Dental implants have become a widely accepted solution for restoring both function and esthetics in partially or fully edentulous patients, through the use of either fixed or removable prostheses. Systematic reviews have consistently demonstrated high survival rates for both implants and prosthetic components, with an overall success rate of approximately 94.6%.1,2
However, peri-implant diseases remain a significant complication associated with implants and the associated prostheses. These inflammatory conditions affect the surrounding soft and hard tissues, leading to potential implant or prosthesis failure. Initial reports estimated the prevalence of peri-implantitis to be approximately 10% at the implant level and 20% at the patient level within 5 to 10 years post-implant placement.3
In recent years, systematic reviews and meta-analyses have provided further insights into the incidence of peri-implant disease. A comprehensive review revealed that the prevalence of peri-implantitis up to 16% among patients without a history of periodontitis, and increased up to 66.7% in patients with a prior history of periodontitis.4 Recently peri-implantitis prevalence was reported to be 19.53% at the patient level and 12.53% at the implant level.5
Given the substantial prevalence of peri-implant diseases, it is imperative to ensure continuous clinical monitoring of the soft and hard tissues surrounding implants and their prostheses. This paper will provide a comprehensive overview of the etiology, prevention, and treatment strategies (Fig. 1) for peri-implant diseases that compromise the long-term health of implants and associated prostheses.
Fig. 1
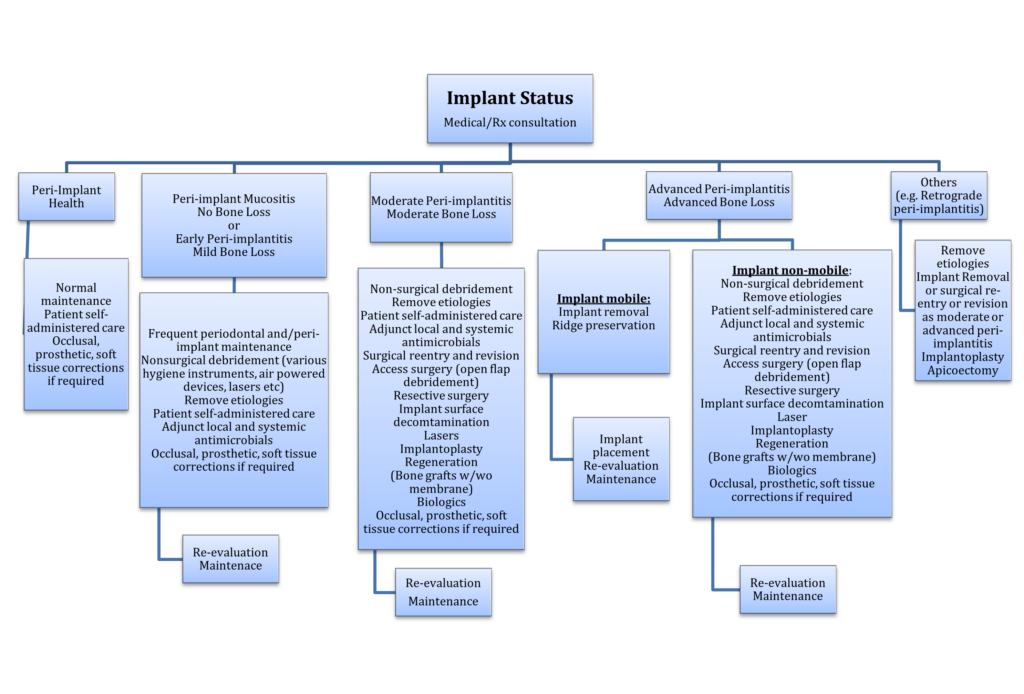
Preventive strategies for peri-implant diseases
A comprehensive maintenance regimen coupled with optimal oral hygiene is critical for ensuring the health and longevity of dental implants. Mechanically controlling plaque with the use of patient-administered hygiene tools—such as manual or power toothbrushes, interdental brushes, dental floss, and irrigation devices—combined with chemical control via antimicrobial mouth rinses and dentifrices, have been demonstrated to be an effective strategy in preventing peri-implant diseases.6,7
Evaluation of medical and medication history in patients
According to the guidelines issued by the American Academy of Oral Medicine, it is recommended that patients and clinicians conduct a well-documented review of the patient’s medical and medication history at least annually.8 Moreover, it is advised that any changes in the patient’s medical and medication history be reviewed at each dental appointment. This practice is essential for clinicians to identify any alterations in the patient’s health status that could affect the current or future health of dental implants and related oral health.
Systemic conditions, including uncontrolled diabetes mellitus, osteoporosis, autoimmune and connective tissue disorders, as well as depression, along with social behaviors such as smoking, vaping, and alcohol consumption, have been increasingly recognized as significant factors that may adversely affect implant health.9,10
Additionally, medications, including corticosteroids, oral and intravenous bisphosphonates, biologics, monoclonal antibodies, antidepressants, proton pump inhibitors, organ transplant anti-rejection drugs, and antineoplastic agents, have the potential to exert pharmacological effects that could impact implant health.9,10
Maintenance frequency:
The frequency of maintenance appointments should be tailored to the patient’s systemic, local, and environmental risk factors. According to the 2017 World Workshop on the Classification of Periodontal and Peri-Implant Diseases and Conditions11, patients with a history of chronic periodontitis, poor plaque control skills, and who do not adhere to regular maintenance regimens following implant therapy are at a higher risk of developing peri-implant diseases.11 Furthermore, additional evidence suggests that factors such as residual cement, insufficient keratinized mucosa, and suboptimal implant positioning may impede effective oral hygiene and professional maintenance.11
Retrospective longitudinal studies12 demonstrated that patients that adhered to regular maintenance therapy exhibited an 18% incidence of peri-implantitis compared to those without maintenance, who showed a 44% incidence. Routine maintenance protocols have been shown to effectively reduce plaque accumulation and bleeding indices over time, thereby controlling plaque buildup and preventing clinical attachment loss.13 Optimal home care combined with routine maintenance visits are essential for the ongoing evaluation and preservation of implant health.
The 11th European Workshop on Periodontology (2014),14 which focused on the topic of “Effective prevention of periodontal and peri-implant diseases”, concluded that professional supportive care should be customized to the individual needs of each patient. The recall intervals should be dependent on patient-specific factors, usually with a time frame of 3, 6, or 12 months. Ensuring patient compliance with these maintenance schedules is critical for the primary prevention of peri-implant diseases.14
Peri-Implant disease classification
According to the 2017 World Workshop on the Classification of Periodontal and Peri-Implant Diseases and Conditions,15 “peri-implant health” is characterized by the absence of visual signs of inflammation and bleeding upon probing. Implants are considered “healthy” when these clinical signs are not present. Additionally, peri-implant health can be maintained around implants with both normal and reduced bone support.15 However, to maintain implant health, it is essential to conduct thorough periodontal and peri-implant examinations during routine maintenance visits.
During maintenance visits, a thorough review and reinforcement of the patient’s self-administered oral hygiene practices must be completed. It is also necessary to confirm that the patient has access to the appropriate homecare products for effective oral hygiene around implant prostheses. Given that occlusal overload is a contributing factor for peri-implant diseases, an occlusal analysis should be routinely performed and adjustments made accordingly.16 In certain cases, modifications or adjustments to the prosthesis may be required to enhance cleansability and prevent accumulation of biofilm.17,18
The amount and health of keratinized tissue around the implant prosthesis is recognized to protect implants from mechanical and bacterial challenges, enhancing oral hygiene, and ensuring the long-term success and health of the implant. In cases of soft tissue compromise, soft tissue augmentation should be completed, as soft and hard tissue conditions may remodel in response to functional loading over time.
Peri-implant mucositis/no bone loss
Peri-implant mucositis is characterized by inflammation of the soft tissues surrounding a dental implant in the absence of supporting bone loss.19 Peri-implant mucositis is widely regarded as a precursor to peri-implantitis. The primary etiological factors contributing to peri-implant mucositis include the presence of biofilm, calculus, food debris, foreign bodies, and residual cement,20,21 which disrupt the host-microbe homeostasis at the implant-mucosa interface. Fortunately, peri-implant mucositis is a reversible condition when these etiological factors are removed, and oral hygiene is appropriately managed.
The etiology of peri-implant diseases is multifactorial, involving microbial, host-related, and environmental factors.22 Microbial biofilm and foreign bodies, such as cement, floss remnants, and food debris on implant surfaces, may initiate a localized immune response, leading to inflammation and, subsequently, affecting both soft and hard tissues around dental implants. The removal of these etiological factors is critical to reducing infection and inflammation.23,24,25
The management of peri-implant mucositis involves non-surgical debridement using selected curettes, elimination of causative factors, patient-administered oral hygiene care, adjunctive use of local chemotherapeutics, systemic antibiotics, soft tissue correction, prosthetic adjustments, and occlusal adjustments as needed during routine maintenance visits.26
Various hygiene instruments are recommended for debriding implants, including metallic curettes (e.g., titanium), non-metallic curettes (e.g., carbon, plastic, resin), rubber cups, titanium brushes, air-powered abrasive instruments, ultrasonic scalers with plastic tips, and lasers.27,28,29
Air-powered devices utilizing abrasive powders, such as erythritol, sodium bicarbonate, sodium hydrocarbonate, or amino acid glycine, have demonstrated effectiveness in biofilm and stain removal. More recent studies have employed erythritol or glycine powder for the decontamination of implant surfaces and prostheses.30,31,32
The pathogenesis and clinical manifestations of peri-implant diseases resemble periodontal diseases. Therefore, the management of peri-implant diseases follows periodontal principles.33 Due to the multiple risk factors, including the patient’s systemic health, social history, and the complexity of biofilm, adjunctive treatment regimens have been proposed to enhance the management of both periodontal and peri-implant diseases. Complete elimination of biofilm is challenging with mechanical debridement alone, particularly in the presence of complex systemic risk factors. Consequently, chemotherapeutic approaches, whether local or systemic, have been proposed as additional strategies for managing periodontal and peri-implant diseases.34,35
Submucosal debridement combined with adjunctive local delivery of antibiotics, submucosal glycine powder air polishing, or Er laser treatment may reduce the clinical signs of peri-implant mucosal inflammation more effectively than submucosal debridement using curettes with adjunctive irrigation with chlorhexidine.36
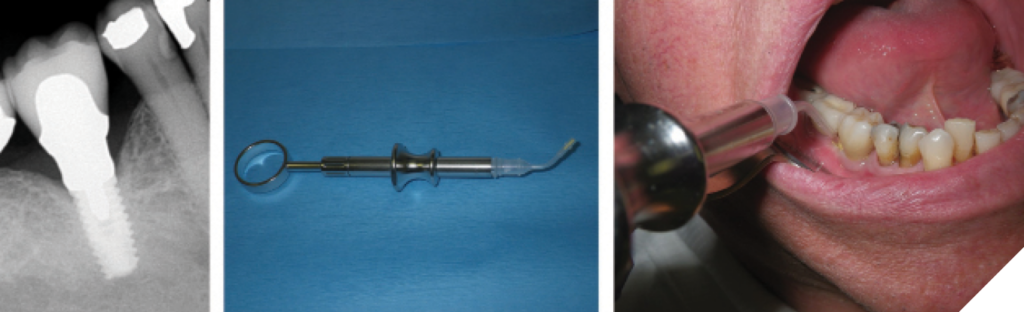
Early peri-implantitis/mild bone loss
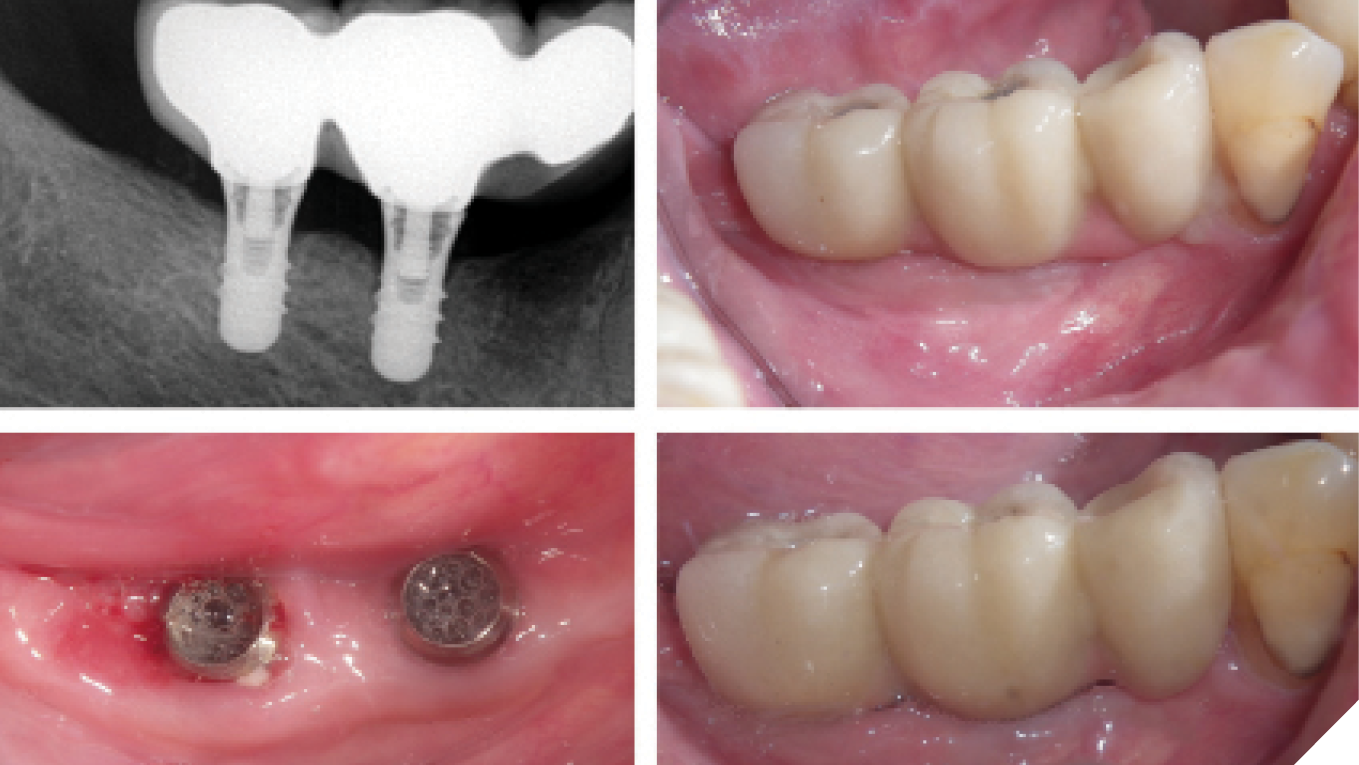
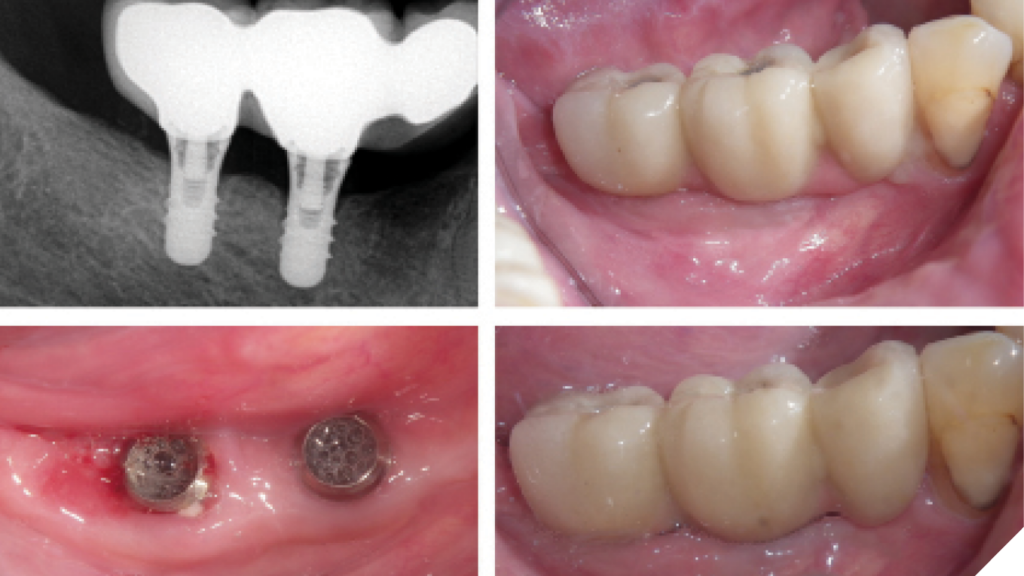
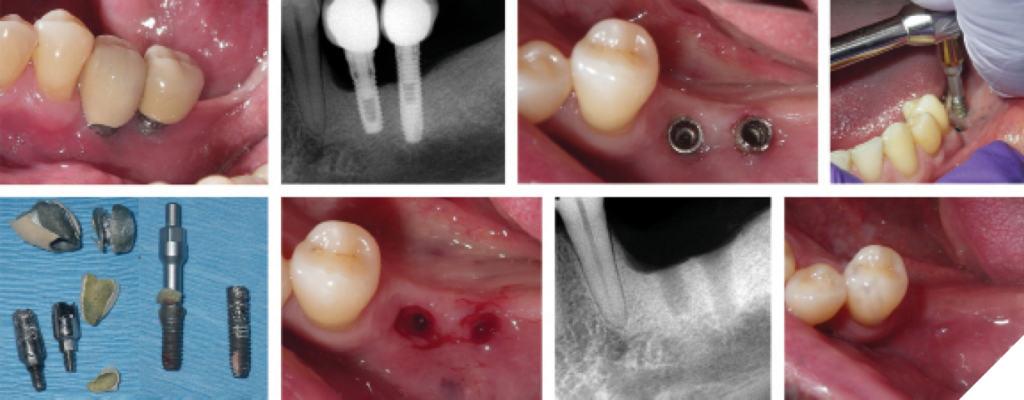
Early peri-implantitis is associated with mild bone loss, limited to the coronal portion of the implant. The management of early peri-implantitis follows a similar approach to that of peri-implant mucositis. It involves a comprehensive review of the patient’s current medical and medication history, non-surgical debridement using selected curettes, elimination of etiological factors, reinforcement of patient-administered oral hygiene care, and the application of adjunctive local chemotherapeutics or systemic antibiotics as needed.26 Additionally, interventions may include soft tissue correction, prosthetic adjustments, and/or occlusal modifications to ensure optimal implant health (Figs. 2&3).26
Fig. 2
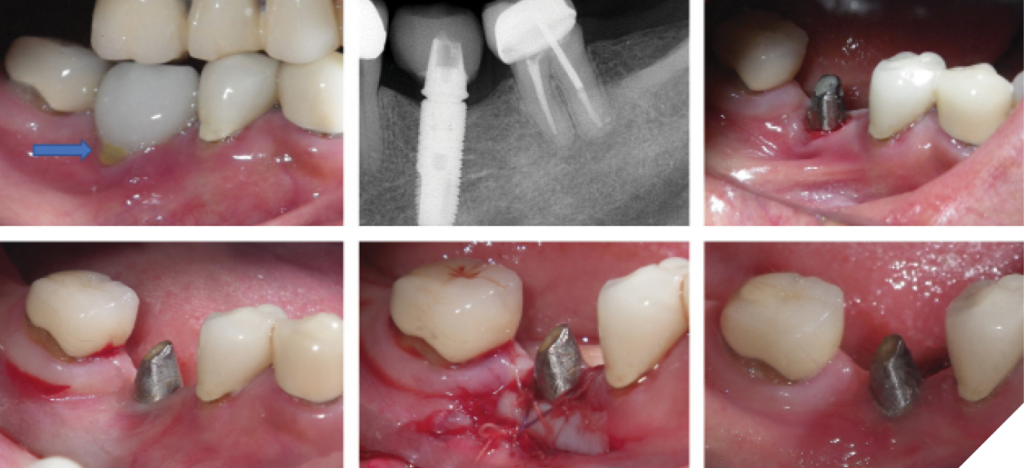
Fig. 3
Moderate peri-implantitis/moderate bone loss
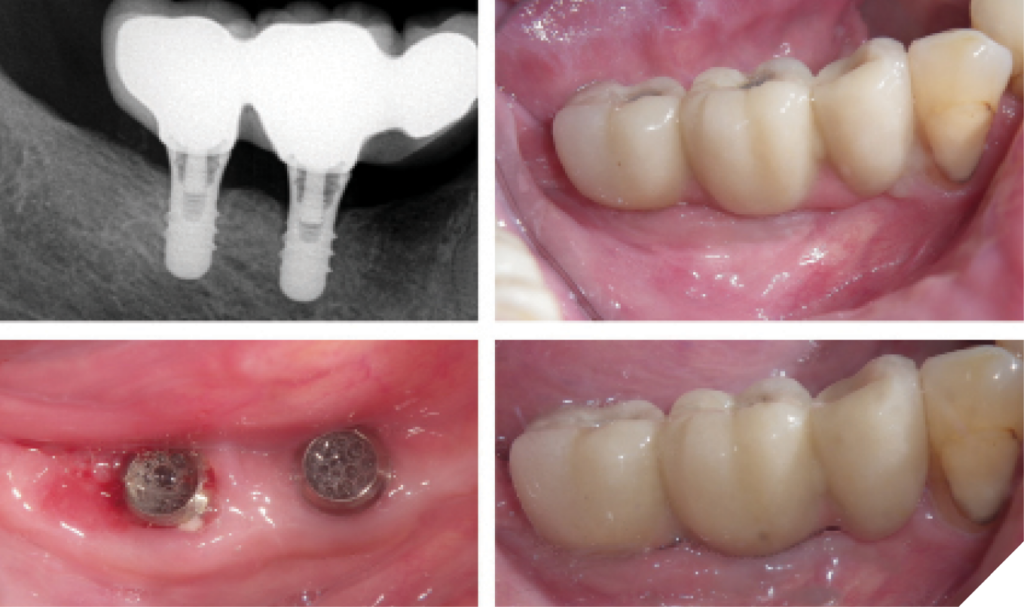
Moderate peri-implantitis is associated with significant bone loss, extending beyond the coronal portion of the implant, however not involving greater than 50% length of the implant. The management of moderate peri-implantitis includes non-surgical therapies, such as debridement and the removal of etiological factors, alongside patient-administered oral care and the use of adjunctive local and systemic chemotherapeutics to reduce infection and inflammation. However, non-surgical therapy yields less predictable outcomes due to the challenges associated with accessing exposed implant surfaces for effective decontamination.37,38,39,40
The primary goal of surgical intervention in moderate peri-implantitis is to gain access to contaminated implant surfaces and osseous defects. Various surgical approaches have been documented, including access surgery (open flap debridement), resective procedures with or without modification of implant surfaces (implantoplasty), and regenerative techniques (Figs. 4&5).41,42,43
Fig. 4
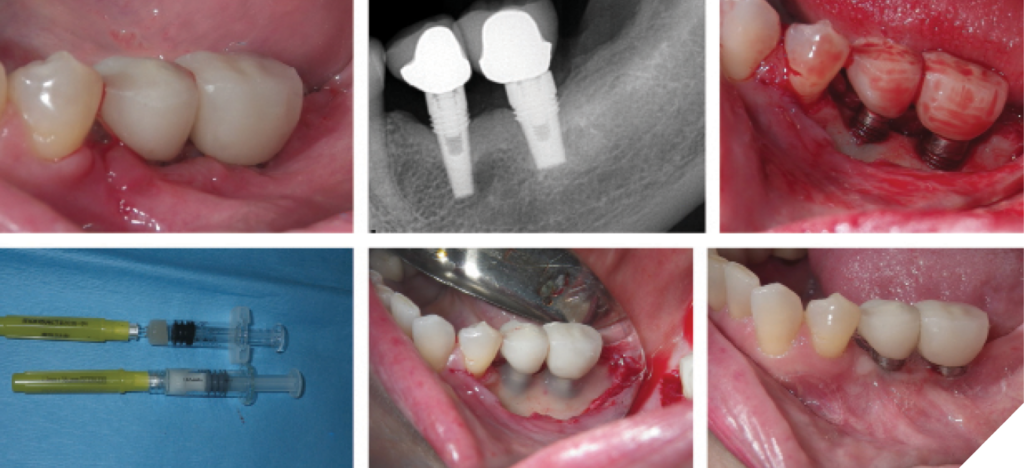
Fig. 5
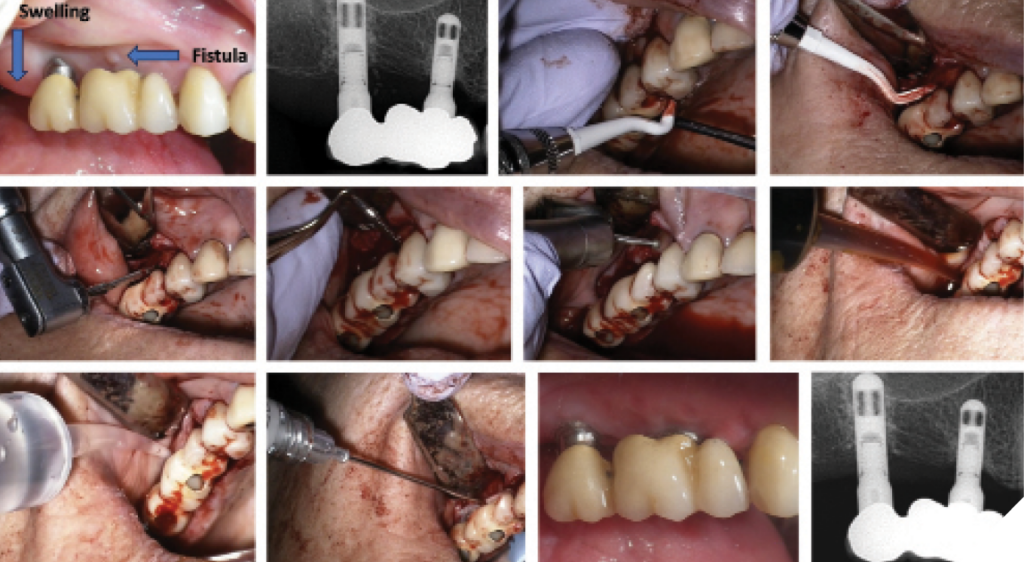
Access Flap Surgery: Also known as “open flap debridement”, this procedure involves elevating periodontal flaps to directly visualize and decontaminate the implant surface.44 Mechanical debridement is performed using selected curettes, and additional decontamination may involve adjunctive chemotherapeutic agents, such as chlorhexidine, iodine, hydrogen peroxide, doxycycline, sterile saline, and others, or the use of lasers.43 Adjunctive use of systemic antibiotics following surgical treatment of peri-implantitis can positively impact treatment outcomes, including reduced probing depth, decreased bleeding on probing/suppuration, and minimal bone loss.45
Resective Surgical Approaches: These procedures involve bone recontouring and the apical positioning of the mucoperiosteal flap, which reduces probing depths around infected implants and improves access for homecare.42,43 Additionally, implantoplasty—modifying the exposed implant threads to reduce surface roughness—may be performed to make the surface less conducive to plaque retention. This process typically involves the use of diamond burs or carbide bone cutters, followed by polishing with Arkansas stones and silicone polishers.46 Systematic reviews have indicated that implantoplasty does not result in significant mechanical or biological complications in the short to medium term.47
Regenerative Surgical Approaches: These techniques focus on regenerating the infrabony defect and attempting to achieve reosseointegration around exposed implant threads.48,49 The consensus from the 15th European Workshop on Periodontology49 recommends augmentation procedures for defects with a minimum depth of 3 mm and 3- to 4-wall contained defects (craters). Augmentation can involve the use of membranes alone, bone grafts with or without membranes, biologics, and lasers, depending on the defect’s type and depth.49 Bone graft options include autogenous grafts, allografts, xenografts, and alloplastic materials, depending on patient and clinician preferences.50 Comparative studies have shown that xenografts may provide better clinical and radiographic outcomes than autogenous bone or alloplastic particles due to slower resorption rates.51
Use of Biologics: Biologic agents, such as enamel matrix derivative (EMD, Amelogenin), platelet-derived growth factors (GEM 21), platelet-rich fibrin (PRF), plasma rich in growth factors (PRGF), and bone morphogenic proteins (BMP), have been proposed as adjuncts in the management of peri-implant diseases. These biologics contain and stimulate various bioactive molecules, promoting cell proliferation, differentiation, and tissue regeneration. A systematic review suggests that the use of biologics may improve clinical outcomes in peri-implant mucositis, their efficacy in peri-implantitis management remains inconclusive based on available evidence.52
For surgical management of peri-implant diseases, it may be necessary to remove the implant prosthesis to achieve proper access and ensure primary closure for subgingival healing. However, studies indicate no significant differences in treatment response whether the prosthesis is removed or retained after regeneration surgery.53 Surgical procedures may also lead to soft tissue recession, necessitating additional soft tissue augmentation.
Advanced peri-implantitis/advanced bone loss
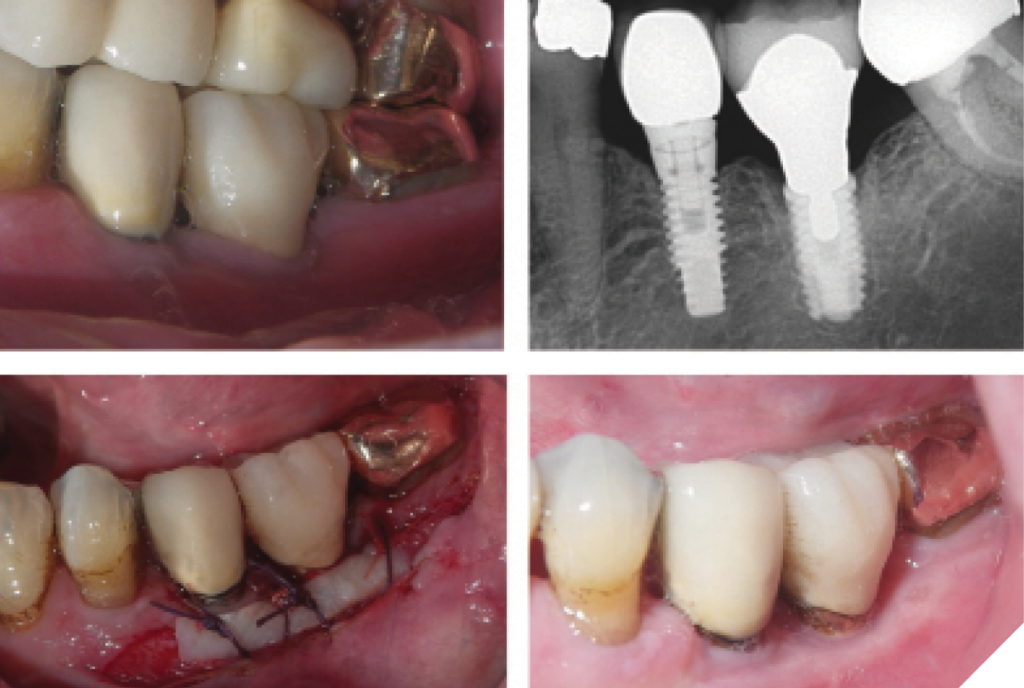
Advanced peri-implantitis is associated with extensive bone loss, often involving greater than 50% bone loss of the implant. In cases of advanced peri-implantitis, where the implant remains stable (i.e., non-mobile), treatment may still be viable. The treatment protocol is similar to that for moderate peri-implantitis, beginning with a thorough review of the patient’s medical and medication history. Initial management involves non-surgical therapies, including debridement, removal of etiological factors, patient-administered care, and adjunctive local and systemic chemotherapeutics. Surgical therapy follows, encompassing resective procedures with or without implantoplasty and/or regenerative approaches. Mucogingival and prosthetic assessments and corrections may also be required. (Figs. 6&7)
Fig. 6
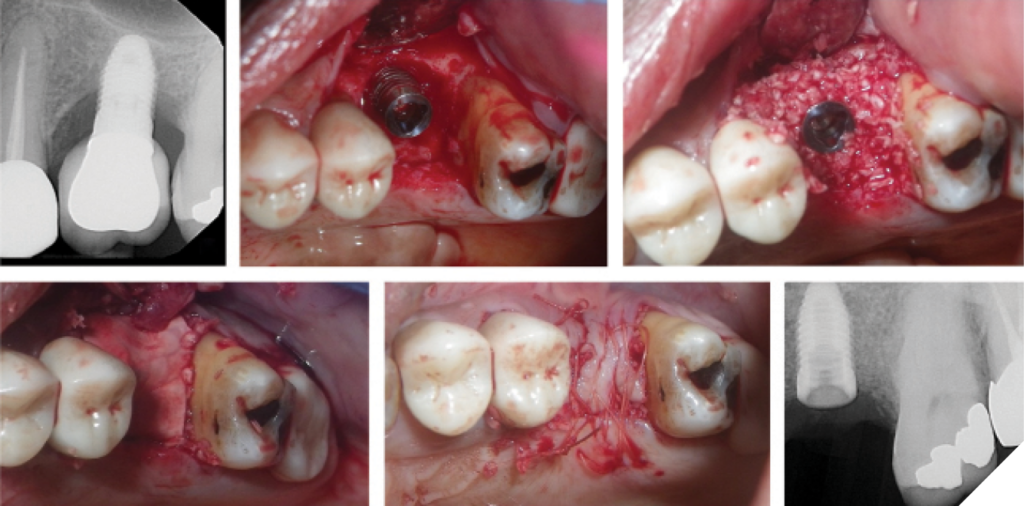
Fig. 7
If the implant is fractured, mobile, or cannot be maintained after surgical resective or regenerative therapy, implant removal (explantation) is recommended. Various methods for implant removal include reverse torque, elevators, forceps, trephines, burs, piezosurgery, laser-assisted explantation, or combinations therapies.54,55 Post-explantation management of the alveolar ridge may involve spontaneous healing or guided bone regeneration for ridge augmentation.
Retrograde peri-implantitis
Retrograde peri-implantitis is a specific type of peri-implant lesion that occurs at the apex of a dental implant. This condition is characterized by localized bone loss at the apex of the implant, often in the absence of peri-implant disease around the coronal part of the implant.
If the retrograde peri-implantitis lesions are asymptomatic, they may not require immediate intervention unless clinical signs and symptoms develop. Therapeutic options include implant removal, surgical re-entry, or revision as in moderate or advanced peri-implantitis, or apical resection, which may involve apicoectomy of adjacent endodontically involved teeth or removal of the implant apex.56
Adjunctive peri-implantitis treatments
Lasers for Peri-implantitis Rescue therapy: Recent progress for use of lasers to rescue failing dental implants has been recognized. A systematic review57 reports NdYAG (1064 nM wavelength) supports regeneration of peri-implantitis cases based upon radiographic analyses of multiple peer-reviewed publications. Laser-assisted peri-implantiitis protocol (“LAPIP”) involves a well-documented sequence of clinical procedures including NdYAG laser (1064 nM wavelength) applications of:
- De-epithelize pocket epithelium of the diseased implant collar.
- Microbial reduction focusing on black-pigmented bacteroides bacteria and the “red complex” of pathogenic bacteria.58,59
- Control of inflammation in the contiguous soft tissues surround the diseased implant
- Biostimulation of wound healing cells involved in regeneration. 60
Prosthetic Correction and Accessibility: There is a recognized association between prosthetic design and peri-implant health. Factors such as the implant-abutment connection, restorative material selection, prosthetic design, and cleansability can significantly influence peri-implant soft tissue health.17 Inadequate prosthetic design may predispose the implant to food trapping, biofilm attachment, and calculus formation. A high proportion of implants diagnosed with peri-implantitis are associated with poor accessibility for adequate oral hygiene, emphasizing the need for routine evaluation and correction of prosthetic design to minimize these risks (Fig. 8).18
Fig. 8
Occlusal adjustments/occlusal guards
Both natural teeth and implant prostheses should be in physiologic occlusion, harmonizing with the functions of the masticatory system. Various factors, including masticatory function, dietary intake, aging, bite force, temporomandibular joint disorders, and parafunctional habits, can alter the occlusal scheme over time.61,62 Biomechanical complications, such as those affecting screws, abutments, prostheses, and implant structures, as well as biological complications impacting osseointegration and surrounding alveolar bone, have been linked to occlusal discrepancies and overload.63,64
Recent systematic reviews have demonstrated a positive correlation between occlusal overload and peri-implant crestal bone loss.16 Therefore, regular occlusal evaluations, adjustments, and the use of occlusal guards are recommended to protect the final prosthesis and reduce the risk of implant failure and peri-implant diseases.63
Soft tissue correction—Increasing keratinized gingiva
The dimension of keratinized tissue around implants is crucial for maintaining peri-implant health.65,66 The incidence of inadequate keratinized gingiva around implants has been reported to range from 46% to 74%.67 A wide band of keratinized mucosa is associated with improved peri-implant health parameters, including plaque index, modified gingival index, mucosal recession, and attachment loss.68 A recent systematic review also suggests that the lack of buccal keratinized mucosa may be associated with peri-implant parameters, such as bleeding on probing and marginal bone loss.69 Consequently, soft tissue correction may be necessary when a lack of keratinized tissue is linked to peri-implant diseases. (Fig. 9)
Fig. 9
Conclusion
Clinicians must regularly assess the soft and hard tissue conditions of implants and implant prostheses, providing professional maintenance tailored to the individual needs of each patient. Patients must be consistently instructed and reinforced in their self-administered care to ensure the long-term success of implant therapy. A variety of treatment options are available, depending on the level of implant health and the severity of peri-implant diseases. 
Oral Health welcomes this original article.
References
- Mancini L, Strauss FJ, Lim HC, Tavelli L, et al. Impact of keratinized mucosa on implant-health related parameters: A 10 year prospective re-analysis study. Clin Implant Dent Relat Res. 2024 Jun;26(3):554-563.
- Howe MS, Keys, W & Richards, D. Long-term (10-year) dental implant survival: A systematic review and sensitivity meta-analysis. J. of Dent. 2019; 84:9-21.
- Moraschini V, da C Poubel, Ferreira VF, Barboza E dos S.P. Evaluation of survival and success rates of dental implants reported in longitudinal studies with a follow-up period of at least 10 years: a systematic review. Int. J. Oral Maxillofac. Surg. 2015 Mar; 44(3):377-88.
- Mombelli A, Muller N., Cionca N. The epidemiology of peri-implantitis. Clin Oral Implants Res. 2012 Oct:23 Suppl 6:67-76.
- Ting M, Craig J, Balkin BE, Suzuki JB. Peri-implantitis: A comprehensive overview of systematic reviews. J Oral Implantol. 2018 June; 44(3):225-247.
- Diaz P, Gonzalo E, Gil Villagra LJ, Miegimolle B, Suarez MJ. What is the prevalence of peri-implantitis? A systematic review and meta-analysis. BMC Oral Health 2022; Oct 19;22(1):449.
- Bidra AS, Daubert DM, Garcia LT, Gauthier MF, et al. A systematic review of recall regimen and maintenance regimen of patients with dental restorations. Part 2: Implant-Borne Restorations. J Prosthodont. 2016 Jan 25: Suppl 1:S16-31.
- Salvi GE & Ramseier CA. Efficacy of patient-administered mechanical and/or chemical plaque control protocols in the management of peri-implant mucositis. A systematic review. J. Clin Periodontol 2015 Apr: 42 Suppl 16:S187-201.
- Bader J. Risk-based recall intervals recommended. Evid Based Dent. 2005;6(1):2-4.
- Misch CE & Resnik RR. Misch’s avoiding complications in oral implantology, 1st edition. 2018 Elsevier.
- Resnik RR. Misch’s contemporary implant dentistry. 4th Edition. 2020 Elsevier.
- Schwarz F, Derks J, Monje A, Wang HL. Peri-implantitis. J Periodontol 2018 June: 89 Suppl 1:S267-S290.
- Costa FO, Takenaka-Martinez S, Miranda Cota LO, Ferreira SD, et al. Peri‐implant disease in subjects with and without preventive maintenance: A 5‐year follow up. J Clin Periodontol. 2012 Feb;39(2): 173-181.
- Corbella S, Del Fabbbro M, Taschieri S, De Siena F, Francetti L. Clinical evaluation of an implant maintenance protocol for the prevention of peri-implant diseases in patients treated with immediately loaded full-arch rehabilitations. Int J Dent Hyg. 2011 Aug;9(3):216-222.
- Jepsen S, Berglundh T, Genco R, Aass AM, Demirel K, et al. Primary prevention of peri-implantitis: managing peri-implant mucositis. J Clin Perodontol 2015 Apr:42 Suppl 16:S152-157.
- Caton JG, Armitage G, Berglundh T, Chapple ILC, et al. A new classification scheme for periodontal and peri-implant diseases and conditions- introduction and key changes from the 1999 classification. J Clin Periodontol. 2018 June: 45 Suppl 20:S1-S8.
- Di Fiore A, Montagner M, Sivolella ST, Stellini E, et al. Peri-implant bone loss and overload: a systematic review focusing on occlusal analysis through digital and analogic methods. J Clin Med 2022 Aug 17;11(16):4812.
- Hamilton A, Putra A, Nakapaksin P, Kamolroongwarakul, Gallucci GO. Implant prosthodontic design as a predisposing or precipitating factor for peri-implant disease: A review. Clin Implant Dent Relat Res 2023 Aug;25(4):710-722.
- Serino G & Ström C. Peri-implantitis in partially edentulous patients: association with inadequate plaque control. Clin Oral Implan. Res 2009 Feb;20(2):169–174
- Heitz-Mayfield LJA & Salvi GE Peri-implant mucositis. J Periodontol 2018; Jun:89 Suppl 1:S257-S266.
- Staubli N, Walter C, Schmidt JC, Weiger R, Zitzmann NU. Excess cement and the risk of peri-implant disease – A systematic review. Clin Oral Implants Res. 2017 Oct;28(10):1278–1290.
- Wilson Jr. TG. The positive relationship between excess cement and peri-implant disease: A prospective clinical endoscopic study. J Periodontol. 2009 Sep;80(9):1388–1392.
- Carvalho ÉBS, Romandini M, Sadilina S, Sant’Ana ACP, Sanz M. Microbiota associated with peri-implantitis-A systematic review with meta-analyses. Clin Oral Implants Res. 2023 Nov;34(11):1176–1187.
- Lafaurie GI, Sabogal MA, Castillo DM, Rincon MV, et al. Microbiome and microbial biofilm profiles of peri-implantitis: a systematic review. J Periodontol. 2017 Oct;88(10):1066–1089.
- Van Velzen FJJ, Lang NP, Schulten EAJM, Bruggenkate CMT. Dental floss as a possible risk for the development of peri-implant disease: an observational study of 10 cases. Clin Oral Implants Res. 2016 May;27(5):618-21.
- Dos Reis INR, Fukuoka GL, Nagay BE, Pannuti CM, et al. Incidence of peri-implant disease associated with cement and screw retained implant supported prostheses: a systematic review and meta-analysis. J Prosthet Dent. 2023 Oc5 2:S0022-3913(23)00563-2.
- Ata-Ali J, Ata-Ali F, Galindo-Moreno P. Treatment of periimplant mucositis: a systematic review of randomized controlled trials. Implant Dent 2015 Feb;24(1):13–18.
- Valderrama P, Blansett JA, Gonzalez MG. Cantu MG, Wilson TG. Detoxification of Implant Surfaces Affected by Peri-Implant Disease: An Overview of Nonsurgical Methods. Open Dent. J. 2014 May 16:8:77–84.
- Nee WY, Awang RAR, Hassan A. 2022 Effects on the Titanium Implant Surface by Different Hygiene Instrumentations: A Narrative Review Cureus Oct 30;14(10):e30884.
- Chen A, Ghaffar H, Taib H, Hassan A. A review of bacterial colonization on dental implants with various hygiene instruments. Cureus 2023 Oct 22; 15(10):e47483.
- Menini M, Setti P, Dellepiane E, Zunino P, Pera P, Pesce P. Comparison of biofilm removal using glycine air polishing versus sodium bicarbonate air polishing or hand instrumentation on full-arch fixed implant rehabilitations: a split-mouth study. Quintessence int. 2019;50(9):722-730.
- Menini M, Delucchi F, Bagnasco F, Pera F, et al. Efficacy of air polishing devices without removal of implant supported full arch prostheses. Int J Oral Implantol (Berl) 2021 Nov 2; 14(4):401-416.
- Abdulbaqi HR, Shaikh MS, Abdulkareem AA, Zafar MS, et al. Efficacy of erythritol powder air polishing in active and supportive periodontal therapy: a systematic review and meta-analysis. Int J Dent Hyg. 2022 Feb; 20(1):62-74.
- Leonhardt, A., Renvert, S. & Dahlén, G. Microbial findings at failing implants. Clin. Oral Implants Res. 1999 Oct;10(5):339-45.
- Krayer JW. Leite R, & Kirkwood KLl. Non-surgical chemotherapeutic treatment strategies for the management of periodontal diseases. Dent Clin North Am. 2010 Jan: 54(1):13-33.
- Valderrama P, Blansett JA, Gonzalez MG, Cantu MG, Wilson TG. Detoxification of implant surfaces affected by peri-implant disease: An overview of nonsurgical methods. Open Dent. J. 2014 May 16:8:77–84.
- Muthukuru M, Zainvi A, Esplugues EO, Flemming TF. Non-surgical therapy for the management of peri-implantitis: a systematic review. Clin Oral Implants Res 2012 Oct 23 Suppl 6:77–83.
- Lindhe J, Meyle J., Group D of European Workshop on Periodontology. Peri-implant diseases: consensus report of the Sixth European Workshop on Periodontology. J Clin Periodontol. 2008 Sep;35 (8 Suppl):282-285.
- Schwarz F, Schmucker A & Becker J. Efficacy of alternative or adjunctive measures to conventional treatment of peri-implant mucositis and peri-implantitis: a systematic review and meta-analysis. Int J Implant Dent. 2015 Dec;1(1):22.
- Klinge B, Meyle J & Working Group 2. Peri-implant tissue destruction. The third EAO consensus conference 2012. Clin Oral Implants Res. 2012 Oct:23 Suppl 6:108-110.
- Khoury F, Keeve PL, Ramanauskaite A, Schwarz F, Koo KT, Sculean A, Romanos G. Surgical treatment of peri-implantitis—consensus report of Working Group 4. Int Dent J. 2019 Sep;69 Suppl 2(Suppl 2): 18-22.
- Renvert S., Polyzois I., & Clafey N. Surgical therapy for the control of peri-implantitis. Clin. Oral. Implant. Res. 2012 Oct:23 Suppl 6:84–94.
- Keeve P.L., Koo KT, Ramanauskaite A, Romanos G, et al. Surgical treatment of peri-implantitis with non-augmentative techniques. Implant. Dent. 2019 Apr;28(2):177–186.
- Schwarz F, Jepsen S, Obreja K, Galarraga-Vinueza ME, Ramanauskaite A. Surgical therapy of peri-implantitis. Periodontol 2000. 2022 Feb;88(1):145-181.
- Figuero E, Graziani F, Sanz I, Herrera D, Sanz Mariano. Management of peri-implant mucositis and peri-implantitis. Periodontol 2000. 2014 Oct;66(1):255-273.
- Carcuac O, Derks J, Charalampakis G, Abrahamsson I, Wennstrom J, Berglundh T. Adjunctive systemic and local antimicrobial therapy in the surgical treatment of peri-implantitis: a randomized controlled clinical trial. J Dent Res. 2016 Jan;95(1):50-57.
- Ramel C.F., Lussi A, Ozcan M, Jung RE, Hammerle CHF, Thoma DS. Surface roughness of dental implants and treatment time using six different implantoplasty procedures. Clin. Oral Implant. Res. 2016;Jul;27(7):776–781.
- Stavropoulos A, Bertl K, Eren S, Gotfredsen K. Mechanical and biological complications after implantoplasty- A systematic review. Cin Oral Implants Res 2019 Sep;30(9):833-848
- Ramanauskaite A, Obreja K, Sader R, Khoury F. Surgical treatment of periimplantitis with augmentative technique. Implant Dent. 2019 Apr;28(2):187-209.
- Jepsen S, Schwarz F, Cordaro L, Derks J, Hammerle CHF et al. Regeneration of alveolar ridge defects. Consensus report of Group 4 of the 15th European Workshop on Periodontology on Bone Regeneration. J Clin Periodontol. 2019 Jun;46 Suppl 21:277-286.
- Schwarz F, Sahm N, Bieling K, Becker J. Surgical regenerative treatment of peri-implantitis lesions using a nanocrystalline hydroxy-apatite or a natural bone mineral in combination with a collagen membrane: a four-year clinical follow-up report. J Clin Periodontol. 2009 Sep;36(9):807-814.
- Aghazadeh A, Persson GR, Renvert S. A single-centre randomized controlled clinical trial on the adjunct treatment of intra-bony defects with autogenous bone or a xenograft: results after 12 months. J Clin Periodontol. 2012 Jul;39(7):666-673.
- Khouly I, Pardinas-Lopez S, Ruff RR, Strauss FJ. Efficacy of growth factors for the treatment of peri-implant diseases: a systematic review and meta-analysis. Clin Oral Investig. 2000 Jul;24(7):2141-2161.
- Astolfi V, Gomez-Menchero A, Rios-Santos JV, Bullon P, Galeote F, et al. Influence of removing or leaving the prosthesis after regenerative surgery in peri-implant defects: retrospective study: 32 clinical cases with 2 to 8 years of follow up. Int J Environ Res Public Health 2021 Jan 13;18(2):645.
- Monje A & Nart J. Management and sequelae of dental implant removal. Periodontology 2000. 2022 Feb;88(1):182-200.
- Masaki C, Kondo Y, Tomoeda, Nodai T, et al. Treatment strategies for dental implant removal: A literature review. Japanese Dental Science Review. Japanese Dental Science Review. 2024 Dec (60):120-127.
- Sarmast ND, Wang HH, Soldatos NK, Angelov N, et al. A novel treatment decision tree and literature review of retrograde peri-implantitis. J Periodontol 2016 Dec;87(12):1458-1467.
- Ting, M. C Alluri LS, Sulewski JG, Suzuki JB, da Silva APB. Laser treatment of peri-implantitis: a systematic review of radiographic outcomes. Dent J (Basel). 2022 Jan 28;10(2):20.
- Socransky SS & Haffajee AD. The nature of periodontal diseases. Ann Periodontol. 1997 Mar;2(1):3-10.
- Socransky SS & Haffajee AD. Periodontal microbial ecology. Periodontol 2000. 2005;38:135–87.
- Suzuki JB. Salvaging implants with an Nd:YAG laser: a novel approach to a growing problem. Compend Contin Educ Dent. 2015 Nov-Dec;36(10):756-61.
- Sheridan RA, Decker AM, Plonda AB, Wang HL. The role of occlusion in implant therapy: a comprehensive updated review. Implant Dent. 2016 Dec; 25(6): 829-838.
- Higashi K, Harra K, Mameno T, Takahashi T, et al. The relationship between changes in occlusal support and masticatory performances using 6-year longitudinal data from the SONIC study. J Dent. 2023 Dec:139:104763.
- Lobbezzo F, Brouwers JEIG, Cune MS, Naeije Me. Dental implants in patients with bruxing habits. J Oral Rehabil. 2006 Feb; 33(2):152-159.
- Misch CE, Suzuki JB, Misch-Dietsh FM, Bidez MW. A positive correlation between occlusal trauma and peri-implant bone loss: literature support. Implant Dent. 2005 Jun;14(2):108-16.
- Thoma DS, Gil A, Hammerle CH, Jung RE. Management and prevention of soft tissue complications in implant dentistry. Periodontol 2000. 2022 Feb;88(1):116-129.
- Benic GI, Mokti M, Chen CJ, Weber HP, Hammerle CHF, Gallucci GO. Dimensions of buccal bone and mucosa at immediately placed implants after 7 years: a clinical and cone beam computed tomography study. Clin Oral Implants Res. 2012 May;23(5):560-566.
- Wennström JL & Derks J. Is there a need for keratinized mucosa around implants to maintain health and tissue stability? Clin Oral Implants Res. 2012; Oct 23 Suppl6:136-146.
- Lin GH, Chan HL, Wang HL. The Significance of Keratinized Mucosa on Imp lant Health: A Systematic Review. J. Periodontol. 2013, Dec;84(12):1755–1767.
About the Authors

Dr. Yueh J. Hsiao, Graduate Periodontology and Oral Implantology Program Director, Associate Professor, Temple University, Philadelphia, PA, USA.

Dr. Randy R. Resnik, Clinical Professor, Temple University, Philadelphia, PA, USA. Clinical Professor, University of Pittsburgh School of Dental Medicine, Pittsburgh, PA, USA. Clinical Professor, Allegheny General Hospital, Pittsburgh, PA, USA.

Dr. Jon B. Suzuki, Clinical Professor, University of Maryland, Baltimore, MD, USA. Clinical Professor, University of Washington, Seattle, WA, USA. Clinical Professor, Nova-Southeastern University, Ft. Lauderdale, FL USA. Professor Emeritus, Temple University, Philadelphia, PA, USA.