Abstract
This manuscript explores the burgeoning collaboration between dentistry and nuclear engineering, showcasing their shared principles and synergistic potential through examples such as research programs focusing on radiation-based diagnostic tools, joint clinical initiatives optimizing radiation therapy for oral cancers, and innovative applications of nuclear imaging modalities such as PET/CT (Positron Emission Tomography/Computed Tomography) in oral health assessments. By examining advancements in medical imaging, radiation therapy, materials science, radiation safety, and emerging technologies, this interdisciplinary dialogue uncovers innovative approaches poised to transform oral healthcare. Medical imaging techniques, such as PET and SPET (Single-Photon Emission Computed Tomography), enhance diagnostic accuracy, while advancements in radiation therapy improve targeted cancer treatments, minimizing harm to surrounding tissues. A comprehensive analysis of these areas highlights how interdisciplinary collaboration fosters innovation and drives progress in oral healthcare. By bridging the gap between these fields, novel diagnostic and therapeutic approaches can be developed to improve patient outcomes and elevate the overall quality of care. This cross-disciplinary collaboration sets the stage for future innovations with the potential to advance oral health on a global scale.
Dentistry and nuclear engineering, traditionally distinct disciplines, are increasingly converging to address common challenges and opportunities in healthcare. Nuclear medicine has emerged as a critical imaging modality, particularly in the diagnosis and treatment of diseases affecting the oral and maxillofacial regions. While conventional radiographs, Cone Beam Computed Tomography (CBCT), and Computed Tomography (CT) are commonly employed in dentistry, nuclear medicine’s ability to visualize metabolic and physiological processes using radionuclides offers distinct advantages.1 This capability is particularly beneficial in diagnosing and treating diseases affecting the oral and maxillofacial regions, offering precision beyond that of traditional imaging techniques.2
Historically, the evolution of nuclear engineering has provided safer and more precise diagnostic and therapeutic technologies across various medical fields.3 This progress is now beginning to extend meaningfully into dentistry, enabling professionals to address key challenges such as minimizing patient radiation exposure, improving imaging clarity, and advancing the precision of therapeutic interventions.4
This article investigates the multifaceted relationship between these fields, highlighting key areas of collaboration and innovation. By leveraging insights from dentistry and nuclear engineering, researchers and practitioners can forge new pathways for advancing oral healthcare, addressing pressing societal needs and enhancing the effectiveness of existing treatments. This exploration emphasizes the importance of interdisciplinary approaches in tackling complex health issues and improving the integration of technology into clinical practice.
Medical imaging
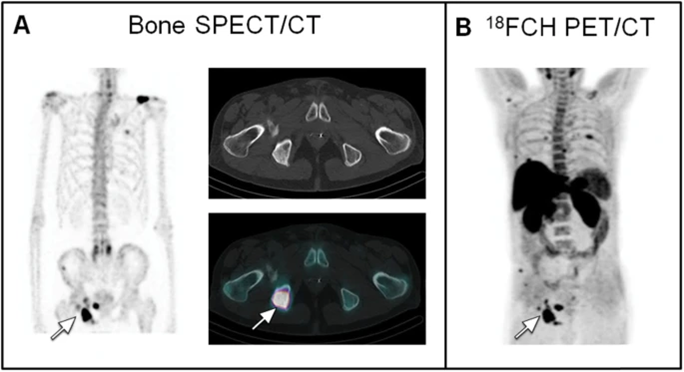
Nuclear imaging modalities, such as positron emission tomography (PET) and single-photon emission computed tomography (SPECT), offer valuable insights into the physiological processes underlying oral diseases. PET, for instance, is particularly effective in identifying metabolic changes at a cellular level, which can be crucial in detecting malignancies and inflammatory processes in their early stages.5 A 2017 prospective cohort study by Rohde et al., found that PET/CT was notably more effective in identifying distant metastases and simultaneous cancers in patients with oral cavity cancer (T1-T4) compared to chest CT and head and neck MRI (magnetic resonance imaging).6 SPECT, on the other hand, provides three-dimensional imaging that is particularly useful for assessing bone metabolism and temporomandibular joint (TMJ) disorders.7 Figure 1 highlights the complementary use of PET/CT and SPECT/CT for detecting bone metastases, illustrating the enhanced sensitivity of PET/CT in identifying additional lesions.8
Fig. 1
Recent advancements in hybrid imaging technologies, such as PET/CT and SPECT/CT, have enabled comprehensive anatomical and functional assessment of oral pathologies by combining the strengths of both nuclear and structural imaging.9,10 These hybrid modalities provide high-resolution images, allowing clinicians to precisely localize pathological areas, making it easier to plan targeted treatments and surgical interventions.11,12 PET/CT has shown promise in identifying sentinel lymph nodes in cases of oral cancer, aiding in more accurate staging and treatment planning.13
Integrating these modalities into dental practice could revolutionize diagnostic capabilities, facilitating early detection and personalized treatment planning. For patients with complex cases, such as osteonecrosis of the jaw or severe periodontitis, nuclear imaging may provide essential information on tissue viability and metabolic status that conventional radiography cannot offer.14 Additionally, nuclear imaging can help assess treatment efficacy over time by visualizing changes in tissue metabolism, enabling a more dynamic approach to care that can adapt to the patient’s individual response to therapy.3
This integration of nuclear imaging into dental care represents a significant shift toward precision medicine, where diagnostic insights inform tailored treatment plans, ultimately improving patient outcomes and reducing the risk of overtreatment or complications.15,16
Nuclear imaging, such as PET, presents many challenges in terms of health risks, cost and availability. While radiation exposure during PET imaging is similar to CT imaging, the combination of PET-CT scans increases the dose of radiation the patient is exposed to.17 The purchase price and operational costs of PET-CT units, cause difficulties to its expansions in medical clinics.18
Regarding the accessibility of PET-CT imaging, a survey developed by the CADTH (Canadian Agency for Drugs and Technologies in Health) was sent to numerous medical professionals concerning the potential future of PET-CT in Canada to gain different perspectives of clinicians from many Canadian provinces.19 The respondents indicated several issues concerning the availability and accessibility of PET-CT scans to patients. To list a few of the problems: the reliability on only one single supplier which leads to service disruption, extended use of PET-CT leads to issues with the availability and cost of PET-CT, difficulty sourcing from alternative vendors as well as limited number of radiopharmaceutical suppliers, lack of funding for radiopharmaceuticals resulting in restrictions to the use of PET-CT services.19 In 2022-2023, the CADTH had done national surveys that indicated there were only sixty PET-CT units in nine provinces in Canada, as well as concluding that Canada has 1.5 PET-CT units per million people.18 Although these challenges present themselves, it is stated by the Canadian Cancer Society that the benefit of having a PET scan outweighs the risks of exposure to radiation.20
Radiation therapy
Optimizing radiation therapy for oral cancer treatment requires precise targeting of tumors while minimizing radiation exposure to surrounding healthy tissues. Intensity-modulated radiation therapy (IMRT) and image-guided radiation therapy (IGRT) have emerged as promising approaches for achieving this goal.21,22 IMRT allows for highly accurate radiation dose distribution by modulating the intensity of each beam, thereby sparing adjacent critical structures, such as salivary glands and jawbone, from unnecessary radiation damage.23,24 IGRT further enhances precision by incorporating real-time imaging during treatment, ensuring that radiation is accurately directed even as tumors or patient positioning shifts over time.25
These advanced radiation techniques have demonstrated significant reductions in side effects commonly associated with oral cancer treatment, such as xerostomia, osteoradionecrosis, and trismus.26,27 Collaborative efforts between dentists and nuclear engineers are essential in refining treatment protocols that leverage IMRT and IGRT technologies, which optimize therapeutic efficacy and minimize adverse effects, ultimately improving patient outcomes. Nuclear engineers contribute expertise in radiation dosimetry and imaging, which plays a critical role in designing custom radiation fields that conform to each patient’s unique anatomy, minimizing collateral damage to healthy tissues.28
Recent developments also include proton therapy, which uses positively charged particles that can be more precisely controlled, allowing higher doses to the tumor with even less exposure to surrounding tissues.29 This method is especially advantageous in treating complex cases near critical structures for instance, at the base of the skull.30 Dental involvement is critical in proton therapy planning, as pre-treatment dental assessments can help anticipate and prevent oral complications that might arise from high-dose radiation.31
Nuclear medicine technique: Salivary gland scintigraphy
Salivary gland scintigraphy, also known as sialography, is a nuclear medicine technique that utilizes radiotracers to assess the functional status of salivary glands.32 This non-invasive imaging modality plays a crucial role in identifying disorders such as Sjögren’s syndrome, salivary duct obstruction, and gland inflammation.33 By providing dynamic imaging of gland function, it offers insights beyond what traditional ultrasound or CT imaging methods can achieve.32
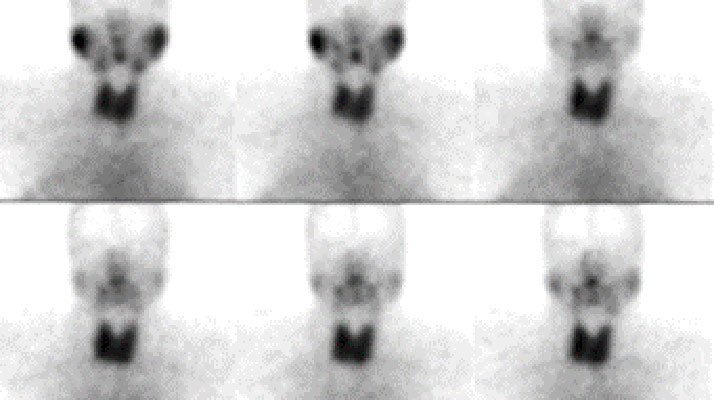
In salivary scintigraphy, a small amount of radiotracer, commonly technetium-99m pertechnetate, is injected intravenously.33 The tracer is selectively taken up by the salivary glands, and gamma cameras capture real-time images as the glands concentrate and excrete the tracer, as seen in Figure 2.33,34 The procedure typically includes stimulating the glands with a sour substance (e.g., citric acid) to observe excretory function.35
Fig. 2

This technique is particularly effective in diagnosing Sjögren’s syndrome, it enables early detection of functional decline before structural changes occur, aiding in the identification and management of xerostomia (dry mouth), commonly seen in patients undergoing radiation therapy or those with systemic diseases.36-38 Identifying the underlying cause aids in tailoring effective treatment plans, including salivary substitutes or stimulants.
Scintigraphy is also used to detect salivary obstructions, such as sialolithiasis, and evaluate inflammation or infections, providing actionable insights for dental interventions such as gland massage or surgery.39,40 Compared to traditional imaging modalities, salivary scintigraphy excels in offering functional data, making it possible to detect physiological abnormalities that static imaging, such as CT or ultrasound might miss and, while its spatial resolution is relatively low, innovations such as hybrid systems (e.g. SPECT/CT and new radiotracers with higher tissue specificity) are addressing these limitations.41
Despite minimal radiation exposure and limited availability in some regions, salivary scintigraphy remains a critical tool in dentistry, providing precise evaluations that contribute to tailored treatment plans and improved patient outcomes. Other nuclear medicine applications such as bone scintigraphy is used for detecting osteomyelitis and other jawbone health concerns. This method is sensitive and can detect the osteoblastic activity of the skeleton.42 Bone scintigraphy is also used for detecting bone abnormalities, bone tumors and several other bone injuries.43 Highlighting the broader scope of nuclear engineering’s role in dentistry.
Radiation safety and protection
Ensuring radiation safety is paramount in dental and nuclear environments, necessitating robust safety protocols and shielding measures. Strict adherence to protocols for dose optimization, shielding, and quality control is critical to minimizing exposure levels while maintaining diagnostic and therapeutic efficacy. The International Commission on Radiological Protection (ICRP) and the American Dental Association (ADA) offer guidelines for optimizing radiation protection in dentistry, emphasizing dose optimization and quality assurance.44,45 In dental practices, these protocols involve the use of protective gear, such as lead aprons and thyroid collars, as well as the careful selection of low-dose imaging techniques to protect sensitive tissues without compromising image quality.46
Although the radiation emitted from x-rays present to patients’ radiation in a low dose, studies have found that repeated exposure to radiation through dental examinations can increase the risk of brain cancer, tumors of the parotid gland, breast cancer and thyroid cancer.4
Collaborations between fields of dentistry and nuclear engineering are instrumental in developing innovative radiation shielding materials and technologies, enhancing workplace safety and patient care. Materials such as lead-free shielding and lightweight composite materials are being tested for effectiveness and comfort in clinical environments.47 These advances not only enhance workplace safety but also reduce patient radiation exposure, supporting safer and more comfortable diagnostic procedures.
Emerging technologies in radiation monitoring, such as wearable dosimeters and real-time radiation sensors, also contribute to a safer environment for healthcare providers. These devices allow for continuous monitoring and immediate feedback on radiation levels, enabling quick adjustments to shielding and exposure time, if necessary.48
Efforts are made to develop alternative diagnostic tools that are non-radiation based such as ultrasounds. Ultrasounds are a non-invasive and inexpensive method to detect hard and soft tissue. It’s done by ultrasonic waves, which transmit energy through a transducer, which converts them into an electrical signal and black and white pictures.49 Ultrasounds are favourable to use with patients whose MRI is contra-indicated because of cardiac pacemakers, claustrophobia and/or metallic prostheses.49
Emerging technologies
The integration of emerging technologies, such as artificial intelligence (AI) and 3D printing, holds immense potential for transforming oral healthcare. AI-powered diagnostic tools are capable of rapidly analyzing complex and large volumes of imaging data, such as radiographs, CBCT scans, and even nuclear imaging modalities, enabling clinicians to identify subtle pathologies that may be missed by the human eye.50 These tools assist in early detection, risk assessment, and personalized treatment planning, streamlining workflows and improving accuracy in diagnosis.50,51 Additionally, AI is being applied in radiation dose optimization, tailoring doses to each patient’s unique anatomy and treatment needs, which enhances safety and reduces unnecessary exposure.52
Furthermore, 3D printing is another transformative technology, particularly valuable for the customization of dental implants and prostheses, enhancing fit, comfort, aesthetics, and surgical guides.53 By creating patient-specific devices, 3D printing allows for a better anatomical fit, improved comfort, and superior aesthetics in dental restorations.53 The precision offered by 3D printing also reduces the time required for adjustments and ensures more accurate, efficient surgical procedures.53
By embracing these cutting-edge technologies, dentistry and nuclear engineering can pioneer new frontiers in patient care and treatment innovation. As these fields continue to explore the applications of AI and 3D printing, they are setting the stage for transformative advancements in oral healthcare, potentially reducing treatment times, enhancing patient comfort, and paving the way for innovative treatment modalities.51
The integration of nuclear engineering in dentistry holds immense promise for advancing personalized medicine. By combining nuclear imaging techniques, such as PET and SPECT, with genomic data, clinicians could tailor diagnostics and treatments to individual genetic profiles.54 This approach has the potential to improve the precision of oral cancer therapies and predict patient-specific responses to radiation treatments, minimizing side effects and optimizing outcomes.
Additionally, nuclear engineering could revolutionize the development of next-generation dental implants and prosthetics. Incorporating radiopaque markers or embedded micro-sensors into implants could enable real-time monitoring of osseointegration, improving post-operative care.55 Furthermore, innovations in biocompatible materials designed using nuclear technologies may enhance implant longevity and functionality, addressing complex oral health challenges.
Collaborations between nuclear engineering and related fields, such as biomedical engineering and pharmacology, could further amplify these advancements. Joint efforts might lead to breakthroughs in radiopharmaceuticals that target specific oral diseases or biomaterials optimized for precision diagnostics.56 Such interdisciplinary approaches could also expand the use of nuclear imaging in dental research, enabling early detection of metabolic or structural changes associated with conditions such as osteonecrosis or TMJ disorders.
The integration of AI in cancer diagnosis, as demonstrated by Dr. April Khademi, enhances diagnostic precision and personalization by improving pathologists’ ability to assess tissue samples.57 This aligns with the potential for AI to revolutionize dentistry, particularly in nuclear imaging. Just as AI refines oncology diagnostics, it could also optimize the analysis of nuclear imaging techniques, for example, PET and SPECT, in diagnosing oral cancers, enabling more tailored and effective treatment plans.57 Both fields stand to benefit from AI’s capacity to improve diagnostic accuracy and patient care.
Despite the transformative potential of advanced imaging techniques and AI healthcare, there are significant global disparities in their implementation. Access to cutting-edge technologies such as PET/CT or AI-driven diagnostics remains limited in low-resource settings due to high costs, inadequate infrastructure, and a shortage of trained professionals.58 These challenges exacerbate health inequities, particularly in rural and underserved areas, where advanced imaging modalities are scarce.59 Addressing these limitations requires a global effort to improve healthcare access, ensure equitable distribution of technology, and train healthcare professionals worldwide to bridge these gaps in care.
Ethical considerations
The integration of radiation-based technologies, such as nuclear imaging and radiation therapy, into oral healthcare raises important ethical considerations, particularly regarding radiation exposure. While techniques namely PET/CT and SPECT provide significant diagnostic benefits, they also involve ionizing radiation, which can pose long-term health risks, including cancer.60 Therefore, balancing the benefits of these technologies with their potential risks is essential for patient safety.
Informed consent is critical when using radiation-based technologies. Patients must be fully informed about the risks and benefits, ensuring they understand the necessity of the procedure and the associated potential long-term effects.61 This allows for informed decision-making and respects patient autonomy.
Additionally, the principle of “justification” dictates that radiation should only be used when the clinical benefit justifies its risks. Healthcare providers must adhere to the “ALARA” (As Low As Reasonably Achievable) principle, minimizing radiation doses while still achieving effective diagnostic results.62
Ethical concerns also extend to healthcare equity. Disparities in access to advanced technologies may lead to unequal care, particularly in low-resource areas.63 Ensuring equitable access to these technologies is essential to address fairness in healthcare.
In summary, integrating radiation-based technologies into oral healthcare must prioritize patient safety, informed consent, and responsible use, ensuring both ethical standards and effective patient care.
Conclusion
The interdisciplinary collaboration between dentistry and nuclear engineering represents a paradigm shift in healthcare, fostering innovation and synergy across diverse domains. By harnessing the collective expertise and resources of these fields, researchers and practitioners can address complex challenges in diagnostic precision, treatment effectiveness, and patient safety, ultimately unlocking new possibilities for enhancing oral health outcomes. This partnership encourages advancements in technology, such as AI-drive diagnostics, precision radiation therapy, novel biomaterials and sialography which contribute to safer and more personalized patient care. Embracing this collaborative spirit is essential for driving sustainable progress and ensuring that oral healthcare continues to evolve in ways that address global health challenges and improve the quality of life for patients worldwide. By championing interdisciplinary approaches, dentistry and nuclear engineering can lay the foundation for a new era of innovation, reshaping healthcare for future generations. 
Oral Health welcomes this original article.
References
- Buch, S. et al. (2017). Nuclear imaging in the field of dentistry: a review, Journal of Turgut Ozal Medical Center, p. 1.
- Baur, D.A., Heston, T.F. and Helman, J.I. (2004). Nuclear Medicine in Oral and Maxillofacial Diagnosis: A Review for the Practicing Dental Professional, The journal of contemporary dental practice, 5(1), pp. 94–104.
- National Research Council (US) and Institute of Medicine (US) Committee on State of the Science of Nuclear Medicine. Advancing Nuclear Medicine Through Innovation. Washington (DC): National Academies Press (US); 2007.
- Hwang SY, Choi ES, Kim YS, Gim BE, Ha M, Kim HY. Health effects from exposure to dental diagnostic X-ray. Environ Health Toxicol. 2018 Dec;33(4):e2018017.
- Kapoor M, Kasi A. PET Scanning. [Updated 2022 Oct 3]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2024 Jan-.
- Rohde M, Nielsen AL, Johansen J, Sørensen JA, Nguyen N, Diaz A, Nielsen MK, Asmussen JT, Christiansen JM, Gerke O, Thomassen A, Alavi A, Høilund-Carlsen PF, Godballe C. Head-to-Head Comparison of Chest X-Ray/Head and Neck MRI, Chest CT/Head and Neck MRI, and 18F-FDG PET/CT for Detection of Distant Metastases and Synchronous Cancer in Oral, Pharyngeal, and Laryngeal Cancer. J Nucl Med. 2017 Dec;58(12):1919-1924.
- López DF, Ríos Borrás V, Muñoz JM, Cardenas-Perilla R, Almeida LE. SPECT/CT Correlation in the Diagnosis of Unilateral Condilar Hyperplasia. Diagnostics (Basel). 2021 Mar 8;11(3):477.
- de Leiris, N., Leenhardt, J., Boussat, B. et al. Does whole-body bone SPECT/CT provide additional diagnostic information over [18F]-FCH PET/CT for the detection of bone metastases in the setting of prostate cancer biochemical recurrence?. Cancer Imaging 20, 58 (2020).
- Jadvar, H., Desai, B., & Ji, L. (2018). PET/CT Imaging in Oral Cancers: A Review. Journal of Nuclear Medicine, 59(6), 882-889.
- Baheti, A. D., Chandanwale, S. S., & Vaidya, A. D. (2020). Role of PET/CT in Head and Neck Cancer. Indian Journal of Nuclear Medicine, 35(2), 107-112.
- Yandrapalli S, Puckett Y. SPECT Imaging. [Updated 2022 Oct 3]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2024 Jan-.
- Beyer T, Townsend DW, Czernin J, Freudenberg LS. The future of hybrid imaging-part 2: PET/CT. Insights Imaging. 2011 Jun;2(3):225-34.
- Madsen CB, Rohde M, Gerke O, Godballe C, Sørensen JA. Diagnostic Accuracy of Up-Front PET/CT and MRI for Detecting Cervical Lymph Node Metastases in T1-T2 Oral Cavity Cancer-A Prospective Cohort Study. Diagnostics (Basel). 2023 Nov 9;13(22):3414.
- Watanabe S, Nakajima K, Kinuya S. ☆Symposium: Imaging modalities for drug-related osteonecrosis of the jaw (5), utility of bone scintigraphy and 18F-FDG PET/CT in early detection and risk assessment of medication-related osteonecrosis of the jaw (secondary publication). Jpn Dent Sci Rev. 2019 Nov;55(1):76-79.
- Malcangi G, Patano A, Guglielmo M, Sardano R, Palmieri G, Di Pede C, de Ruvo E, Inchingolo AD, Mancini A, Inchingolo F, Bordea IR, Dipalma G, Inchingolo AM. Precision Medicine in Oral Health and Diseases: A Systematic Review. J Pers Med. 2023 Apr 25;13(5):725.
- Barbet J, Bernaudin M, Payoux P, Cicone F, Gaugler MH, Kraeber-Bodéré F. Editorial: Nuclear Medicine in the Context of Personalized Medicine. Front Med (Lausanne). 2020 Jun 9; 7:252.
- Mafraji, Mustafa. Positron Emission Tomography (PET) – Positron Emission Tomography (PET). Merck Manual Consumer Version, Nov. 2023.
- CADTH. Canadian Medical Imaging Inventory 2022–2023: PET-CT and PET-MRI. Canadian Journal of Health Technologies. 4(8), 13 August 2024.
- CADTH. Canadian Medical Imaging Inventory Service Report the Future of PET-CT in Canada Canadian Medical Imaging Inventory Service Report the Future of PET-CT in Canada Context. Oct. 2021
- Lee, Sid. Positron Emission Tomography (PET) Scan. Canadian Cancer Society, 2015.
- Lee, T. F., Chao, P. J., & Fang, F. M. (2017). Intensity-Modulated Radiation Therapy for Head and Neck Cancer: Emphasis on Clinical Outcomes and Radiation-Related Toxicities. Head & Neck, 39(5), 1082-1088.
- Nutting, C. M., & Dearnaley, D. P. (2018). Image-Guided Radiation Therapy for Head and Neck Cancer: Where Do We Go from Here? Clinical Oncology, 30(1), 5-8.
- Cheung K. Intensity modulated radiotherapy: advantages, limitations and future developments. Biomed Imaging Interv J. 2006 Jan;2(1): e19.
- Studer G, Zwahlen RA, Graetz KW, Davis BJ, Glanzmann C. IMRT in oral cavity cancer. Radiat Oncol. 2007 Apr 12; 2:16.
- Dawson LA, Sharpe MB. Image-guided radiotherapy: rationale, benefits, and limitations. Lancet Oncol. 2006 Oct;7(10):848-58.
- Ma SJ, Rivers CI, Serra LM, Singh AK. Long-term outcomes of interventions for radiation-induced xerostomia: A review. World J Clin Oncol. 2019 Jan 10;10(1):1-13.
- Somay E, Yilmaz B, Topkan E, et al. Assessment of the Impact of Osteoradionecrosis on Quality-of-Life Measures in Patients with Head and Neck Cancer. In: Sergi CM, editor. Advancements in Cancer Research [Internet]. Brisbane (AU): Exon Publications; 2023 Aug 17. Chapter 3.
- Bortfeld T, Jeraj R. The physical basis and future of radiation therapy. Br J Radiol. 2011 Jun;84(1002):485-98.
- Nuyts S, Bollen H, Ng SP, Corry J, Eisbruch A, Mendenhall WM, Smee R, Strojan P, Ng WT, Ferlito A. Proton Therapy for Squamous Cell Carcinoma of the Head and Neck: Early Clinical Experience and Current Challenges. Cancers (Basel). 2022 May 24;14(11):2587.
- Tang DM, Cutri RM, Wu AW, Patil C, Zumsteg ZS. Proton Therapy for Skull Base Chondrosarcoma. J Neurol Surg Rep. 2023 Nov 20;84(4):e144-e145.
- Falek S, Regmi R, Herault J, Dore M, Vela A, Dutheil P, Moignier C, Marcy PY, Drouet J, Beddok A, Letwin NE, Epstein J, Parvathaneni U, Thariat J. Dental management in head and neck cancers: from intensity-modulated radiotherapy with photons to proton therapy. Support Care Cancer. 2022 Oct;30(10):8377-8389.
- Anton Faria R, Chagas Jaguar G, Nóbrega Pereira Lima E. Parametric imaging in salivary gland scintigraphy. Nucl Med Commun. 2024 Dec 1;45(12):1098-1104.
- Nadal M, Levy M, Bakhsh A, Joly A, Maruani A, Vaillant L, Erra B, Samimi M. Salivary scintigraphy for Sjögren’s syndrome in patients with xerostomia: A retrospective study. Oral Dis. 2018 May;24(4):552-560.
- Loutfi I, Nair MK, Ebrahim AK. Salivary Gland Scintigraphy: The Use of Semiquantitative Analysis for Uptake and Clearance. Journal of Nuclear Medicine Technology. 2003 Jun 1;31(2):81–5.
- Gardner A, So PW, Carpenter G. Endogenous salivary citrate is associated with enhanced rheological properties following oral capsaicin stimulation. Exp Physiol. 2020 Jan;105(1):96-107.
- Saito T, Fukuda H, Horikawa M, Ohmori K, Shindoh M, Amemiya A. Salivary gland scintigraphy with 99mTc-pertechnetate in Sjögren’s syndrome: relationship to clinicopathologic features of salivary and lacrimal glands. J Oral Pathol Med. 1997 Jan;26(1):46-50.
- Baer AN, Walitt B. Sjögren Syndrome and Other Causes of Sicca in Older Adults. Clin Geriatr Med. 2017 Feb;33(1):87-103.
- Álvarez Troncoso J, Giraldo González LF, Coronado Poggio M, Sorriguieta Torre R, Ruiz Bravo-Burguillos E, Domínguez Gadea L, Soto Abánades C. Salivary Gland Scintigraphy in Sjögren’s Syndrome: A Retrospective Study of Diagnostic Accuracy and Correlation With Histological and Immunological Biomarkers. Cureus. 2024 Jul 24;16(7):e65305.
- Chen YC, Chen HY, Hsu CH. Recent Advances in Salivary Scintigraphic Evaluation of Salivary Gland Function. Diagnostics (Basel). 2021 Jun 28;11(7):1173.
- Yoshimura Y, Morishita T, Sugihara T. Salivary gland function after sialolithiasis: scintigraphic examination of submandibular glands with 99mTc-pertechnetate. J Oral Maxillofac Surg. 1989 Jul;47(7):704-10; discussion 710-1.
- Klutmann, S. et al., 1999, Quantitative Salivary Gland Scintigraphy.
- Ferreira RI, de Almeida SM, Bóscolo FN, Santos AO, Camargo EE. Bone scintigraphy as an adjunct for the diagnosis of oral diseases. J Dent Educ. 2002 Dec;66(12):1381-7.
- Bombardieri E, Aktolun C, Baum RP, Bishof-Delaloye A, Buscombe J, Chatal JF, Maffioli L, Moncayo R, Morteímans L, Reske SN. Bone scintigraphy: procedure guidelines for tumour imaging. Eur J Nucl Med Mol Imaging. 2003 Dec;30(12): BP99-106.
- International Commission on Radiological Protection (ICRP). (2018). Radiation Protection in Dentistry. ICRP Publication 142. Oxford: Elsevier.
- American Dental Association (ADA). (2020). Radiation Safety in Dental Practice: Recommendations of the ADA Council on Scientific Affairs. Journal of the American Dental Association, 151(7), 537-544.
- Health Canada (2019) Radiation Protection in Dentistry – Recommended Safety Procedures for the Use of Dental X-Ray Equipment – Safety Code 30 – Canada.ca.
- Tungsten-based material as promising new lead-free gamma radiation shielding material in nuclear medicine. Jamal AbuAlRoos, Nadin et al. Physica Medica: European Journal of Medical Physics, Volume 78, 48 – 57.
- P. Papadimitroulas et al., “A Review on Personalized Pediatric Dosimetry Applications Using Advanced Computational Tools,” in IEEE Transactions on Radiation and Plasma Medical Sciences, vol. 3, no. 6, pp. 607-620, Nov. 2019.
- Shah N, Bansal N, Logani A. Recent advances in imaging technologies in dentistry. World J Radiol. 2014 Oct 28;6(10):794-807.
- Hartoonian S, Hosseini M, Yousefi I, Mahdian M, Ghazizadeh Ahsaie M. Applications of artificial intelligence in dentomaxillofacial imaging: a systematic review. Oral Surg Oral Med Oral Pathol Oral Radiol. 2024 Nov;138(5):641-655.
- Schwendicke, F., Golla, T., & Dreher, M. (2019). Artificial Intelligence in Dentistry: Chances and Challenges. Journal of Dental Research, 98(10), 1055-1062.
- Ng CKC. Artificial Intelligence for Radiation Dose Optimization in Pediatric Radiology: A Systematic Review. Children (Basel). 2022 Jul 14;9(7):1044.
- Chander, R., & Kapoor, D. (2019). 3D Printing in Restorative Dentistry: A Review. Journal of Prosthodontics, 28(2), 146-153.
- Fullwood MJ, Wei CL, Liu ET, Ruan Y. Next-generation DNA sequencing of paired-end tags (PET) for transcriptome and genome analyses. Genome Res. 2009 Apr;19(4):521-32.
- Scherer MD, Roh HK. Radiopaque dental impression method for radiographic interpretation, digital alignment, and surgical guide fabrication for dental implant placement. J Prosthet Dent. 2015 Apr;113(4):343-6.
- Dhoundiyal S, Srivastava S, Kumar S, Singh G, Ashique S, Pal R, Mishra N, Taghizadeh-Hesary F. Radiopharmaceuticals: navigating the frontier of precision medicine and therapeutic innovation. Eur J Med Res. 2024 Jan 5;29(1):26.
- Canadian Cancer Society. How Dr April Khademi Is Improving Cancer Diagnosis with AI. Cancer.ca, Nov. 2024.
- Rashid, A., Ashfakul K. AI Revolutionizing Industries Worldwide: A Comprehensive Overview of Its Diverse Applications. Hybrid Advances. 2024 August 1. 7;100277:(100277–100277).
- Gallach M, Mikhail Lette M, Abdel-Wahab M, Giammarile F, Pellet O, Paez D. Addressing Global Inequities in Positron Emission Tomography-Computed Tomography (PET-CT) for Cancer Management: A Statistical Model to Guide Strategic Planning. Med Sci Monit. 2020 Aug 27;26:e926544.
- Nievelstein RA, Quarles van Ufford HM, Kwee TC, Bierings MB, Ludwig I, Beek FJ, de Klerk JM, Mali WP, de Bruin PW, Geleijns J. Radiation exposure and mortality risk from CT and PET imaging of patients with malignant lymphoma. Eur Radiol. 2012 Sep;22(9):1946-54.
- Hurwitz MD, Chundury A, Goodman CR, Jones J, Lo SS, Saeed H, Small W Jr, Schechter NR. ACR-ARS Practice Parameter on Informed Consent Radiation Oncology. Am J Clin Oncol. 2023 Jun 1;46(6):231-235.
- Strauss KJ, Kaste SC. The ALARA (as low as reasonably achievable) concept in pediatric interventional and fluoroscopic imaging: striving to keep radiation doses as low as possible during fluoroscopy of pediatric patients–a white paper executive summary. Pediatr Radiol. 2006 Sep;36 Suppl 2(Suppl 2):110-2.
- Lawson MB, Scheel JR, Onega T, Carlos RC, Lee CI. Tackling Health Disparities in Radiology: A Practical Conceptual Framework. J Am Coll Radiol. 2022 Feb;19(2 Pt B):344-347.
About the authors

Dr. Sherif Elsaraj earned his dental degree and a Master’s in Oral Biology from the University of Manitoba, followed by a Ph.D. in Craniofacial Pain and Health Sciences from McGill University. He currently supervises residents in the General Residency Program at the Jewish General Hospital, specializing in temporomandibular disorders (TMD) and orofacial pain at the hospital’s clinic.

Raquel Kasha-Blois, a graduate in Biomedical Sciences from the University of Ottawa, is committed to advancing dental research and healthcare, with a notable publication reflecting her dedication to the field.

Dr. Hossam Shalabi, a Senior Nuclear Fire & Explosion Expert with more than 20 years of experience, represents Canada on the global stage and contributes to the advancement of engineering through his research and teaching.

Emad Shalabi, a Biomedical Science student at the University of Ottawa with a prestigious full scholarship, has authored two publications that demonstrate his commitment to advancing scientific research.