Abstract
This study aimed to compare the effectiveness of 450 nm blue diode laser and 465 nm blue LED light in reducing oral Lactobacillus bacteria. A total of 102 human participants were randomly assigned to three groups: Sugar-Free Gum (control), Blue LED, and Blue Laser. Post-treatment saliva samples showed average bacterial reductions of 19% (control), 54% (LED), and 86% (laser). Within the limitations of this study, blue light phototherapy — especially the 450 nm laser — demonstrated significant antimicrobial potential in oral care.
Keywords
Oral bacteria, bacterial reduction, blue laser therapy, blue LED therapy, photobiomodulation, saliva analysis, non-invasive treatment, dental hygiene, antimicrobial effect, laser dentistry.
The antimicrobial effects of blue light (400-500 nanometers) are well documented: it disrupts microbial biofilms via reactive oxygen species (ROS) produced by endogenous bacterial chromophores, such as porphyrins and flavins, and has been shown to significantly reduce key oral pathogens including Streptococcus mutans and Lactobacillus species.1 Since these bacteria are primary contributors to dental caries,2 optimizing light-based therapies could advance non-invasive cavity prevention, as well as reducing antibiotic resistance by reducing the need for prescriptions.
While previous studies have predominantly focused on LEDs (e.g., 405 nm or 450 m),3 blue laser therapy — with its coherent, monochromatic output — may deliver more targeted antimicrobial action, deeper penetration, and less dispersion compared to broader-band LEDs.4 This experiment therefore aimed to directly compare a blue LED device (Multi Radiance MR5 Activ Pro – Ohio, USA, 465 nm, FDA-Class I) against a blue diode laser (Pioon UTU, China, 450 nm, FDA-Class IV) in their ability to reduce oral Lactobacillus. The null hypothesis is that there is no difference between these two devices.
Lactobacillus species were chosen specifically because they play a significant role in caries progression and biofilm persistence; it is acidogenic and often more resilient in acidic micro ecosystems, making its management highly relevant to oral health.5
Related article: What’s BLUE in dentistry?!
Values in Table 1 represent the dose per quadrant and reflect a meaningful difference in total light exposure. The higher energy density of the laser allows for deeper or more targeted bacterial disruption compared to the lower-intensity, broad-spectrum LED,6 clarifying how irradiance, coherence, and energy may impact microbial reduction in the oral cavity.
Table 1: Device specifications and dosimetric parameters for laser and LED treatments
| Device | Wavelength | Power | Area | Time | Power Density | Energy Density |
| Laser | 450 nm | 3 Watts | 6 cm2 | 300 seconds | 0.5W/cm2 | 150J/cm2 |
| LED | 465 nm | .5 Watts | 6 cm2 | 300 seconds | 0.083W/cm2 | 25J/cm2 |
We obtained the following: Both treatments were applied over a fixed area of 6 cm2 for 300 seconds. The laser delivered a total energy density of 150 J/cm2, while the LED delivered 25 J/cm2.
Methods and materials
Study design and participant allocation
This study is a randomized controlled clinical trial designed to compare the antimicrobial effects of blue laser therapy and blue LED therapy against a control condition in a human population. A total of 102 participants were enrolled and randomly assigned (34 per group) to one of three groups: (1) chewing sugar free gum (control), (2) blue LED therapy, or (3) blue laser therapy. To provide a meaningful baseline, the control group utilized sugar-free gum chewing — a recognized method for stimulating saliva flow without introducing fermentable sugars.
Xylitol-containing gum in particular has been shown to reduce levels of key oral bacteria, including Streptococcus mutans and Lactobacillus species.7 Randomization and labeling were performed prior to sample collection to reduce allocation bias. Participants were randomly assigned to one of the three treatment groups using a computerized random number generator to ensure equal distribution across groups. Each participant was labeled with a unique identifier using tape, such as “1.1” or “2.2,” where the first digit represented the participant number and the decimal indicated the sample stage (1 = pre-treatment, 2 = post-treatment). The assigned treatment group — G (gum), LE (LED), or LA (laser) — was also marked on the label to allow for sample tracking.
Laser/light parameters
The light parameters were selected for both safety and comparability: the LED was at factory preset wavelength of 465 nm (bandwidth ± 20–30 nm), and the laser at 450 nm (±2 nm), with stationary contact delivery mode for 20 minutes total (5 minutes per quadrant). The broader bandwidth and lower coherence of LED radiation leads to greater dispersion and reduced irradiance uniformity, even at matched power settings.
The laser was operated in continuous wave (CW) mode, consistent with clinical decontamination protocols. Device attachment sizes were determined via manufacturer specifications: both the laser and LED utilized wide, quadrant-shaped applicator heads designed to cover multiple teeth simultaneously (Fig. 1 and 2). As each device was held in stationary contact with the teeth for five minutes per quadrant, the estimated irradiated area per quadrant was approximately 6 cm2.
Fig. 1

Fig. 2

Sample collection and preparation
Saliva samples were collected immediately before and after the assigned intervention for each participant. Samples were plated onto MRS (de Man, Rogosa, and Sharpe – the three researchers who developed the medium) agar, a selective growth medium for Lactobacillus spp., minimizing growth of non-target oral flora and reducing confounding variables.8 Each sample was plated under sterile conditions using sterile swabs. To ensure anaerobic conditions conducive to Lactobacillus growth, petri dishes were sealed using laboratory-grade tape to restrict oxygen infiltration. All samples were then stored in a dark, enclosed environment maintained at approximately 98°F (37°C)9 to simulate human oral temperature.
Fig. 3

Fig. 4A
Fig. 4B
Incubation and colony counting
All plates were incubated for 72 hours, after which colony growth was analyzed. Each plate was digitally photographed, and CFUs (colony-forming units) were manually counted using visual dot-annotation software to ensure accurate quantification. CFU counts were recorded for each participant before and after treatment (Fig. 5).
Fig. 5
Results
The effectiveness of each intervention on oral bacterial reduction was assessed by comparing percent reduction in colony-forming units (CFUs) from pre- to post-treatment saliva samples. As shown in Figure 6, participants in the sugar-free gum control group exhibited a mean bacterial reduction of 19%, while those treated with blue LED and blue laser therapy demonstrated reduction of 54% and 86%, respectively.
Fig. 6
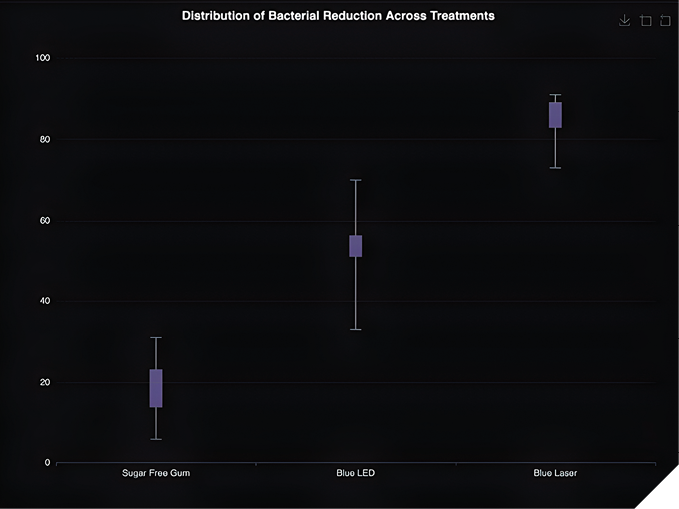
Box plot analysis reveals increasing median percent reduction across the three groups, with the greatest interquartile spread observed in the sugar free gum group. The blue laser group and blue LED group demonstrated the smallest interquartile ranges, indicating more uniform reductions among participants.
Statistical analysis
Statistical evaluation was performed on percentage-reduction values (post-treatment CFU relative to each subject’s baseline) to control for baseline differences in bacterial load between subjects. Preliminary diagnostics confirmed that the data satisfied the core assumptions for parametric inference: Shapiro-Wilk tests applied to residuals for each treatment group were non-significant (p > 0.10 – Table 2) and Q-Q (quantile – quantile) inspection showed no significant departures (Table 2) from normality. A one-way analysis of variance (ANOVA) subsequently revealed a pronounced main effect of treatment on bacterial reduction (F₂,₉₉ = 993.32, p = 1.11 × 10-16), with an associated η² of 0.77, demonstrating that roughly three-quarters of the total outcome variance was attributable to treatment modality. Descriptively, the gum-only control achieved a mean reduction of 19.46 ± 6.94 % (Table 2), exposure to a blue LED produced 54.31 ± 6.73 % (Table 2), and blue-laser irradiation yielded 85.94 ± 4.48 % (all n = 34 – Table 2). Post-hoc Tukey HSD comparisons indicated that every pairwise difference remained highly significant (all adjusted p < 0.001): the LED outperformed the control by 34.8 percentage points, the laser exceeded the control by 66.5 points, and the laser surpassed the LED by 31.6 points. Collectively, these findings substantiate the hypothesis that photobiomodulation markedly enhanced antibacterial efficacy relative to mastication alone, with blue-laser treatment providing the greatest benefit and blue LED offering an intermediate effect. The null hypothesis can be rejected.
Table 2: One-way ANOVA summary for treatment effects.
| Treatment ➞ | Gum | LED | Laser | Pooled Total |
| Observations N | 34 | 34 | 34 | 102 |
| Sum ∑xi | 661.7961 | 1846.538 | 2921.8679 | 5430.202 |
| Mean X | 19.4646 | 54.3099 | 85.9373 | 53.2373 |
| Sum of Squares ∑Xi2 | 14471.6227 | 101778.0271 | 251760.9302 | 368010.5801 |
| Sample Variance s2 | 48.1828 | 45.2321 | 20.1067 | 781.4003 |
Discussion
This study demonstrates a clear gradient in antimicrobial efficacy: blue laser therapy (86% mean reduction) significantly outperformed blue LED (54%) and sugar-free gum (19%). These findings are consistent with prior literature indicating that coherent, high fluence laser exposure achieves more rapid bacterial killing than broader band LED light.10 An in vitro study done by pediatric dentists systematically explored the effects of varying output powers of a 445 nm blue diode laser used in antimicrobial photodynamic therapy (aPDT) with photosensitizers (curcumin or riboflavin) on Streptococcus mutans adhered to orthodontic brackets.11 The authors observed a clear dose-dependent relationship, with the highest bacterial reductions occurring at 500 mW output when paired with curcumin, achieving CFU reductions statistically equivalent to 0.2% chlorhexidine (p<0.001). Although this study did not incorporate photosensitizers, the laser fluence (estimated at 150 J/cm²) and stationary contact application mirror the BDL (Blue Diode Laser) exposure parameters in their curcumin groups. The key overlap lies in both studies’ demonstration that coherent, high-intensity blue laser therapy yields greater bacterial suppression than low-intensity or broad-spectrum alternatives. Their use of orthodontic brackets also extends relevance to plaque-prone anatomical surfaces, paralleling the real world application to human oral microbiota.
Another literature review outlined the antibacterial potential of 405 nm diode lasers in cavity and endodontic sterilization, particularly when paired with photosensitizers such as erythrosine.12 One of the most compelling findings is that under specific energy densities (e.g., 25.34 J/cm² for 75 seconds), Streptococcus mutans mortality reached up to 99.26% with curcumin as a photosensitizer and 100% when using erythrosine. These data highlight the amplifying role of photosensitizers in blue-light therapy but also suggest that wavelengths within the 405–450 nm spectrum alone carry strong bactericidal capability. Importantly, the review notes that power, exposure time, and biofilm structure critically determine outcomes — an observation directly aligned with this study’s finding that laser therapy (20 minutes, continuous wave, stationary contact) vastly outperformed LED in both efficacy and consistency. Unlike many purely in vitro experiments cited in this review, the use of human saliva samples introduced host factors such as salivary proteins and surface irregularities. Despite these added complexities, the laser still achieved an 86% reduction — comparable in magnitude to some of the highest performing photodynamic protocols in this review.
A randomized clinical trial done by doctors in the Department of Conservative Dentistry and Endodontics at the Government Dental College and Hospital in Ahmedabad, India evaluated the antibacterial efficacy of 940 nm diode laser, photodynamic therapy (PDT) with Rose Bengal-functionalized chitosan nanoparticles (CS-NPs), and 2% chlorhexidine in deep carious lesions.13 The study found that while all three interventions led to significant reductions in Streptococcus mutans and Lactobacillus counts, chlorhexidine exhibited the highest bacterial reduction, followed by PDT, with the 940 nm diode laser performing an effectiveness with the following percentages: TVC (total viable count) reduction 81%; S. mutans: 74.6%; Lactobacillus: 86.8%. While the 940 nm diode laser group achieved an 86.8% reduction in Lactobacillus count, our 450 nm blue laser yielded a comparable 86% reduction — close despite targeting bacterial colonies in live saliva without adjunctive photosensitizers. Notably, their diode laser achieved only a 74.6% reduction in S. mutans and 81% in TVC, supporting the effectiveness of blue-spectrum lasers for selective bacterial suppression.
Table 3: Descriptive statistics for bacterial reduction across treatments.
| Source | Sum of Squares SS | Degrees of Freedom υ | Mean Square MS | F statistic | p-value |
| Treatment | 75175.2166 | 2 | 37587.6083 | 663.617 | 1.11E-16 |
| Error | 3746.2104 | 99 | 37.8405 | ||
| Total | 78921.4271 | 101 |
The accessibility of LED devices may allow for longer treatment durations or more frequent application in a home setting.14 This may ultimately offset the higher upfront clinical efficacy of laser therapy, particularly for patients with limited access to in office care. Within the limitations of this study, which suggests a superiority of blue-laser irradiation over both blue LED exposure and mastication alone, it also opens several clinically relevant questions that warrant exploration. First, a time-efficiency optimization study is needed: by repeating the trial with progressively shortened periods (e.g., 5,10 and 15 minutes versus the current 20 minutes benchmark) investigators can determine the minimal exposure that still achieves an equivalent outcome (margin ≤ 5 percentage points in bacterial-load reduction). Such duration modeling would enhance patient acceptability and chair side efficiency/workflow. Second, the durability of the antibacterial effect should be assessed with longitudinal sampling.15 Collecting follow-up saliva at meaningful intervals (e.g., 24 hours, 7 days, and 30 days post treatment) would reveal whether bacterial counts rebound, and if so, at what rebound threshold (e.g., 50% of baseline). Mapping this rebound curve will, in turn, suggest an evidence-based maintenance schedule: the frequency (times per week or per month) at which repeated laser sessions are required to keep bacterial load below a predefined ceiling. A design combining shortened duration with varied treatment intervals would allow calculation of an optimal time vs. frequency schedule that maximizes efficacy while minimizing patient burden and resource utilization. Finally, extending this study to include microbiome sequencing and biomarkers would clarify whether sustained reductions in salivary CFU translate into long term ecological and host-response benefits. Most notably, the blue LED light offers a more practical application pathway due to its affordability, safety, and potential for at-home use, without requiring chair time or professional oversight.
This study demonstrates that blue diode laser photobiomodulation significantly outperforms both blue LED light and non-light-based mechanical stimulation (chewing sugar-free gum) in reducing salivary bacterial load. Quantitative analysis revealed a clear, statistically significant gradient of efficacy, with blue laser achieving the greatest mean percentage reduction. These findings suggest that short-term application of specific light wavelengths, particularly coherent blue-laser energy, may serve as a powerful non-invasive addition in oral hygiene protocols.
Table 4: Tukey HSD pairwise comparisons between treatment groups.
| Treatment Pair | Tukey HSD Q Statistic | Tukey HSD p-value | Tukey HSD inference |
| Gum vs. LED | 33.0928 | 0.0010053 | ** p<0.01 |
| Gum vs. Laser | 63.0092 | 0.0010053 | ** p<0.01 |
| LED vs. Laser | 29.9795 | 0.0010053 | ** p<0.01 |
Conclusion
Within the limitations of this study, both 450 nm blue laser and 465 nm blue LED light demonstrated bacterial reduction in oral samples. While immediate effects were observed, further research is needed to evaluate long-term outcomes and optimal retreatment intervals. These findings support the potential of blue light phototherapy as a promising avenue in dental care. 
Oral Health welcomes this original article.
References
- Wang, Y., Wang, Y., Wang, Y., Murray, C. K., Hamblin, M. R., Hooper, D. C., & Dai, T. (2017). Antimicrobial blue light inactivation of pathogenic microbes: State of the art. Drug resistance updates : reviews and commentaries in antimicrobial and anticancer chemotherapy, 33-35, 1–22. https://doi.org/10.1016/j.drup.2017.10.002
- Lena, A., Marino, M., Manzano, M., Comuzzi, C., & Maifreni, M. (2023). An Overview of the Application of Blue Light-Emitting Diodes as a Non-Thermic Green Technology for Microbial Inactivation in the Food Sector. Food Eng Reviews 16, 59–84 (2024). https://doi.org/10.1007/s12393-023-09355-1
- Caufield, P. W., Schön, C. N., Saraithong, P., Li, Y., & Argimón, S. (2015). Oral Lactobacilli and Dental Caries: A Model for Niche Adaptation in Humans. Journal of Dental Research, 94(9 Suppl), 110S–8S. https://doi.org/10.1177/0022034515576052
- Heiskanen, V., & Hamblin, M. R. (2018). Photobiomodulation: lasers vs. light emitting diodes?. Photochemical & photobiological sciences : Official journal of the European Photochemistry Association and the European Society for Photobiology, 17(8), 1003–1017. https://doi.org/10.1039/c8pp90049c
- Huang, S., Lin, S., Qin, H., Jiang, H., & Liu, M. (2023). The Parameters Affecting Antimicrobial Efficiency of Antimicrobial Blue Light Therapy: A Review and Prospect. Biomedicines, 11(4), 1197. https://doi.org/10.3390/biomedicines11041197
- Zein, R., Selting, W., & Hamblin, M. R. (2018). Review of light parameters and photobiomodulation efficacy: dive into complexity. Journal of biomedical optics, 23(12), 1–17. https://doi.org/10.1117/1.JBO.23.12.120901
- Mäkinen, K. K., Alanen, P., Isokangas, P., Isotupa, K., Söderling, E., Mäkinen, P. L., Wenhui, W.,
- Weijian, W., Xiaochi, C., Yi, W., & Boxue, Z. (2008). Thirty-nine-month xylitol chewing-gum programme in initially 8-year-old school children: a feasibility study focusing on mutans streptococci and lactobacilli. International Dental Journal, 58(1), 41–50. https://doi.org/10.1111/j.1875-595x.2008.tb00175.x
- Vinderola, C. G., & Reinheimer, J. A. (1999). Culture media for the enumeration of Bifidobacterium bifidum and Lactobacillus acidophilus in the presence of yoghurt bacteria. International Dairy Journal, 9(8), 497–505. https://doi.org/10.1016/S0958-6946(99)00120-X
- Śliżewska, K., & Chlebicz-Wójcik, A. (2020). Growth Kinetics of Probiotic Lactobacillus Strains in the Alternative, Cost-Efficient Semi-Solid Fermentation Medium. Biology, 9(12), 423. https://doi.org/10.3390/biology9120423
- Amaroli, A., Ravera, S., Zekiy, A., Benedicenti, S., & Pasquale, C. (2022). A Narrative Review on Oral and Periodontal Bacteria Microbiota Photobiomodulation, through Visible and Near-Infrared Light: From the Origins to Modern Therapies. International journal of molecular sciences, 23(3), 1372. https://doi.org/10.3390/ijms23031372
- Pordel, E., Ghasemi, T., Afrasiabi, S., Benedicenti, S., Signore, A., & Chiniforush, N. (2023). The Effect of Different Output Powers of Blue Diode Laser along with Curcumin and Riboflavin against Streptococcus mutans around Orthodontic Brackets: An In Vitro Study. Biomedicines, 11(8), 2248. https://doi.org/10.3390/biomedicines11082248
- Chalisha, T. N., Febrianti, N. N., Kunarti, S., & Setyabudi, S. (2021). Photodynamic Therapy 405 nm Diode Laser as Antibacterial for Cavity and Root Canal Sterilization. Conservative Dentistry Journal, 11(2), 62–66. https://doi.org/10.20473/cdj.v11i2.2021.62-66
- Disodia, N., Dhanak, N., Pidaparthi, A., Asthana, G., & Kanodia, S. (2025). Evaluating the effectiveness of various disinfectants in deep carious lesions: An in vivo study. Journal of Conservative Dentistry and Endodontics, 28(3), 258–263. https://doi.org/10.4103/JCDE.JCDE_833_24
- Cronshaw, M., Parker, S., Hamadah, O., Arnabat-Dominguez, J., & Grootveld, M. (2025). Photobiomodulation LED Devices for Home Use: Design, Function and Potential: A Pilot Study. Dentistry Journal, 13(2), 76. https://doi.org/10.3390/dj13020076
- Ma, S. Y., Zhou, Q. N., Cai, S., Zhou, Y., Zhang, X. Y., Feng, X. Y., Diao, S., Xi, J. Q., Yu, G. X., Shang, J. J., & Yang, N. Y. (2024). A comparative study of microbial changes in dental plaque before and after single- and multiappointment treatments in patients with severe early childhood caries. BMC oral health, 24(1), 695. https://doi.org/10.1186/s12903-024-04458-5
About the authors

Adarsh Venkatesh is a high school senior with research experience in the use of lasers in dentistry. He’s passionate about pursuing a career in dentistry and aims to contribute to the advancement and innovation of the field.

Dr. Sangita Venkatesh earned her DMD from Tufts University School of Dental Medicine and has been practicing general dentistry in private practice for over 10 years. She is proficient in many aspects of dentistry and is dedicated to patient care, continuously attending seminars to stay up to date on the latest innovations in dental technology.

Marina Polonsky is a gold medal University of Toronto ’99 graduate, she maintains private general practice in Ottawa, Ontario with focus on multi-disciplinary treatment utilizing lasers of different wavelengths. She holds a Mastership from World Clinical Laser Institute (WCLI), Master of Science in Lasers in Dentistry from RWTH University in Aachen, Germany. She is the founder of the Canadian Dental Laser Institute (CDLI), the only study club affiliated with the Academy of Laser Dentistry. She serves on the Executive Committee for Oral Health and is the editor of the Laser Dentistry issue.