
Background: Periodontitis is a common chronic inflammatory disease, with many patients unaware that they have this condition. This is partly because it progresses slowly and often comes with mild or intermittent symptoms. Diagnosing periodontal disease usually requires gingival probing, a reliable method, but one that often gets skipped during routine dental check-ups. Patients who cannot afford regular dental care may only visit the dentist when things worsen—like when they are in pain, they have bleeding and/or swollen gums, or loose teeth. At that point, the focus is on fixing the immediate problem, and the underlying periodontal issues might get missed. In some cases, people who live far from dental services may not get regular care, including periodontal screenings, so their disease condition goes unnoticed. To address these challenges, a new test was developed to make periodontal screenings easier and help dental practitioners keep better track of their patients’ oral health. This article will discuss the concept behind this test and how it can be incorporated into routine dental practice.
Test Concept: This quick test checks for certain inflammatory proteins that are released by inflammatory cells, specifically oral neutrophils, during oral inflammation. When periodontal tissues get inflamed—often because of harmful bacteria—oral neutrophils are the first to respond. They move into the gingival crevice to fight off the infection. The number of oral neutrophils entering the oral cavity correlates with the Oral Inflammatory Load (OIL) and can act as a biomarker for active oral inflammation. This biomarker can identify the presence of inflammation as well as monitor its severity. Oral Inflammatory Load can be measured in a lab by counting neutrophils, or as a point of care chair-side screening test we are introducing here.
How is the test performed? The test consists of three simple steps, does not require specific training, and can be performed by non-dental personnel:
- The patient rinses with 15 mL of tap water for 15 seconds to remove debris and then waits for two minutes.
- The patient rinses with 10 mL of water provided in the test kit, for 30 seconds and then expectorates in the disposable cup.
- A test strip is dipped in the rinse for one second. After one minute, its colour is checked against a colorimetric scale to determine the OIL.
Clinical Application: The chair-side colorimetric test is a helpful tool for screening patients to identify those with higher levels of oral inflammation, most often linked to gingivitis and periodontitis. The test is meant as an aid to diagnosis, for triaging patients and flagging those who have moderate or high levels of inflammation in need of urgent attention. This can be especially useful in busy clinics or remote areas where a full periodontal exam is not possible due to limited staff or limited time. For patients who show elevated inflammation levels, a more thorough assessment can then be scheduled. After comprehensive diagnosis and treatment planning has been established, this test can help track oral inflammation levels on periodontal patients to determine the efficacy of their treatment.
Case series
This series includes three patients seen at the One Smile Research Program, Faculty of Dentistry, University of Toronto. The study was conducted to determine if accessing cost-free service enables people to improve and maintain their oral health.
Case 1: Female, 69 years old. Bleeding gums was her chief complaint. Extraorally, the main finding was temporomandibular joint clicking. Intraorally, the patient presented a poor level of oral hygiene and a Bleeding on Probing (BOP) index of 72%. The patient also showed malocclusion and dental caries in three teeth. The baseline chairside colorimetric test revealed a high oral inflammatory load (Fig. 1A).
Fig. 1A
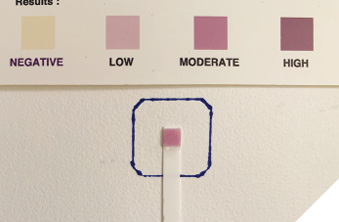
From the 186 sites probed, the patient had 160 sites with probing depth (PD) ≤ 3mm, 19 sites with PD= 4mm, and 7 sites with PD=5mm. The patient received oral hygiene instruction, prophylaxis, scaling and root planning, and restorative treatment for dental caries. Fifteen days after initial treatment, the patient presented at a moderate OIL (Fig. 1B). On her six-month follow-up, a new scaling and root planning treatment was performed. Her oral condition had improved significantly, presenting a low OIL (Fig. 1C).
Fig. 1B
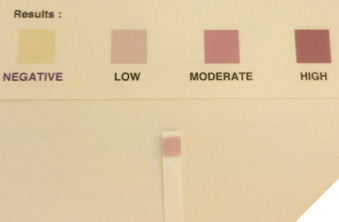
Fig. 1C

Case 2: Male, 65 years old, ex-smoker, Asian. His chief complaint was the presence of dental caries for the past five years. The patient had no significant findings extraorally. Intraorally, a generalized presence of dental calculus was observed in both arches. The patient also presented a pronounced malocclusion, gingival recession, broken teeth, dental caries, and significant external staining. (Figs. 2A-C). A BOP index of 40% was determined upon examination. The initial oral inflammatory level was high (Fig. 3A).
Fig. 2A
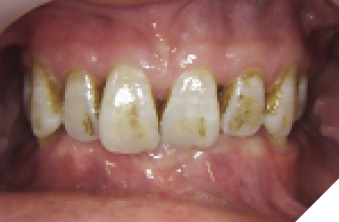
Fig. 2B
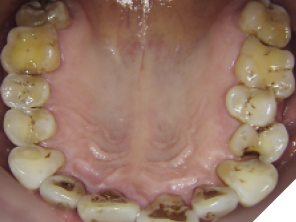
Fig. 2C
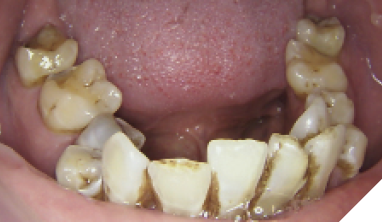
Fig. 3A

After receiving dental care, including oral hygiene education, scaling and root planning, and restorative procedures, the patient presented a significant improvement in his oral condition, evidenced by a marked reduction in his oral inflammatory load (Figs. 3B-C).
Fig. 3B
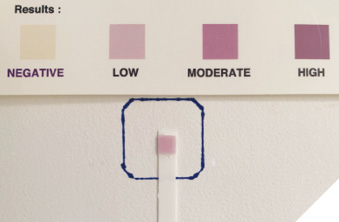
Fig. 3C

Case 3: Female, 32 years old, Caucasian. No major oral complaints. The patient had no significant findings extra orally and presented a good overall oral condition and a mild plaque buildup. A BOP index of 29% was observed. The initial Oral Inflammatory Load was low and remained low in the second and third follow-up appointments (Figs. 4A-C).
Fig. 4A

Fig. 4B

Fig. 4C

A summary of the main clinical and demographic patient characteristics is available in Table 1.
Table 1: Clinical and demographic characteristics of the patients.
| Variable | Case 1 | Case 2 | Case 3 |
| Sex | Female | Male | Female |
| Age | 69 | 56 | 22 |
| Alcohol/ Tobacco/ Vaping/ Cannabis | No | Ex-smoker | No |
| Bleeding index | 72% | 40% | 29% |
| Oral hygiene | Poor | Poor | Fair |
| Plaque | Moderate | Moderate | Mild |
| Calculus | Severe | Moderate | Moderate |
| oPMNs/mL | 240000.00 | 1250000.00 | 400 |
| Baseline OIL | High | High | Low |
Discussion: This case series illustrates how a colorimetric chair-side test can effectively assess a patient’s oral inflammatory profile. The results can confirm observed oral conditions but most importantly can serve as a screening device for what is yet unseen. The test’s utility is similar to the use of the blood pressure gauge in assessing hypertension. Just as elevated blood pressure necessitates a comprehensive evaluation by a physician to determine the underlying cause and appropriate treatment, a moderate or high oral inflammatory load indicates active disease, requiring a thorough and timely examination to identify the cause. This test serves as a valuable tool to screen for oral disease, help determine appropriate treatment, and monitor this treatment for its efficacy.
In the first case, the patient had a high BOP index with significant periodontal involvement. The high BOP index and the presence of periodontal pockets were the likely cause for her initial high inflammation test results. After initial therapy, this case already demonstrated improvement, as demonstrated in the moderate inflammation test results. However, it was at the six-month follow-up, after a second phase of dental care, that the patient reached a low level of OIL. The test helped to capture the improvement of the inflammatory conditions in this patient, who reported feeling encouraged to maintain her oral health by the test results.
In the second case, the elevated neutrophil test result aligned with the patient’s poor overall oral health, presenting moderate plaque and calculus and nearly 50% of the probed sites showing bleeding. Following treatment, the patient’s oral health improved significantly, and the colorimetric test effectively captured the reduction in oral inflammation. This highlights the clinical applicability of the test in monitoring the improvement of oral inflammatory diseases, such as periodontal disease, and in evaluating the effectiveness of dental therapy, as supported by other studies.5, 6, 7
In a scenario where periodontal health is maintained, the test yields a negative or low Oral Inflammatory Load result, as seen in case 3. In such cases, the test result can be used to reinforce patients’ efforts in maintaining good oral hygiene. All three patients were keen to see their test results and appreciated how the test reflected the improvement in their oral condition in the moderate and high Oral Inflammatory Load cases. They also found the test to be educational, as it helped them better understand what oral inflammation and periodontal disease mean—terms that can be difficult for patients to grasp. The colorimetric test helped patients gain a clearer understanding of their oral health status. When asked during follow-ups, patients reported feeling more motivated to maintain their oral hygiene after understanding the importance of reducing oral inflammation through proper hygiene and dental care. This demonstrates the test’s educational value. None of the patients reported any discomfort during the test, emphasizing its non-invasive nature.
Test Limitations: It is important to note that this chair-side test, designed for screening purposes, does not replace a full periodontal evaluation. While it indicates the presence of active inflammation, the test alone cannot substitute for a comprehensive oral examination (with additional diagnostic tools such as biopsies and x-rays), which is required to arrive at a final diagnosis of oral disease. 
Oral Health welcomes this original article.
Acknowledgements: Green Shield Canada for funding. Sonila Dergini, Kamini Kaura and Rida Sbaiti for providing care to these cases.
References
- Scannapieco FA, Gershovich E. The prevention of periodontal disease-An overview. Periodontol 2000. 2020 Oct;84(1):9-13. doi: 10.1111/prd.12330. PMID: 32844421.
- Janakiram C, Dye BA. A public health approach for prevention of periodontal disease. Periodontol 2000. 2020; 84: 202–214. https://doi.org/10.1111/prd.12337
- Levy BB, Goodman J, Eskander A. Oral healthcare disparities in Canada: filling in the gaps. Can J Public Health. 2023 Feb;114(1):139-145. doi: 10.17269/s41997-022-00692-y. Epub 2022 Sep 23. Erratum in: Can J Public Health. 2023 Feb;114(1):146. doi: 10.17269/s41997-022-00710-z. PMID: 36149572; PMCID: PMC9510506.
- Khoury W, Glogauer J, Tenenbaum HC, Glogauer M. Oral inflammatory load: Neutrophils as oral health biomarkers. J Periodontal Res. 2020 Oct;55(5):594-601. doi: 10.1111/jre.12758. Epub 2020 May 6. PMID: 32372438.
- Elebyary O, Sun C, Batistella EA, Van Dyke TE, Low SB, Singhal S, Tenenbaum H, Glogauer M. Utilizing Oral Neutrophil Counts as an Indicator of Oral Inflammation Associated With Periodontal Disease: A Blinded Multicentre Study. J Clin Periodontol. 2024 Aug 19. doi: 10.1111/jcpe.14054. Epub ahead of print. PMID: 39161194.
- Landzberg M, Doering H, Aboodi GM, Tenenbaum HC, Glogauer M. Quantifying oral inflammatory load: oral neutrophil counts in periodontal health and disease. J Periodontal Res. 2015 Jun;50(3):330-6. doi: 10.1111/jre.12211. Epub 2014 Jul 14. PMID: 25040400.
- Bender, J. S., H. Thang, and M. Glogauer. 2006. “Novel Rinse Assay for the Quantification of Oral Neutrophils and the Monitoring of Chronic Periodontal Disease.” Journal of Periodontal Research 41, no. 3: 214–220. https://doi.org/10.1111/j.1600-0765.2005.00861.x.
About the Authors

Dr. Elis Angela Batistella is an Assistant Professor at the College of Dentistry, University of Saskatchewan. Her research focuses on discovering oral biomarkers for oral and systemic diseases and on understanding how oral and systemic health are interconnected.

Dr. Sonica Singhal is an Assistant Professor and the Graduate Program Director of the Dental Public Health specialty at the Faculty of Dentistry, University of Toronto.

Dr. Imon Pal is a Researcher Associate at the Faculty of Dentistry, University of Toronto.

Dr. Anuj Aggarwal is the dentist of The One Smile Research Program, Faculty of Dentistry, University of Toronto.

Dr. Michael Glogauer is a clinician-scientist and leader in the fields of neutrophil biology, innate immunity, oral microbiome, and periodontology. He is a Fellow of the Canadian Academy of Health Sciences. Dr. Glogauer is also a Professor at the Faculty of Dentistry, University of Toronto (since 2002), Head of Dentistry for University Health Network and Chief of Dental Oncology at Princess Margaret Cancer Centre.
