Abstract
Tooth wear has become an increasingly prevalent condition across age groups. With this article, we want to explore the etiology, classification, diagnosis, and management of tooth wear, focusing on the interplay of mechanical, chemical, and biomechanical factors such as abrasion, attrition, erosion, and abfraction. Dietary habits, parafunctional activities, systemic diseases, and oral hygiene practices are examined as contributing risk factors. In this article we outline a risk-based, phased approach to treatment, incorporating preventive strategies, non-invasive interventions, and restorative techniques using contemporary materials like composites, glass ionomers, and ceramics. Additionally, the psychosocial impact of tooth wear and its effect on quality of life are highlighted.
Emerging research in biomimetic materials, remineralization technologies, and digital dentistry offer promising future directions for minimally invasive, patient-centered care. This review emphasizes the importance of early diagnosis, interdisciplinary collaboration, and long-term maintenance to successfully manage tooth wear and preserve oral function and aesthetics.
As we know, tooth wear is the irreversible loss of dental hard tissues caused by mechanical, chemical, or biomechanical processes and is posing a growing concern worldwide.1,2 Unlike dental caries or trauma, tooth wear lacks infectious etiology but has a pronounced functional, aesthetic, and psychosocial toll. It is our duty as dental professionals to understand the complex causality, early signs, risk stratification, and multi-modal management to preserve dentition and oral health.
Epidemiology
Tooth wear prevalence varies by age and geography. In adults, moderate wear affects ~30–45%, severe in 10–15%; among adolescents, mild wear may reach 60%, with destructive erosion in ~5%.3 Erosion prevalence parallels dietary acid intake and increased reflux occurrence.
Types of tooth wear
1- Abrasion1 is defined to be the loss of tooth substance due to mechanical friction from foreign objects (Fig. 1).
Fig. 1
Causes: Incorrect brushing technique (hard bristles of tooth brushes, horizontal strokes with abrasive pastes), certain oral habits, habitual manipulation with different objects (toothpicks, pens), denture clasp friction.
Clinical presentation: V-shaped cervical notches at the cervical margin of teeth, often associated with a specific brushing technique, abrasive toothpaste or habits like holding objects between teeth (e.g. pen biting or pipe smoking). These surfaces are often smooth with dentin exposure.
Histology: Enamel prisms may fracture; dentin tubules smear; lesion walls show fatigue failure lines.
Management: Behavior modification and restoration once beyond enamel.
2- Attrition3 is the act of wearing or grinding down by friction resulting from mastication or parafunction, limited to the contacting surfaces of the teeth (Figs. 2A and 2B).
Fig. 2A
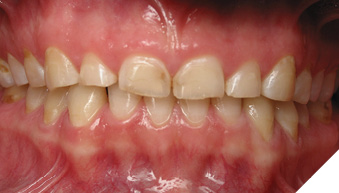
Fig. 2B
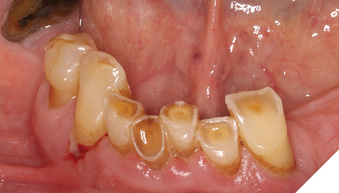
Mechanism: Occluding teeth wear opposing surfaces and bruxism accelerates this process.
Lesion characteristics: flat, polished wear facets on occlusal and incisal surfaces that often match opposing teeth. With progression, it can lead to reduction of cusp height, flattening the occlusal inclined planes. It can also lead to dentinal exposure which will likely result in an increase in the rate of wear and sensitivity. Depending on the rate of wear, hypersensitivity may not be present due to the formation of secondary dentine.
Contributing features: Parafunctional habits (awake clenching, sleep bruxism), malocclusions, occlusal interferences.
Implication: Can destabilize occlusion, change vertical dimension, and lead to temporomandibular disorders (TMD) syptoms. Esthetics become also a concern when the attrition is moderate to advanced.
3- Erosion3-5 is the loss of dental hard tissues due to a chemical process of acid dissolution not involving bacteria and not directly associated with traumatic factors (Figs. 3 and 4).
Fig. 3
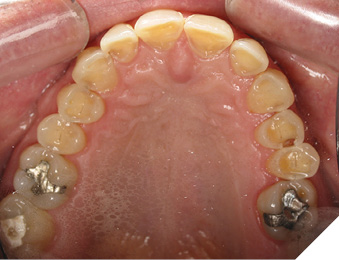
Fig. 4
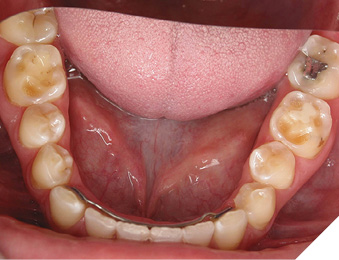
Sources: Extrinsic acids (citrus, sodas, sports drinks); intrinsic acids (GERD, bulimia). Other cofactors may exist from the consumption and/or use of certain medications or oral hygiene products. Some recreational drugs and dietary supplements have also been associated with an increased risk of developing erosion.
Lesion characteristics: Smooth, scooped-out, or cupped-out lesions, often glazed appearance, loss of surface texture, translucency of incisal edges, and “dentine islands” where dentine is exposed. It can lead to “restoration halo” where fillings appear to stand proud of the worn tooth surface. Erosion may reach root surfaces in gingival recession.
Implications: Acid-softened surfaces are more susceptible to abrasive forces.
4- Abfraction2 is the loss of tooth structure at the cervical area caused by flexural forces on the tooth during occlusal loading (e.g. clenching and grinding) (Fig. 5).
Fig. 5
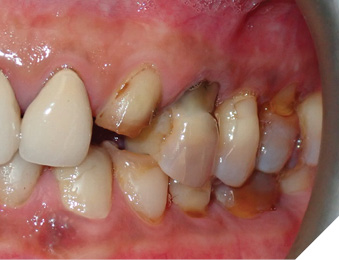
Proposed mechanism: It is produced by the tensile flexural stresses at cervical enamel-complex during mastication or parafunction.
Lesion characteristics: Sharp, wedge-shaped cervical lesions, often asymmetrical.
Controversy: Role debated; may represent susceptibility zones for abrasion when combined with acid/brush forces.
Consultation appointment
We should be thorough during the consultation appointment to gather all the information to reach a proper diagnosis and offer the best treatment to our patients who will present to us with different needs. We should keep in mind that there could be a combination of tooth wear etiologies.7
Patient History
Dietary diary: The patient should tell us about their diet and frequency/timing of acidic food/drink intake if any. We should also ask about the nature of these drinks which could be rich in citric, phosphoric or carbonic acids. Lifestyle factors like lemon-squeezing or lozenge use should also be documented.
Medical review: Has our patient been diagnosed with gastroesophageal regurgitation disorder (GERD) and if so, what are the medications she/he is taking. If we suspect eating disorders, we should be careful how we approach this subject with our patients. They might have not told anyone about this problem. We should show interest and compassion without judgment. They might also be taking multiple medications that produce xerostomia, which will work against salivary buffer capacity. Sleep disturbances have been associated with sleep bruxism. The prevalence of sleep bruxism in adult patients with obstructive sleep apnea (OSA) and other sleep disorders has been found to be higher than that in the general population. The specific mechanisms behind these positive associations has not been identified.8
Frequency matters: Erosion extent depends more on repeated acid exposure than pH alone. We should ask how often do they ingest acidic drinks/foods or how often do they vomit.
Parafunctional behaviors: High amplitude forces fatigue enamel faster than masticatory loading. Are the patients aware of stress-related clenching? Do they wear night guards? Do they break their nightguards often due to bruxism? Do they wake up with morning jaw fatigue?
Brushing habits: We should ask patients to show us their brushing technique, the type of brush and toothpaste they use and the timing of brushing after acid intake.
Habits: Do they have a tendency to nail or pen biting, to hold objects in their mouth, or to chew on hard foods/seeds?
Multifactorial Synergy: Tooth wear rarely exists in isolation. Acid-related enamel softening plus abrasive brushing leads to accelerated cervical wear; bruxism plus erosion can increase incisal shortening; abfraction zones may fill debris and increase plaque retention causing dental caries. Acid regurgitation in a xerostomic patient will increase the tooth structure loss.
Clinical examination
A thorough and proper clinical examination is important to document the extent of tooth wear and to determine appropriate treatment and management. Some key areas to consider are:
Extraoral examination:
- Temporomandibular Joints (TMJs) and associated musculature
- Cervical lymph nodes and salivary glands
- Facial (and dental-facial) features (proportions, symmetry, facial shape, profile and width and lip morphology and mobility
An evaluation of the TMJ will include an assessment of the range of movement including the maximum mandibular opening as measured by the inter-incisal distance. Any distance less than 35mm is considered restricted movement. Similarly, lateral movement should be assessed and the normal range is considered to be around 12mm. Any deviation on opening and closure path should also be noted.9
The TMJ complex can be palpated in the pre-auricular region for any signs of tenderness. In addition, the placement of index fingers into the extra auricular meatus (EAM) and asking the patient to open and close and attempting to manipulate the mandible into a retruded position are all part of the examination.
Along with mandibular manipulation and palpation of the joint area, the use of a stethoscope can be used to detect any anomalous joint sounds such as dislocation or crepitation sounds. The presence of a click along with any correlation with symptoms as well as the timing of the click can give insights on the joint structure. When it comes to the muscular examination, this is often performed by palpating the muscles, noting any signs of hypertrophy, tenderness or discomfort in areas of muscle insertion as well as any evidence of asymmetric representation of the chewing muscles (masseter and temporalis). As for the lymph nodes and the salivary glands, they should be non-tender when palpated. Of more clinical significance, the enlargement of parotid glands may be related to alcoholism, diabetes, Sjogren’s syndrome, eating disorders and HIV. Any signs of hyposalivation may be considered a cofactor of wear lesion as stated before. Patients with hyposalivation may have difficulty in swallowing, controlling their dentures, phonation, unpleasant taste or even loss of taste as well as halitosis.
Facial features should be assessed carefully as they can impact management strategies. Specifically, the vertical facial proportions, facial symmetry, facial profile and shape and width are all considered.
The assessment of the vertical dimension of occlusion can be done using the facial of thirds for proportion. The freeway space using a set of callipers can be assessed with the suggested space to be in the range of 2-4mm. The use of sibilant sounds by ways of phonetics is another method of determining vertical dimension of occlusion.
Facial symmetry can be evaluated by looking at the facial midline and the interpupillary line as vertical and horizontal reference planes. These will serve as part of an aesthetic evaluation. In addition, the interpupillary line is also used as a reference in determining the incisal, gingival and occlusal planes which will be relevant in anterior tooth wear cases. Another important evaluation is to note whether there is a cant in these reference planes.
The lateral facial profile is also an important assessment. The use of E-line formed by connecting the tip of the nose to the tip of the chin and to see if the upper and lower lips are 4mm and 2mm posterior to this E-line helps to indicate whether the patient has a normal profile, convex profile and concave profile. This determination may influence the treatment decision of how to set the upper anterior teeth for proper lip support and whether to increase or keep the vertical dimension of occlusion in supporting the current facial profile or to improve the profile.
The assessment of facial shape from ovoid, square, tapering or square tapering historically has been used to determine moulds selection for removable prosthesis. For severe wear cases where the patient has lost a good portion of tooth structure, such information may be useful for the practitioner to restore anterior worn dentition to proper morphological structures.
In addition, the lip evaluation is also an important step especially in anterior wear dentition. The morphology, the lip fullness and symmetry can serve an important evaluation for a smile design in restoring anterior wear dentition. The amount of lip movement that occurs when a patient smiles and the amount of tooth display at rest and in dynamic position will influence smile design in where to position the incisal edges of the anterior teeth reconstruction that will be both functional and aesthetic.
According to Vig and Brundo,10 tooth display varies with age. The average tooth display at rest is around 3.0 to 3.5mm for 30 years old or younger. Patients aged 50 years will show around 1.0- 1.5mm of tooth structure. For 70 years and older, tooth display may be around 0.0 to 0.5mm at rest. It is important to note these averages, especially when contemplating at lengthening incisal edges which may impact tooth display at rest. Other consideration would be phonetic tests of fricative sounds which can help determine the proper relationship of upper incisal edges to the lower lip.
It is recommended to use intra-oral and extra-oral pictures and short videos to evaluate the patient at rest, during speaking and laughing to evaluate the aesthetic need and the ideal length of the incisors.
Intra oral examination
- Soft tissue assessment.
- An assessment of dental hard tissues.
- A periodontal examination.
- An occlusal assessment.
- An evaluation of the aesthetic zone.
- An appraisal of any edentulous spaces.
- Document wear patterns: Incisal edge losses, flattened cusp tips, cervical abrasions, smooth erosion patches.
- Shade changes imply dentin exposure.
- Check occlusion/TMD signs: TMJ noise, pain, muscle palpation tenderness.
- Dentin hypersensitivity: Air/water triggers.
Diagnostic tools
Intraoral photographs: Photos are very important. Ideally have photos taken at same angulation with same lighting and same camera settings for future comparison.
Intraoral scans/study casts and occlusal records: Visualizing vertical dimension and wear facets.
Salivary analysis: check for volume milking the parotid and submandibular glands since low saliva increases risk.
Classification systems
Any simple and effective method to screen for the presence of tooth wear will be helpful. This provides a means for determining progression and/or quiescence. Furthermore, it can form the basis for determining a protocol for future care.
Currently, there are a number of indices proposed. Unfortunately, there is no universally accepted index for the recording of tooth wear. The problem stems from having a system that has minimal inter-examiner to intra-examiner variability, along with a level of sensitivity that permits the monitoring of subtle changes over a period of time.
Nonetheless, an appropriate grading will permit targeted management strategies. Below is one of many of indexes on tooth wear.
BEWE risk score grading and clinical management guide
| Risk Level | Cumulative Score of all Sextants | Management Guide |
| None | Less than or equal to 2 | Routine maintenance and observation. Review at two yearly intervals. |
| Low | Between 3 and 8 | As above plus oral hygiene, dietary assessment, advice. |
| Medium | Between 9 and 13 | As above plus identify the main aetiological factors and eliminate them. Avoid restorations, apply fluorides, casein-derived pastes, surface sealants. Monitor at 6-12 month intervals (casts,photos, scratch test) |
| High | 14 and over | As above plus minimal restorations |
Basic Erosive Wear Examination (BEWE)
This tooth wear index was proposed by Bartlett et al in 2008.11 It aims at recording the severity of erosive tooth wear for patients in a primary care setting. It is designed to be simple to use. The table above is the protocol for using the Basic Erosive Wear Examination:
- Diagnose the presence of tooth wear, eliminating teeth with trauma and developmental defects from the score.
- Examine all teeth and all surfaces of teeth in the mouth for tooth wear.
- Identify each quadrant that has the most severely affected tooth wear.
- Conduct the BEWE
The BEWE records the severity of wear on a scale from 0 to 3 in each sextant based on the following criteria:
- 0 = no erosive wear
- 1 = initial loss of surface texture
- 2 = distinct defect; hard tissue loss less than 50% of the surface area*
- 3 = hard tissue loss equal to or greater than 50% of the surface area*
*dentine often involved but may be difficult to detect, especially in the cervical areas
The limitation of this screening is that it does not consider multifactorial etiology nor the measurement of progression. A visual appraisal can be difficult to distinguish between more or less than 50% of the surface area loss and it does not provide suggestions on clinical management.
Risk assessment and treatment planning
Risk factor identification:
Behavioral: Brushing patterns, diet, parafunction.
Medical: Acid reflux, xerostomia, medications.
Occlusal: Malalignment, wear facet mapping.
Saliva: Flow, composition.
Diagnostic tools
Wear scoring systems: BEWE, TWI.
Salivary testing: Unstimulated flow, buffering.
pH monitoring: If intrinsic acid is suspected.
Objectives of treatment
- Stop progression: this will depend on the etiology of the tooth wear.
- Relieve symptoms: endodontic treatment, immediate restorations, prescription medications, Botulinum toxin injections.12
- Rehabilitate form and function: it will also depend on the patient’s expectations and main complaints.
- Preserve vertical dimension and occlusal stability.
- Reinforce preventative maintenance: recalls to preserve and maintain our restorations as well as ensure the patient continues to be motivated.
Phases of care
Phase 1: Risk control and stabilization.
Phase 2: Restoration of tissue loss (non-invasive to invasive).
Phase 3: Maintenance and recall monitoring.
Preventive and non-restorative strategies
Dietary management:
- Reduce acidic intake; or consume with meals.
- Avoid sipping sports or energy drinks.
- Use straws, rinse with water post-acid exposure.
- Encourage dairy consumption to help buffer acids.
Oral hygiene education:
- Use soft-bristle brush and low-abrasivity toothpaste (RDA <70).
- Avoid brushing within at least 30 minutes of acid ingestion.
- Recommend gentle circular brushing.
Fluoride and desensitizing regimens:
- Prescription-level fluoride toothpaste (≥5000 ppm F).
- 6–12 month topical fluoride varnish or gel application.
- Use of potassium nitrate-based toothpaste for hypersensitivity.
Salivary support:
- Stimulate saliva via sugar-free gum or lozenges.
- Hydration reminders.
- Prescription sialagogues if medically appropriate.
Medical collaboration:
- Referral for management of GERD (H2-blockers, PPIs, lifestyle).14
- Mental health support for eating disorders.
- Referral for botulinum toxin injections on masticatory muscles.
- Referral for diagnosis and management of OSA. 8
Protective appliances:
- Hard acrylic night guards to intercept attrition.
- Soft silicone guards when bruxism causes soreness.
- Temporary nocturnal guards to protect newly placed restorations.
Restorative management
Discuss with patients what they want to achieve when addressing their tooth wear. Is it mainly to restore function? Is it aesthetics? Is it to get rid of tooth sensitivity or TMJ pain?15
Indications for restoration:
- Cosmetic concern
- Functional impairment
- Sensitivity or pain
- Microleakage/caries risk in lesions
- Risk of progressive tissue loss
Material choices
Composite restorations
Direct composites: This option is conservative, bioadhesive, and can be repaired often.16,17
Indirect resin/onlay systems: Improved occlusal anatomy and wear resistance. It is more costly than direct composites but offers great aesthetics.18
Glass Ionomer Cements (GIC)
- Useful in cervical lesions, fluoride release, but it has limited mechanical properties. It can be used when caries risk is moderate to high.16
Ceramic and non-ceramic materials
- Lithium disilicate, zirconia: Indicated for larger load-bearing or esthetically critical restorations.18
- Gold crowns, especially when there is limited prosthetic space, like in the second molars.
Sealants and high-fluoride coatings
- Resin sealants to prevent lesion deepening.
- Acidulated phosphate fluoride varnish.16
Occlusal considerations and splint therapy
Occlusal adjustments: Some clinicians will favour some occlusal equilibration:19
- Identify premature closure points via articulating film.
- Use minimal enameloplasty targeting early contacts only.
- Avoid over-adjustment to preserve tooth structure.
Splint design and use: This is a great option for attrition tooth loss, even when the patient chooses to not restore first. The main problem remains user compliance from patients. It is recommended a hard acrylic splint for high-load bruxism, with Full occlusal coverage required. It is recommended for its use overnight and occasional daytime during stress. Regular adjustment to maintain balanced occlusion.19
Bruxism monitoring: Use wear facets or occlusal tablets to evaluate continued parafunction. Regular follow-up for splint checking, patient adherence. The injection of botulinum toxin in the masticatory muscles (masseter and temporalis) has been documented with various results.13
Phased rehabilitation approach
Phase 1 – Stabilization: It is advisable to address the cause of tooth wear first before going to the restorative phase. Depending on the cause, we could advise patients on:
A. Behavior/habit modification
B. Dietary change
C. Oral hygiene refinement
D. Fluoride and saliva management
E. Provision of occlusal therapy appliances.
Phase 2 – Restorative
- Diagnostic wax-ups, mock-ups, and provisional restorations for testing aesthetics and comfort.
- Gradual vertical dimension restoration if necessary.
- Definitive restorative work via composites, partial or full coverage restorations of different materials: gold, porcelain fused to metal, different ceramics.
Phase 3 – Maintenance
- Recall every 3–6 months, depending on the severity of symptoms at the initial appointment.
- Photographic wear-level comparisons.
- Reapply protective agents and check occlusion.
- Update splint treatment as needed.
Special patient populations
Children and adolescents: We should focus on early identification of enamel wear and to limit invasive interventions. Educate caregivers on managing bottle feeding and acidic drinks. Use preventive sealants and monitor diet.
Geriatric patients: There is an increased incidence due to medications inducing hyposalivation, parafunction, and gingival recession. Discuss options with pharmacists and family doctors or specialists.
Medically compromised / special needs: Tailored behavior approach for neurologic disorders, xerostomia, and limited oral hygiene. Xerostomia intervention is paramount in radiation or autoimmune patients. GERD or bulimia patients need multi-disciplinary care.
Psychosocial impact and quality of life:
Esthetic changes to the dentition due to wear can negatively affect patient self-confidence and social engagement, while masticatory dysfunction may limit dietary variety and compromise nutritional status. Pain and dentin hypersensitivity can further interfere with daily activities and overall well-being. Quality-of-life impacts can be systematically measured using validated instruments such as the Oral Health Impact Profile (OHIP-14), which is widely applied in research, clinical practice, and oral health program planning. The OHIP facilitates evaluation of the functional, psychological, and social effects of oral conditions, as well as the outcomes of dental treatments and public health interventions.19 Incorporating motivational interviewing into patient care may enhance engagement and support sustained positive behavior change.
From infinity and beyond: New research
There is a growing body of emerging research focused on mitigating tooth loss and enhancing natural reparative processes. Substantial progress has been achieved in the development of biomimetic and bioactive materials aimed at promoting dentin-pulp complex regeneration and enamel remineralization. Ongoing investigations are evaluating novel remineralizing agents, stem cell–based therapies, and bioengineered scaffolds designed to restore the structural and functional integrity of dental tissues.18-22
Novel remineralizing agents
Casein phosphopeptide–amorphous calcium phosphate (CPP-ACP, Recaldent®) has demonstrated significant increases in enamel resistance to acid challenge in vitro.15 Nano-hydroxyapatite (n-HA) toothpaste is emerging as a promising fluoride substitute, exhibiting uniform enamel surface repair, reduction in biofilm adhesion, and improved resistance to demineralization.20,21
Biomimetic repair
Self-healing glass ionomer composites have shown the ability to seal early micro-cracks through ion exchange and matrix swelling mechanisms. Bioactive restorative materials capable of sustained release of calcium, phosphate, and fluoride ions promote ongoing remineralization and enhance the longevity of restorations.22
Digital modeling and 3D printing
Advances in digital dentistry now allow precise simulation and planning for vertical dimension of occlusion (VDO) increase. Additive manufacturing enables the fabrication of highly customized night guards, occlusal overlays, and provisional prostheses with exceptional fit and functional predictability.23
Genetic and biochemical insights
Recent studies have identified associations between genetic variations in enamel matrix protein genes (e.g., AMELX) and susceptibility to mechanical wear such as bruxism. Proteomic profiling of saliva has revealed specific protein signatures that may serve as biomarkers for early detection of erosive acid exposure.24
Early erosion detection
Non-invasive optical techniques, including laser fluorescence and optical coherence tomography (OCT), allow detection of subclinical enamel softening before visual or tactile changes occur, enabling targeted preventive intervention.25
Tooth wear secondary to parafunction: A case
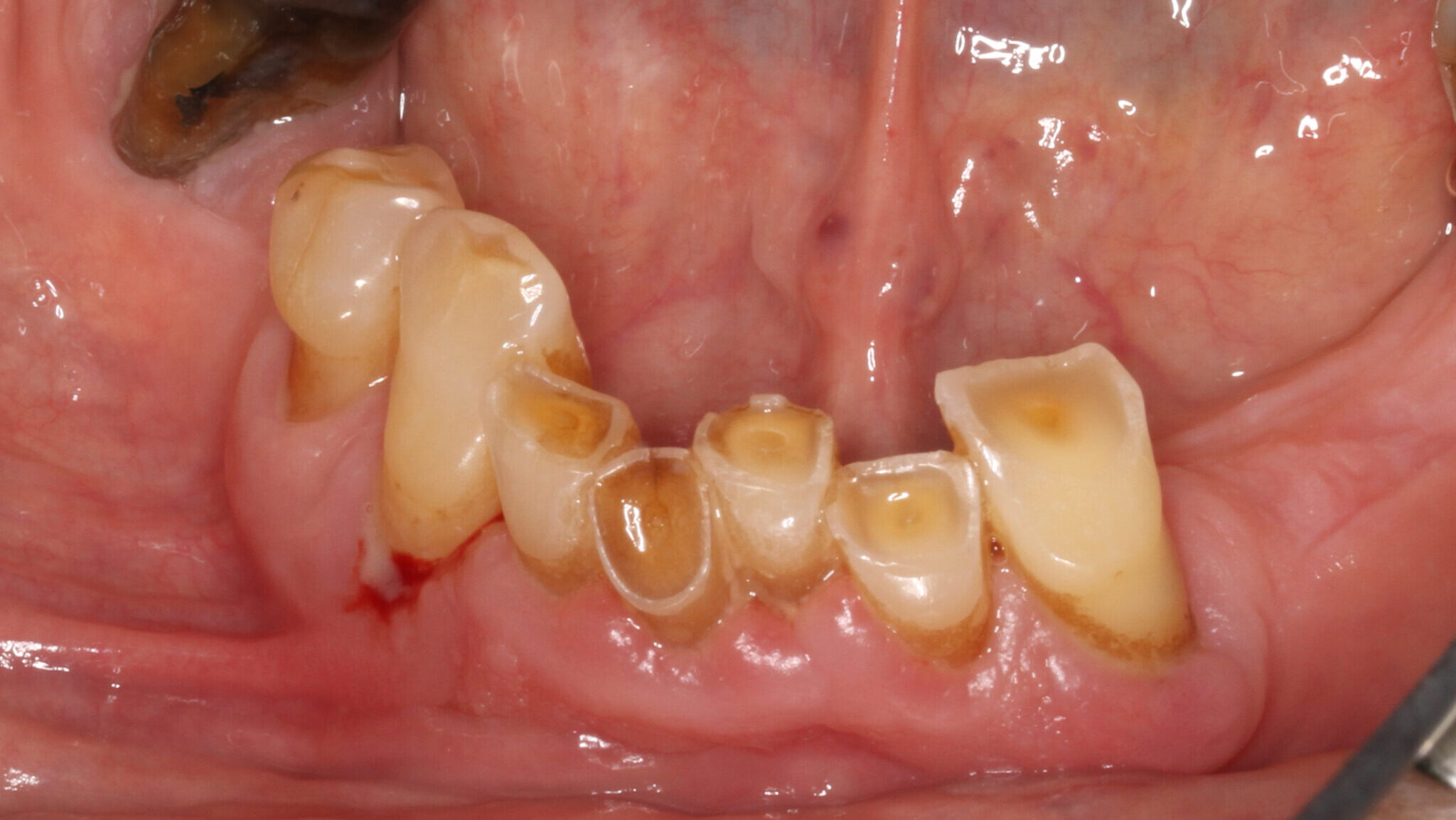
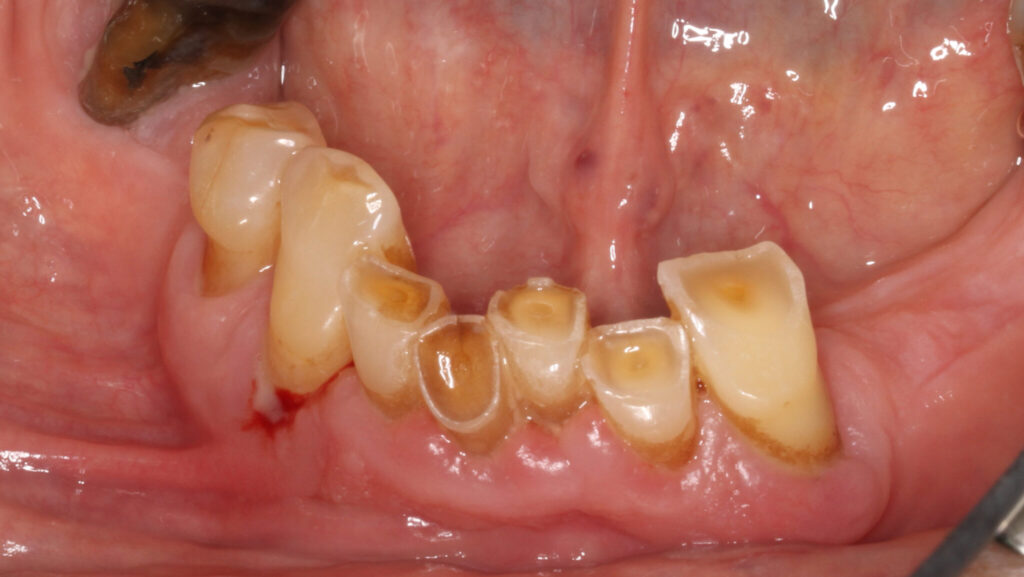
A 65-year-old woman comes to the office with the chief complaint of worn-down teeth, missing teeth, tooth sensitivity, especially on maxillary teeth and TMJ pain. Her past medical history was reviewed and was significant for asthma, insomnia, hypercholesterolemia, hypothyroidism, and anxiety disorder. She denied any sleep disturbances. Her medications included salbutamol, ezetimibe, quetiapine, and levothyroxine. She referred to being allergic to dust and shellfish and sensitive to propofol. She had a right mastectomy in 2014 due to breast cancer. No chemotherapy or radiation was done. Her diet was considered normal, and she denied any consumption of acidic drinks and denied any history of regurgitation. She denied any parafunctional habits during the day. She was using a maxillary transitional partial denture since she had lost some of her teeth due to fracture and caries. No other problems were evidenced. Intraoral exam showed moderate attrition lesions of posterior teeth, porcelain fused to metal crowns on teeth 46 and 47 had the porcelain portion worn down, exposing the metal substructure. Missing teeth included teeth 14,15,16,18,24,28, 36,38,48. Palatal surfaces of maxillary anterior teeth also appeared worn down. An abfraction lesion was found on tooth 23. No carious lesions were found in any of her teeth. Composite restorations on mandibular anterior teeth were chipped or were missing (Figs. 6-11) She also presented with limited mouth opening: 35 mm interincisal distance at maximum opening. There was bilateral pain on palpation of preauricular areas. Her pain was the worst early in the morning which suggested night parafunction. No crepitus, or joint clicking were found.
Fig. 6
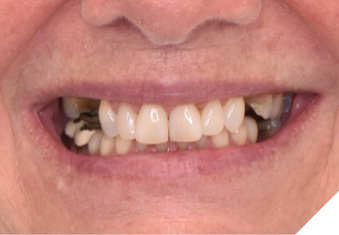
Fig. 7
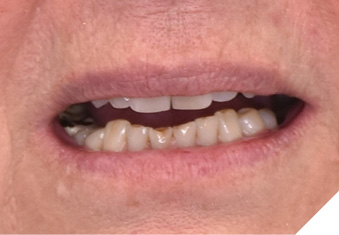
Fig. 8
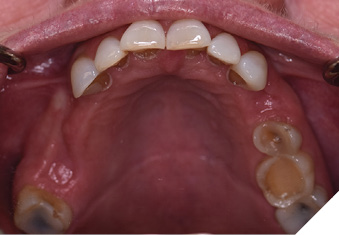
Fig. 9
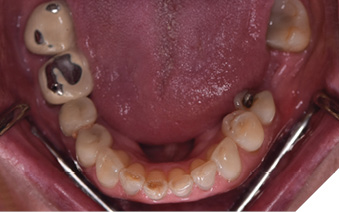
Fig. 10
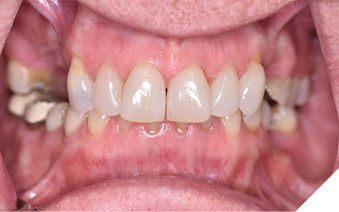
Fig. 11
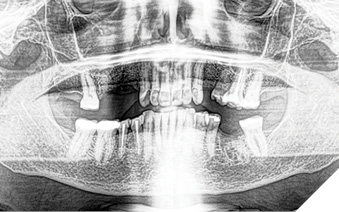
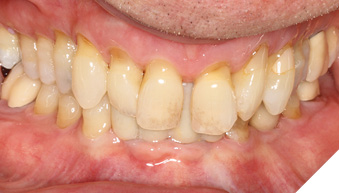
Articulated diagnostic casts showed very limited to no overjet and 90% of overbite. The wear pattern on the palatal aspect of her maxillary teeth appeared to be secondary to her tight occlusion since mandibular anterior teeth were in close contact with these surfaces with no “room” for protrusive movements. The diagnosis for this patient was: partial edentulism, failing existing restorations and attrition and abfraction secondary to parafunction. We discussed different options, including orthodontics in order to increase her overjet so we could have prosthetic space to restore her worn down teeth. We also discussed tooth replacement options. After a second consultation appointment which she attended with her husband, she decided that she did not want to replace her teeth with removable prosthetics. She also declined referral to an orthodontist. After discussion of multiple options of treatment to address all her chief complaints, her treatment consisted of a full mouth reconstruction with full coverage restorations in Zirconia for teeth 11-13,17,22-23,25-27,32-42, 44-47 and partial coverage restorations with lithium disilicate restoration for teeth 33,34 and 43. Replacement of teeth was achieved with a fixed partial denture to replace tooth 36 and a 3-unit implant supported bridge to replace teeth 24-26. In terms of occlusion, we created a more favorable occlusion with a proper anterior guidance, canine disclusion with more freedom for protrusive movements, which hopefully would decrease the parafunction, and alleviate some of her muscle pain. Before completing the case, the patient spent several weeks with provisional restorations which reflected the increase in vertical dimension of occlusion and the new occlusal scheme. During this trial phase, we assessed comfort, esthetics, function, decrease in pain and speech (Figs. 12-17).
Fig. 12
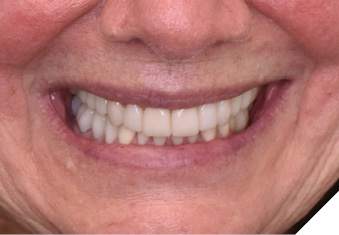
Fig. 13
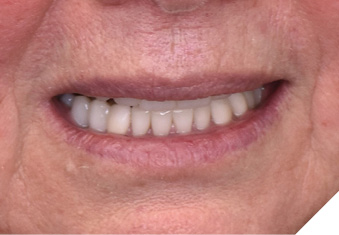
Fig. 14
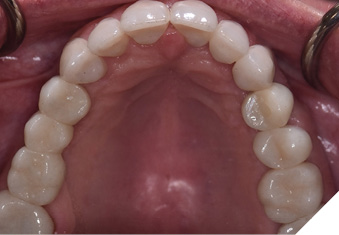
Fig. 15
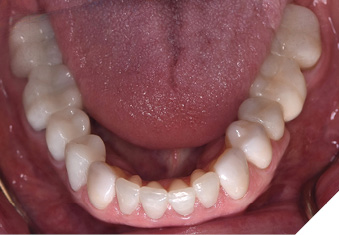
Fig. 16
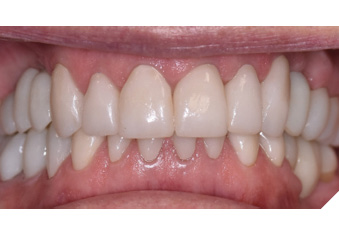
Fig. 17
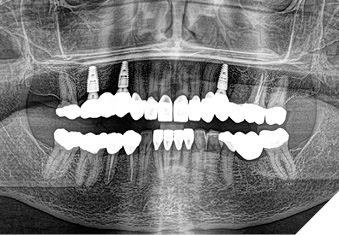
During a 6 month follow up visit, the patient presented with reduced pain in her masticatory muscles and no sensitivity on her teeth. She was also pleased that her chewing function had improved and she loved her brighter smile. She used her night guard every night. She is in a 6-month recall protocol to ensure all restorations are in proper condition and that her occlusion continues to be ideal. We also monitor the health of the soft tissues and peri-implant health.
Conclusion
Tooth wear arises from multiple overlapping causes including abrasion, attrition, erosion, and abfraction, each with distinct yet interconnected mechanisms. Effective dental management requires early recognition, comprehensive risk assessment, preventive behavior change, targeted restorative intervention, occlusal control, and long-term maintenance. Advances in materials science, adhesive dentistry, and diagnostic imaging are refining personalized management strategies. Clinicians should adopt phased, minimally invasive approaches, coordinate with medical and behavioral healthcare providers if possible, and empower patients for sustained improvements in oral health and quality of life. 
Oral Health welcomes this original article.
Disclaimer: The authors have no financial ties, ownership interests, or other affiliations with any companies that make or sell the products or services mentioned in this article.
References
- Bader, J.D. & Shugars, D.A. (2012). Dental wear: I. Prevalence and etiology. Journal of Oral Rehabilitation, 39, 550–563.
- Grippo, J.O., Simring, M., & Schreiner, S. (2012). Abfractions: History, theory, and restoration. Journal of Esthetic and Restorative Dentistry, 24(2), 61–76.
- Lussi, A. & Carvalho, T.S. (2015). Erosive tooth wear: Epidemiology, risk factors and prevention. American Journal of Dentistry, 28(2), 124–133.
- Lussi A, Megert B, Shellis RP. The erosive effect of various drinks, foods, stimulants, medications and mouthwashes on human tooth enamel. Swiss Dent J. 2023 Jul 10;133(7-8):440-455.
- Reid, J., Addy, M., & Newcombe, R. (1999). The erosive potential of cola drinks: An in vivo and in vitro study. Journal of Dentistry, 27, 97–102.
- Zero, D.T. (2000). Reprint of: Etiology of dental erosion – intrinsic factors. European Journal of Oral Sciences, 108, 471–485.
- Eccles, J.D. (1979). Dental Erosion: Aetiology and Clinical Manifestations. Australian Dental Journal, 24(5), 323–329.
- Kuang B, Li D, Lobbezoo F, de Vries R, Hilgevoord A, de Vries N, Huynh N, Lavigne G, Aarab G. Associations between sleep bruxism and other sleep-related disorders in adults: a systematic review. Sleep Med. 2022 Jan;89:31-47.
- Gauer RL, Semidey MJ. Diagnosis and treatment of temporomandibular disorders. Am Fam Physician. 2015 Mar 15;91(6):378-86.
- Vig, R. G., & Brundo, G. C. (1978). The kinetics of anterior tooth display. The Journal of Prosthetic Dentistry, 39(5), 502–504.
- Bartlett, D., Ganss, C., & Lussi, A. (2013). Basic Erosive Wear Examination (BEWE): A new scoring system for scientific and clinical needs. Clinical Oral Investigations, 17, 105–110.
- Delcanho R, Val M, Guarda Nardini L, Manfredini D. Botulinum Toxin for Treating Temporomandibular Disorders: What is the Evidence? J Oral Facial Pain Headache. 2022 Winter;36(1):6-20.
- Dundar A, Sengun A. Dental approach to erosive tooth wear in gastroesophageal reflux disease. Afr Health Sci. 2014 Jun;14(2):481-6.
- Schiff, T., Delgado, E., Zhang, Y.P., Kumar, V., & Qian, F. (2001). Evaluation of dentinal hypersensitivity alleviation in a randomized clinical study. Journal of Clinical Periodontology, 28, 184–191.
- Reynolds, E.C. (2008). Calcium phosphate-based remineralization systems: scientific evidence?. Australian Dental Journal, 53(3), 268–274.
- Soares, C.J., Santos-Filho, P.C., & Consani, R.L. (2013). Long-term survival of composite restorations in worn teeth. American Journal of Dentistry, 26, 58–64.
- Cvar, J.F. & Ryge, G. (1971). Reprint of criteria for the clinical evaluation of dental restorative materials. Clinics in Operative Dentistry.
- Singh BP, Singh N, Jayaraman S, Kirubakaran R, Joseph S, Muthu MS, Jivnani H, Hua F. Occlusal interventions for managing temporomandibular disorders. Cochrane Database Syst Rev. 2024 Sep 16;9(9):CD012850.
- Sheiham, A. (2005). Oral health, general health and quality of life. Bulletin of the World Health Organization, 83(9), 641–720.
- He, Y. & Swain, M.V. (2020). Nanohydroxyapatite versus fluoride toothpaste for enamel remineralisation: a systematic review. Dent Materials, 36(10), 1210–1221.
- Viana, R.B. de, Lussi, A.H. & Tabbakhian, M. (2021). Dentinal fluid biomarkers as predictors of erosive damage – A case-control study. Caries Research, 55, 318–327.
- Malcangi G, et al. Analysis of Dental Enamel Remineralization: A Systematic Review of Technique Comparisons. Bioengineering (Basel). 2023 Apr 12;10(4):472.
- Jeong M, Radomski K, Lopez D, Liu JT, Lee JD, Lee SJ. Materials and Applications of 3D Printing Technology in Dentistry: An Overview. Dent J (Basel). 2023 Dec 19;12(1):1.
- Ahmad P, Hussain A, Carrasco-Labra A, Siqueira WL. Salivary Proteins as Dental Caries Biomarkers: A Systematic Review. Caries Res. 2022;56(4):385-398.
- Chew HP, Zakian CM, Pretty IA, Ellwood RP. Measuring initial enamel erosion with quantitative light-induced fluorescence and optical coherence tomography: an in vitro validation study. Caries Res. 2014;48(3):254-62.
About the authors

Dr. Aragon completed her Master’s degree and specialization in Prosthodontics at the University of Alabama at Birmingham. She served as an Assistant Professor at the Schulich School of Dentistry until 2011, and has since been in full-time private practice in London. Dr. Aragon continues to contribute to academia as an Adjunct Professor at Schulich and lectures on dental implantology to postgraduate residents at the London Health Sciences Centre.

Dr. Leung earned her DDS from the University of Toronto, followed by a General Practice Residency at the Boston Medical Center and a specialty residency in Prosthodontics at the University of Pittsburgh. She is a Fellow of the Royal College of Dentists of Canada and a Diplomate of the American Board of Prosthodontics.