
A five-year NSERC (Natural Sciences and Engineering Research Council of Canada) Discovery Grant to support a transformative research program has been awarded to Dr. Zeeshan Sheikh at Dalhousie University with the aim to develop 3D-printed titanium constructs coated with calcium phosphate for craniofacial reconstruction. While housed at Dalhousie University, this program is designed to attract and foster collaboration with local and international researchers and institutions to refine material formulations and develop broadly applicable implant solutions for various surgical challenges. This research program is not a singular project; it is a comprehensive and integrated initiative that addresses fundamental challenges in biomaterials design, additive manufacturing, and reconstructive surgery. The program leverages Dalhousie’s state-of-the-art infrastructure and interdisciplinary expertise to develop bioactive, customizable, and resorbable titanium implants, ultimately transforming patient care in craniofacial reconstruction.
The clinical challenge: Maxillectomy and reconstructive limitations
Maxillectomy, often necessitated by the treatment of oral and maxillofacial cancers, results in the partial or complete resection of the maxilla.1 This procedure leaves patients with profound anatomical and functional deficits, affecting their speech, mastication, esthetics, and psychological well-being. Restoration of these defects remains one of the most formidable challenges in maxillofacial surgery.2 Conventional reconstruction methods, such as the use of vascularized fibula free flaps, offer some degree of anatomical and functional restoration. However, these procedures are lengthy, typically taking 10-13 hours, and come with significant limitations: donor site morbidity, limited graft availability, and unpredictable long-term outcomes.3 Additionally, current reconstructions often require multiple surgeries and result in less-than-optimal integration and esthetics.
This pressing clinical need calls for the development of alternative biomaterial-based solutions that provide structural support, promote osseointegration, and are tailored to patient-specific anatomical needs. The proposed research program addresses this gap by integrating 3D printing technology with bioactive calcium phosphate coatings, creating patient-specific implants that can potentially replace or augment autologous grafts.
Advanced manufacturing: 3D printing and titanium for craniofacial applications
Titanium is the gold standard in biomedical implants due to its excellent strength-to-weight ratio, corrosion resistance, and inherent osteoconductive properties.4 The alloy Ti-6Al-4V (Ti64) is one of the most used in dental and orthopedic applications as it spontaneously forms a stable oxide layer, further enhancing its biocompatibility and bone affinity.5 Traditional fabrication methods, such as milling or forging, fall short when it comes to creating complex geometries required for patient-specific implants. In contrast, additive manufacturing, particularly Laser Powder Bed Fusion (LPBF)—enables precise control over the internal and external architecture of titanium constructs.6 LPBF builds objects layer by layer, allowing the creation of intricate lattice structures with optimized surface roughness and porosity, which are critical for successful tissue integration.7 Customized titanium plates and meshes are already used in clinical practice, especially to secure fibula flaps in mandibular and maxillary reconstructions.8 However, most of these constructs are solid, dense, and lack internal architecture conducive to tissue infiltration. The use of 3D printing opens the possibility of creating hollow, porous structures that encourage bone ingrowth and reduce implant weight without compromising mechanical integrity.
The role of calcium phosphate coatings in enhancing integration
Despite titanium’s benefits, challenges remain in achieving seamless integration with host bone, especially in complex reconstructive cases. Coating titanium with calcium phosphate materials addresses this limitation by combining mechanical strength with biological functionality. Dicalcium phosphate biomaterials, particularly brushite (CaHPO4·2H2O) and monetite (CaHPO4), are highly bioactive and resorbable.9 Brushite sets rapidly as a hydraulic cement and provides a favorable environment for new bone formation, whereas, monetite, the anhydrous form of brushite, resorbs more quickly and does not convert to hydroxyapatite (HA), enabling better remodeling dynamics.10,11 These coatings can be engineered to control resorption rates, structural integrity, and ionic exchange with the surrounding biological environment.9,10
The challenge lies in applying these coatings uniformly onto complex 3D-printed titanium structures. Traditional coating methods, such as plasma spraying or electrodeposition, are often insufficient for intricate lattice geometries due to poor adhesion, uneven distribution, or coating delamination. The proposed research program seeks to overcome these limitations through innovative application methods, including vibrational infiltration of cement slurries and hydrothermal processing for phase conversion.
Preliminary work and research objectives
To lay the groundwork for this ambitious program, preliminary work was initiated under the joint supervision of Drs. Zeeshan Sheikh, Paul Bishop and Pierre-Luc Michaud at Dalhousie University. A co-op, an undergraduate and a graduate student were engaged to optimize Ti64 powder processing using LPBF. The team successfully printed simple and complex geometries, including cuboids and lattices, with consistent surface roughness and dimensional accuracy. Key achievements included: characterization of Ti64 powder morphology and flow properties, optimization of LPBF parameters (e.g., laser power, scan speed, hatch spacing) for high-resolution printing, printing of complex lattice geometries with consistent porosity and strut thickness, and confirmation of geometric fidelity and resolution down to 0.1 mm. These results provide a strong foundation for subsequent coating and biological evaluation, positioning the team to explore the next phase of research with confidence.
The overarching goal of the research program is to create patient-specific, 3D-printed titanium implants of varying geometries with bioresorbable calcium phosphate coatings for craniofacial applications (Fig. 1). These implants will address current limitations in reconstructive surgery and offer a viable alternative to autologous grafts.
Fig. 1
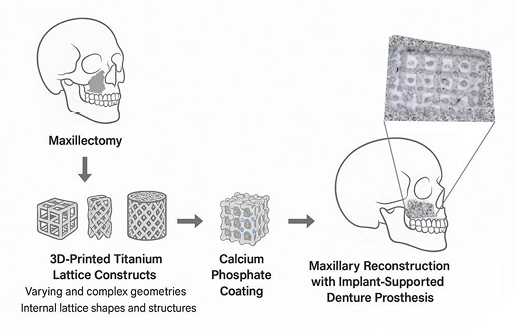
To achieve this, the program will be run via two research streams with the main aim to produce mechanically robust and biologically active implants tailored for craniofacial reconstruction.
Stream 1: Titanium Construct Design and Fabrication
- Characterize and optimize Ti64 powders for LPB.
- Establish LPBF process parameters to fabricate porous scaffolds with controlled surface topography (1–2 µm roughness).
- Design and print grid and octagon lattice geometries with strut thicknesses of 150 µm and varying porosities (Fig. 2).
- Perform mechanical testing and surface characterization.
Fig. 2

Stream 2: Calcium Phosphate Coating Development and Evaluation
- Formulate brushite and monetite cement pastes with varying powder-to-liquid ratios to achieve different viscosities.
- Coat printed titanium scaffolds using vibrational slurry infiltration with brushite and monetite (Fig. 2).
- Evaluate coating integrity, adhesion strength, ionic release, and degradation profiles in vitro.
Methodology and experimental approach
The research program will employ several highly sensitive and unique research methodologies to achieve Powder Characterization and Printing, Lattice Design and Evaluation, Brushite and Monetite Coating Applications, and Coating integrity Evaluations. Plasma-atomized Ti64 powders will be analyzed for particle size, density, and flow characteristics per American Society for Testing and Materials (ASTM) standards. Scanning Electron Microscopy (SEM), Inductively Coupled Plasma Optical Emission Spectroscopy (ICPOES), and O-N-H analysis will confirm morphology and chemical composition. Using the AconityMINI LPBF printer housed at Department of Mechanical Engineering at Dalhousie University, various thin walls and cuboids will be fabricated under different energy density conditions. Mechanical and microstructural properties will be assessed through tensile testing, SEM, and Transmission Electron Microscopy (TEM) imaging. Grid and octagon lattices will be printed using optimized parameters and their dimensions will be verified using micro-CT and confocal microscopy (Fig. 2). Surface roughness will be measured using Atomic Force Microscopy (AFM), and compressive strength assessed with an Instron machine.
Cement slurries of different viscosities will be prepared using β-TCP and monocalcium phosphate. Coatings will be applied using polyvinylsiloxane molds and vibrational infiltration and SEM and TEM will be employed for surface morphology and thickness measurements. X-ray Diffraction (XRD) and Fourier Transform Infrared Spectroscopy (FTIR) will be used to determine phase composition and crystallinity, along with AFM and profilometry for roughness measurement. Tensile adhesion testing and nanoindentation for mechanical properties will be carried out and ion release studies using Inductively Coupled Plasma mass spectrometry (ICP). In addition, this will be followed by in vitro degradation testing in Tris-HCl buffer to evaluate coating stability and dissolution.
Research team and institutional resources
Principal Investigator: Dr. Zeeshan Sheikh, a clinician-scientist with expertise in calcium phosphate biomaterials and titanium implant design, Dr. Sheikh leads the coating development and material characterization aspects of the program.
Collaborator: Dr. Paul Bishop, a Professor in Mechanical Engineering, Dr. Bishop brings critical expertise in metal additive manufacturing, particularly LPBF of titanium alloys. His lab will focus on the printing and structural evaluation of titanium constructs.
Collaborator: Dr. Pierre-Luc Michaud, an experienced maxillofacial prosthodontist, Dr. Michaud will guide scaffold design and clinical translation. His experience with facial prostheses will ensure anatomical accuracy and patient relevance.
Highly Qualified Personnel (HQP) Trainees: The research program provides an exceptional training environment for developing highly qualified personnel (HQP) across multiple disciplines, including dentistry, biomedical engineering, and medical sciences. The team currently includes several talented trainees who are contributing to the ongoing development and optimization of 3D-printed titanium constructs, coated with calcium phosphate for craniofacial reconstruction. Our current trainees include:
Dr.Tiffany Kisway – a master’s student in the Surgical Periodontics program at Dalhousie University, working on translational aspects of biomaterials integration and clinical applications.
Lauren MacRae – an experiential learning student gaining hands-on experience in biomaterials characterization and surface modification techniques.
Ella Lawrence – an Honors thesis and NSERC-USRA research student investigating polymeric membrane development and contributing to the project’s scaffold optimization studies.
Additional volunteers from the Medical Sciences program and Mechanical Engineering at Dalhousie University, who are involved in imaging, mechanical testing, and data analysis, contribute to the multidisciplinary nature of the project. Building on the NSERC Discovery Grant funding, we plan to expand our HQP cohort in the near future by recruiting additional MSc and PhD students who will focus on biomaterials synthesis, surface modification strategies, in-vitro characterization, and translational research. These trainees will benefit from Dalhousie’s state-of-the-art facilities, access to industry partnerships, and exposure to clinical research integration, preparing them to become future leaders in biomaterials research and regenerative medicine.
Institutional Infrastructure: Dalhousie University offers advanced facilities in 3D printing, microscopy, spectroscopy, and mechanical testing. The team also plans to leverage commercialization support from Springboard Atlantic, a regional network that connects researchers with industry partners, facilitates technology transfer, and supports intellectual property development across Atlantic Canada. In addition, Innovacorp, Nova Scotia’s early-stage venture capital organization, can help provide expertise and resources to help accelerate the commercialization of research outcomes, support startup development, and foster industry collaborations. These partnerships are expected to play a pivotal role in translating the team’s research on 3D-printed titanium constructs coated with calcium phosphates into clinically viable solutions, enabling a smoother pathway from lab-based innovation to real-world craniofacial reconstruction applications.
Broader impact and long-term vision
This program is the first of its kind in Canada to combine titanium additive manufacturing with dicalcium phosphate coatings for craniofacial reconstruction. This NSERC-funded research program holds transformative potential for personalized bone repair and reconstructive surgery in Canada and globally. The innovative aspects of this research program include:
- Personalized implant design using patient imaging data
- Bioactive coatings that promote rapid bone integration
- Scalable manufacturing protocols for broader clinical use
The outcomes will impact clinical practice by reducing surgical times, improving healing outcomes, and minimizing complications. Additionally, this research will generate intellectual property and position the team for follow-up in vivo studies, eventually leading to clinical trials. The program provides a rich training ground for highly qualified personnel (HQP) in biomedical engineering, biomaterials science, and surgical innovation, equipping them for future careers in academia, industry, and healthcare.
The long-term objective is to translate these findings into clinical settings. Following in vitro and preclinical validation, the team aims to initiate animal studies and, ultimately, human clinical trials. These implants could serve not only maxillectomy patients but also individuals requiring cranial, mandibular, and orthopedic reconstruction. Future research studies will explore:
- Incorporation of antimicrobial or drug-releasing functionalities.
- Optimization of implant resorption rates.
- Collaboration with industry for device certification and manufacturing scale-up.
This comprehensive approach aligns directly with NSERC’s mandate to advance discovery, train the next generation of researchers, and enhance Canada’s global competitiveness in science and innovation. By cultivating a pipeline of highly qualified personnel, this program will position Dalhousie University as a national leader in biomaterials research, fostering partnerships with industry, health care, and academic collaborators to translate laboratory discoveries into real-world clinical solutions. 
Oral Health welcomes this original article.
Disclosure: This research is supported by the following grants awarded to Dr. Zeeshan Sheikh at Dalhousie University: 1. The Natural Sciences and Engineering Research Council of Canada (NSERC) through a Discovery Grant (2025-2031). 2. Faculty of Dentistry Research Fund, Dalhousie University (2025). 3. NCOHR New Frontiers Seed Grant Program (2024-2025)
References
- Brown, J.S., et al., A modified classification for the maxillectomy defect. Head & Neck: Journal for the Sciences and Specialties of the Head and Neck, 2000. 22(1): p. 17-26.
- Joseph, S.T., et al., Challenges in the reconstruction of bilateral maxillectomy defects. Journal of Oral and Maxillofacial Surgery, 2015. 73(2): p. 349-356.
- Peng, X., et al., Maxillary reconstruction with the free fibula flap. Plastic and reconstructive surgery, 2005. 115(6): p. 1562-1569.
- Khorasani, A.M., et al., Titanium in biomedical applications—properties and fabrication: a review. Journal of biomaterials and tissue engineering, 2015. 5(8): p. 593-619.
- Albrektsson, T. and C. Johansson, Osteoinduction, osteoconduction and osseointegration. European Spine Journal, 2001. 10(2): p. S96-S101.
- Lv, Y., et al., Metal material, properties and design methods of porous biomedical scaffolds for additive manufacturing: a review. Frontiers in Bioengineering and Biotechnology, 2021. 9: p. 641130.
- Sun, S., M. Brandt, and M. Easton, Powder bed fusion processes: An overview. Laser additive manufacturing, 2017: p. 55-77.
- Oh, J.-h., Recent advances in the reconstruction of cranio-maxillofacial defects using computer-aided design/computer-aided manufacturing. Maxillofacial plastic and reconstructive surgery, 2018. 40(1): p. 2.
- Tamimi, F., Z. Sheikh, and J. Barralet, Dicalcium phosphate cements: brushite and monetite. Acta Biomater, 2012. 8(2): p. 474-87.
- Sheikh, Z., et al., Effect of processing conditions of dicalcium phosphate cements on graft resorption and bone formation. Acta biomaterialia, 2017. 53: p. 526-535.
- Tamimi, F., et al., The effect of autoclaving on the physical and biological properties of dicalcium phosphate dihydrate bioceramics: brushite vs. monetite. Acta Biomater, 2012. 8(8): p. 3161-9.
About the author

Dr. Zeeshan Sheikh was trained as a dental clinician, periodontist, and biomaterials scientist at Baqai University, Queen Mary University of London, McGill University, University of Toronto, Mt. Sinai Hospital and Dalhousie University. Dr. Sheikh has published more than 70 original research articles, reviews, clinical case reports and book chapters. He is a Clinical Scientist in Periodontics and an Assistant Professor at Dalhousie University and has a private periodontal practice in Halifax.
