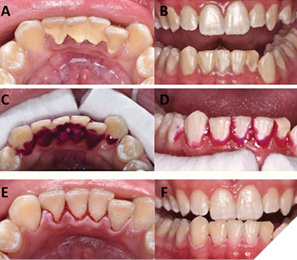
Fig. 3
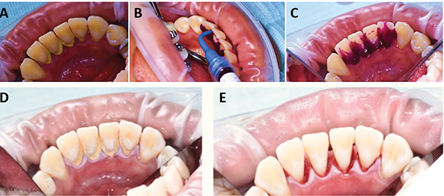
The practitioners can apply HYBENX® products whenever deemed beneficial, with notable advantages in severe dental conditions, such as heavy plaque and calculus buildup. In addition, pre-scaling application can facilitate easier removal during mechanical scaling, while post-scaling application ensures removal of any residual plaque (Figs. 2, 3). A randomized split-mouth clinical trial compared the effect of Scaling and Root Planing (SRP) alone versus SRP plus a desiccant (HYBENX®) using clinical, microbiological, and inflammatory mediator analyses in patients with chronic Periodontitis.12 Both treatments improved the clinical, microbiological, and inflammatory mediator outcomes. However, the SRP + desiccant treatment led to a statistically significant improvement at 12 months of the Probing Depths (PD) and Bleeding On Probing (BOP) compared to the SRP alone.12
The molecular mechanism of action in HYBENX® products centers around their role as powerful liquid contact desiccants. This property arises from their concentrated blends of sulfonic/sulfuric acids, which exhibit a strong affinity for water. Sulfate groups, present in the products, possess a polar internal structure with outer oxygen atoms carrying a strong negative charge. When these sulfate groups interact with water, they form reversible hydrogen bonds, with the sulfate’s negative surface attracting the positive surfaces of water molecules. The strength of hydrogen bonds is influenced by the polarity of the groups involved, leading sulfate groups to compete with other polar molecular groups for water molecules. HYBENX® has a potent affinity for water, allowing it to generate new water molecules through a dehydration elimination reaction. The desiccating effect of concentrated sulfate solutions stems from their successful competition with tissue and material components for water molecules.
Water molecules form contiguous layers, or “shells,” around the sulfate surface. Additional water molecules create layers akin to an onion. As each shell forms, the sulfate’s affinity for water decreases due to dilution. The desiccation intensity of HYBENX® products is determined during formulation by creating the necessary number of water shells around sulfate groups through water dilution. This reduces the net negative polarity to a desired level, preventing destructive reactions or coagulative necrosis on contact with healthy intact tissue. HYBENX®’s effectiveness relies on its ability to compete for hydrogen-bonded water on specific oral tissue surfaces. Successful competition leads to the absorption of water, causing the structure of molecules to become unstable. This instability prompts chemical groups on the desiccated molecule to react, resulting in the collapse into a new, stable arrangement. However, this arrangement is nonfunctional and abnormal, leading to molecular denaturation within tissues.
Molecular denaturation, induced by HYBENX®, results in the gross coagulation of tissues. HYBENX®’s action is limited to desiccating certain oral tissue surfaces to the point of denaturation, without causing destructive elimination reactions or acidification. The components of HYBENX®, being highly concentrated, prevent adequate proton transfer into tissue until the hydrogen bonding capacity of the sulfate is exhausted and unbounded water molecules become readily available. In summary, HYBENX® operates by desiccating oral tissues, inducing molecular denaturation, and promoting tissue coagulation without causing destructive reactions or acidification until specific concentration thresholds are reached.
The interaction of HYBENX® products with microbial biofilms represents a unique application of this technology. Microbial biofilms are common in wet environments, such as teeth, where microorganisms attach to a surface and replicate. As these microbes recruit others, they collectively secrete a polysaccharide material forming an extracellular matrix, creating a biofilm. Research indicates that a significant portion of microbes, including those within the human body, reside within biofilms. HYBENX® products exhibit a distinctive ability to eradicate dental plaque, a form of microbial biofilm, through a potent desiccating action.
Dental plaque, like other microbial biofilms, possesses an aqueous polysaccharide gel structure with adhesive properties, making it resistant to mechanical shear forces and conventional cleansers. HYBENX® products exploit the absence of a protective surface structure in the plaque matrix, allowing for rapid denaturation. This desiccation causes coagulation and shrinkage of the matrix and enclosed microbes, leading to detachment from the tooth surface. This unique action positions HYBENX® as a highly effective dental therapeutic agent for plaque removal and microbial eradication, compensating for the limitations of mechanical anti-plaque techniques, antibiotics, and antiseptics against biofilm microbes.
Recognizing the utility of contact desiccation as a mechanism to eradicate dental plaque and microbial biofilms is a key element of HYBENX®’s intellectual property, as outlined in their patents. Standard plaque removal techniques involve mechanical shear forces, which are often ineffective due to biofilm resistance. HYBENX® products are the first to exploit a structural weakness in the biofilm matrix, enabling easy and rapid eradication of both the matrix and plaque bacteria. For dental practitioners, the adoption of HYBENX® products offers significant clinical advantages. The application does not require expensive equipment or specialized training, making it accessible for routine use. Three main categories of benefits for practitioners include improving the cost efficiency of procedures, expanding patient care options without additional resources, and addressing the link between oral inflammation and systemic disease.
In conclusion, HYBENX® products provide a novel approach to eradicating microbial biofilms, particularly dental plaque, through their potent desiccating action. The flexibility in application methods and the three-fold benefits for practitioners make HYBENX® a promising addition to routine dental care, offering improved efficiency and expanded treatment options. For clinicians, the technology’s ability to ensure a smoother and faster calculus removal process leads to shorter and more convenient appointments. On the patient side, the elimination of dysbiotic biofilm represents a significant breakthrough. Beyond the immediate improvement in oral health, patients benefit from a reduced risk of complications associated with biofilm formation. Overall, HYBENX® technology has the potential to transform the treatment experience, making it a valuable asset in modern dental care. 
Oral Health welcomes this original article.
References
- Marsh, P.D. and D.J. Bradshaw, Dental plaque as a biofilm. Journal of industrial microbiology and biotechnology, 1995. 15(3): p. 169-175.
- Yamashita, Y. and T. Takeshita, The oral microbiome and human health. Journal of oral science, 2017. 59(2): p. 201-206.
- Jiao, Y., M. Hasegawa, and N. Inohara, The role of oral pathobionts in dysbiosis during periodontitis development. Journal of dental research, 2014. 93(6): p. 539-546.
- Christersson, L.A., J.J. Zambon, and R.J. Genco, Dental bacterial plaques: nature and role in periodontal disease. Journal of clinical periodontology, 1991. 18(6): p. 441-446.
- Colombo, A.P.V., et al., Periodontal-disease-associated biofilm: A reservoir for pathogens of medical importance. Microbial pathogenesis, 2016. 94: p. 27-34.
- Kinane, D.F., P.G. Stathopoulou, and P.N. Papapanou, Periodontal diseases. Nature reviews Disease primers, 2017. 3(1): p. 1-14.
- Cochran, D.L., Inflammation and bone loss in periodontal disease. Journal of periodontology, 2008. 79: p. 1569-1576.
- Kwon, T., I.B. Lamster, and L. Levin, Current concepts in the management of periodontitis. International dental journal, 2021. 71(6): p. 462-476.
- Ryan, M.E., Nonsurgical approaches for the treatment of periodontal diseases. Dental Clinics, 2005. 49(3): p. 611-636.
- Merchant, A.T. and W. Pitiphat, Researching periodontitis: challenges and opportunities. Journal of clinical periodontology, 2007. 34(12): p. 1007-1015.
- Shaddox, L.M. and C.B. Walker, Treating chronic periodontitis: current status, challenges, and future directions. Clinical, cosmetic and investigational dentistry, 2010: p. 79-91.
- Isola, G., et al., The effects of a desiccant agent in the treatment of chronic periodontitis: A randomized, controlled clinical trial. Clinical Oral Investigations, 2018. 22: p. 791-800.
About the Authors

Dr. Zeeshan Sheikh has trained at Baqai University, Queen Mary University (London), McGill University, University of Toronto, Mt. Sinai Hospital and Dalhousie University. Author of 70+ original research articles, reviews, he is Clinical Scientist (Periodontics) and Assistant Professor, Dalhousie University and practices at Parklane Dental Specialists.

Martha Szczepulski, DiplDentHygiene (New Caledonia), BDS in Dental Hygiene (UBC), and the British Columbia Provincial Instructors Diploma. She has extensive Dental Hygiene and Dental Assisting teaching experience, and is currently Education Leader, Oral Science Professional. She maintains a mobile hygiene practice in long-term care settings.

Dr. Umer Daood, BDS (Baqai University) MSc Oral Sciences and Biomaterials (National University of Singapore), MSc Dental Materials (Queen Mary University, London), PhD (Prince Philip Dental Hospital Hong Kong University), and a Cert. Periodontology (Queen Elizabeth Hospital, Woolich, London). With >80 high impact international publications, he is a Member MFDS, RCS Glasgow.