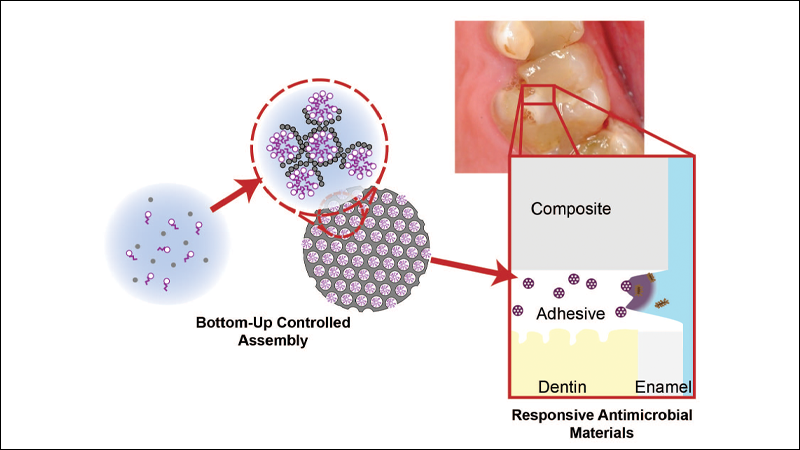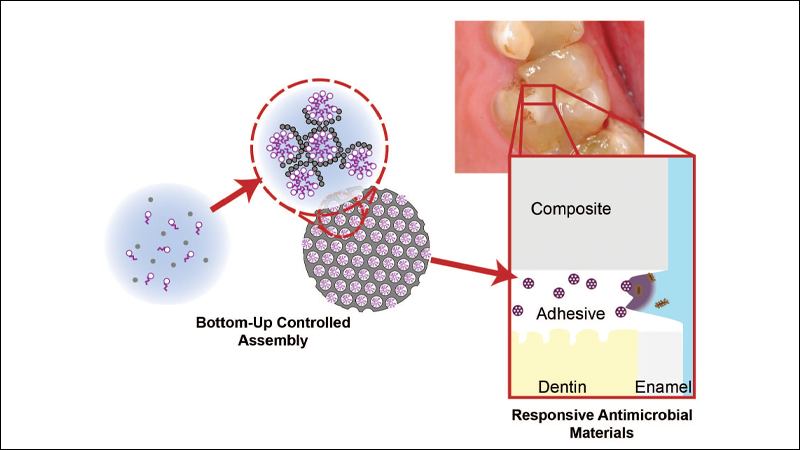
The oral cavity is one of the most challenging environments to design materials for. Awash with hostile bacteria, damaging enzymes, huge temperature swings and massive mechanical forces, creating materials that function well, look good, are easy to use, and last the patient’s entire life has always been challenging. The advent of composite resin-based restoratives and materials satisfied many of these concerns but also introduced a weak point at the connection with the tooth, where bacteria can infiltrate, proliferate, and do more damage, resulting in restoration detachment or secondary caries.
Dr. Yoav Finer’s lab at the University of Toronto has studied these damaging processes for over 20 years, and together with the laboratory spin-off company Mesosil, headed by former lab member Cameron Stewart, has developed a series of bioactive technologies and additives that let dental materials fight back against bacterial attack and allow dentists to provide true lifetime treatments. The key to Mesosil’s breakthroughs has been FDA-approved ultra-long-term antimicrobial capabilities that are integrated into the materials dentists already use: composites, adhesives, cements and other resin- or polymer-based materials. Working as a development partner and ingredient supplier for leading dental materials companies, Mesosil’s technology will be available soon across a range of dental materials to address the biological shortcomings of common treatment options.
Dental caries is ubiquitous: almost every person has or will experience dental caries in their lifetime. 35% of people have active caries at any given time, and 99% of people will get caries and live with some level of tooth restoration or treatment post-caries.1 Despite this near-universal human experience and the potentially debilitating impact on quality-of-life, our understanding of the disease and the tools we have available to treat it have been slow to evolve.
In Canada and the developed world, the most common treatment of severe caries is the removal of diseased tissue (tooth structure that has been demineralized by acid attack and partially digested by bacteria and our own bodies), and the subsequent restoration of the physical structure and function with artificial materials. This is most commonly achieved with resin composite restorative materials – a suite of resin polymer blends based on methacrylate polymerization introduced over 50 years ago, impregnated with a variety of solid fillers to provide structural support, the aesthetic appearance of natural tooth, and to adjust handling.2 Variations of this basic formula exist to fulfill a number of clinical needs: from light-cured low-viscosity flowable composites to chemically cured cements, and filler-free universal adhesives to resin-based caries-arresting medicaments. While dental materials do exist that work on drastically different principles, such as glass ionomer cements or bioceramic root canal sealers, their uses are more niche. Resin based materials have many attractive properties such as favourable handling, quick or variable curing times, mechanical robustness, and their wide variety of aesthetic options.2
In spite of resin materials’ clear benefits, the oral cavity poses one of the most extreme and demanding environments for material engineering in the human body: ever changing mechanical stresses, cycles of heat and cold shock, a complex and difficult to prepare substrate, and constant biochemical assault by acids and enzymes (Fig. 1).3 This constant challenge wears at these materials, eventually exposing weak points at their periphery that progress into secondary caries and/or mechanical failure in about 70% of cases (Fig. 2).3 This material failure and return to a disease-state requires a cycle of retreatment that often leads to escalation or tooth loss.
Fig. 1
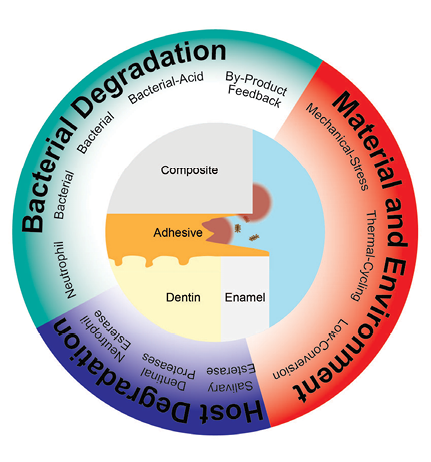
Fig. 2
![Compromised interface: A confocal scanning laser microscopy reconstructed image of an in vitro dentin-adhesive-resin composite interface, with S. mutans cells visible as small dots (green = live, yellow/red = dead) [9]. The adhesive interface has been significantly degraded, allowing bacteria to penetrate.](https://media.oralhealthgroup.com/uploads/2025/12/Stewart-Fig_2.png)
While dental materials scientists and dental manufacturers have made huge advancements in improving the materials’ clinical usability, aesthetics, and mechanical function, the restoration margins remain a persistent weak point. Polymerization shrinkage and mechanical forces pull the material away from the tooth. Bacterial, salivary, and host-immune-system enzymes degrade both the resin polymers and the exposed tooth, creating a gap large enough for cariogenic species such a Streptococcus mutans to colonize. Highly localized acid and enzyme production can start the caries formation process again, while the restoration itself erodes further.3,4 Using materials with lower polymerization shrinkage and good cavity design can reduce marginal breakdown, but the twin challenges of bacterial attack and biochemical degradation will always stress this interface.
Preventing this failure has been an active area of research for many years. Biostable polymers that retain the handling, polymerization and mechanical stability of current resins have recently made significant progress, leveraging advancements in chemistry and biomedical engineering.5 Similarly, a desire to build dental materials that can fight back against active infection or prevent it from occurring, has led to a range of new materials.3 These generally fall into two categories: 1) materials that release a component to affect bacteria, and 2) materials that rely on a permanently bound effect at their surface.
These developments mirror those in implant medicine, with the oral cavity posing distinct requirements. Pathogenic species exist in the mouth alongside critically important commensal species. As well, there is constant fluid flow carrying biological material to foul the surface and dilute any released chemicals from the material. Material development has focused on killing bacteria locally or slowing their attachment and metabolism under these conditions. This effectively reduces their acid and enzyme production and ability to initiate secondary caries.
This is the world Dr. Yoav Finer’s Faculty of Dentistry research group at the University of Toronto, in collaboration with Dr. Benjamin Hatton’s group in Materials Science and Engineering, and the commercial R&D lab spin-off company Mesosil, inhabits. With the goal of creating a technology that can stop bacteria from causing secondary caries, while preserving all the positive traits of dental materials, we undertook the development of a lifetime-lasting antimicrobial technology. Antimicrobial-release appealed to us from the outset due to its advantages over antimicrobial surfaces:
- Free antimicrobial molecules can kill bacteria not only directly on the material surface, but also within a thick accumulated biofilm.
- The material can continue to release antimicrobials and kill bacteria after fouling of the surface.
- Additive antimicrobial releasing fillers could potentially be incorporated by manufacturers without requiring modification of the polymer backbone of the dental material and across a range of materials, meaning the dentist does not experience any procedure change when using the technology.
- Antimicrobial releasing materials can allow the use of a pre-established antimicrobial drug or antiseptic with extensive evidence of efficacy and past use in dentistry.
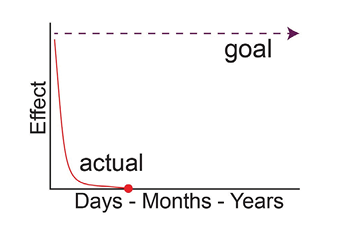
Significant challenges include targeting only the regions of interest and only pathogenic bacteria/biofilms, and ensuring the effect is sustained for the patient’s lifetime. Drug-releasing materials typically have either a very high burst-release that subsides quickly, or a very low level of extended effect (Fig. 3).
Fig. 3
Our breakthrough, to get around these challenges, was a materials science and nanotechnology-based approach to dentistry. We developed a method for the generation of silica particles containing millions of molecule-sized pores pre-loaded with an antimicrobial compound, octenidine, used in oral rinses and wound cleansers.6 The result is a silica particle with a very high payload that is tightly bound and releases very slowly relative to that payload. This is an ideal form factor for dental materials since the silica particles were similar in chemistry, handling, and physical function to the dental glasses that are used across dental composites and even in some adhesives; a small percentage of these could now be replaced with our novel particle. When these particles are loaded into a resin dental material, the polymer matrix acts as a natural diffusion barrier, slowing the release of the antimicrobial, but accelerating release when bacteria are present due to their destruction of the polymer matrix and subsequent water-uptake.7,8
After developing and characterizing the antimicrobial fillers themselves, the next step was to see if they would work in a dental material. We focused on resin adhesives as a demonstrator prototype, since they are used directly at the site of interest, at the restoration-tooth interface (Fig. 4); however, cements, flowable composites and liners, and endodontic sealers may all benefit from the antimicrobial effect. Reproducing this environment in vitro proved to be just as big a challenge as developing the particles. However, our research group’s expertise in artificial enzymes, bacterial biofilms, and drug nanocarriers/drug release modelling proved invaluable. We have been able to demonstrate through a series of pre-clinical trials that by targeting triggered antimicrobial release directly at the restoration margin (dental material-tooth interface), a very small amount of antimicrobial (nanograms) can have an outsized effect. This lets us kill or slow bacterial activity and preserve this critical region where secondary caries forms, without affecting a patient’s broader microbiome (Fig. 5).7 The small size of the marginal microgap we target means we only need a small dose of antimicrobial release at any time to reach an effective concentration, letting the slow-release antimicrobial payload last the patient’s whole life.
Fig. 4
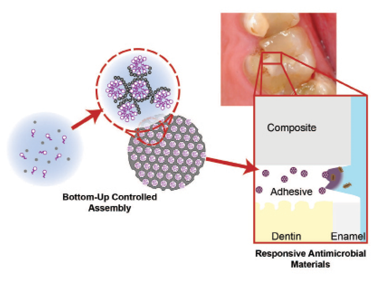
Fig. 5
![Protected Interface: A confocal scanning laser microscopy reconstructed image of an in vitro dentin-antimicrobial silica enabled adhesive-resin composite interface, with S. mutans cells visible as small dots (green = live, yellow/red = dead) [9]. Even after significant microbiological challenge the restoration interface is largely intact (red arrow), with most cells visible dead (red).](https://media.oralhealthgroup.com/uploads/2025/12/Stewart-Fig_5.png)
The antimicrobial effect from the adhesive resulted in reduced demineralization and cavitation when tested on restored extracted 3rd molars exposed to cariogenic bacteria (Fig. 6).9 More recent results have demonstrated other restorative dental materials loaded with our antimicrobial fillers have this effect. Furthermore, we have created a detailed model of antimicrobial release kinetics, letting us model these effects far into the future. This academic research culminated with a rodent secondary caries model, which demonstrated a complete inhibition of secondary caries (Fig. 7).9 Dr. Finer’s group continues to refine this model as well as expand the scope of our porous silica technology’s application into endodontic materials, caries arresting medicaments, and implant coatings.10,11
Fig. 6
![Cavitation and demineralization: A) A micro-CT cross-section of an in vitro resin composite-adhesive-dentin specimen demonstrating significant demineralization (light grey areas) at its perimeter after 1-week incubation in a cariogenic dual-species biofilm (scale bar = 1 mm). B) Quantitative analysis of specimen demineralization and cavitation from the same study as A), demonstrating the significant effects of Mesosil’s additive on secondary caries prevention [9].](https://media.oralhealthgroup.com/uploads/2025/12/Stewart-Fig_6.png)
Fig. 7
![Animal trial: A) A clinical image from our recent animal trial where rat teeth were restored with a Mesosil-enabled adhesive. B) An image of a restored rat 1st molar showing the different areas of interest. C) Quantitative International Caries Classification and Management System™ (ICCMS) scores for rat caries on restored first molars after cariogenic bacteria inoculation and 7-weeks cariogenic diet [9].](https://media.oralhealthgroup.com/uploads/2025/12/Stewart-Fig_7.png)
This is also the point at which our lab-based spin-off company, Mesosil, stepped in. As excited as we were to have developed this technology, we were quite naïve initially as to the dental industry’s operation, commercial material requirements, development costs and timelines, and the need to advocate for our technology. We were lucky that we also found a community of dedicated scientists, engineers and professionals committed to making better materials for dentists, who were very interested in bioactive and antimicrobial materials, and who were willing to share their insights into material development with us. We gathered a team of committed investors and service providers who shared our vision for bringing dental materials to the forefront of biomedical engineering innovation.
This let Mesosil begin tackling several challenges. The production of novel high-tech materials is always difficult; what worked in the lab during your PhD studies, producing only milligrams of a substance, is not commercially compatible with customers who require kilograms of the material. But our team was able to improve upon our porous particle’s original design and generate the material cheaply and easily at scale.
We also began the difficult work of creating dental materials that conformed to industrial standards. As biomedical researchers, aesthetics and shelf-life are typically not front-of-mind, but these areas are critical for use by dentists. Demonstrating conformance with industrial and regulatory expectations and improving our own technology was a fresh challenge; close collaboration with the dental industry and our researchers at the University of Toronto helped us merge the disparate fields of materials science, clinical dentistry, microbiology, and chemical engineering to fully translate our work to clinical use with regulatory approval.
Dental researchers and clinicians can create better tools to treat disease by sharing expertise to find solutions. Dentistry and dental materials have undergone several recent exciting transformations with remineralizing restoratives, and minimum intervention dentistry, that recognize the importance of preserving tooth structure. We believe that Mesosil can leverage our new technology to give dentists better tools to grow upon these improvements. Multiple products will be launching soon, powered by our technology. This is another opportunity for dentistry to lead medicine in technological development, where new solutions for infection prevention can spread to other applications that will benefit the lives of all patients. 
Oral Health welcomes this original article.
Acknowledgement: I would like to acknowledge all those who worked on the scientific research studies described and cited in this article, and my colleagues in Dr. Yoav Finer’s and Dr. Benjamin Hatton’s research groups and Mesosil Inc. The scientific research described here was funded exclusively by the National Institutes of Health [R01DE021385-0]; the Canadian Institutes of Health Research [MOP 115113, PJT-165957]; Canada Foundation for Innovation John R. Evans Leaders Fund (CFI_JELF) [project #35378 and #38794]; Ministry of Research and Innovation (MRI), Ontario Research Fund (ORF) [ORF-35378]; the Natural Sciences and Engineering Research Council of Canada (NSERC) Discovery Grants Program [RGPIN-2020-04595]; the New Frontiers in Research Fund –Exploration [NFRFE-2019-00647]; and the University of Toronto Connaught Innovation Award [IA-2018-19].
Disclaimer: The author Cameron Stewart reports a relationship with Mesosil Inc. that includes board membership, employment, and equity or stocks. Cameron Stewart has patent #WO2017197510A1 issued to Mesosil Inc. Mesosil Inc. did not contribute financially or in kind to the primary research described and cited in this article.
References
- Vilde T, Stewart CA, Finer Y. Simulating the Intraoral Aging of Dental Bonding Agents: A Narrative Review. Dentistry journal. 2022;10.
- Desai H, Stewart CA, Finer Y. Minimally Invasive Therapies for the Management of Dental Caries-A Literature Review. Dentistry journal. 2021;9.
- Stewart CA, Finer Y. Biostable, antidegradative and antimicrobial restorative systems based on host-biomaterials and microbial interactions. Dent Mater. 2019;35:36-52.
- Gitalis R, Bae JH, Preston M, Patel M, Liu Z, Sun C, et al. Human neutrophils compromise the restoration-tooth interface. Acta Biomaterialia. 2020.
- Gouveia Z, Finer Y, Santerre JP. Towards the development of biostable dental resin systems – design criteria and constraints beyond ester-free chemistries. Dental Materials. 2022;38:1827-40.
- Stewart CA, Finer Y, Hatton BD. Drug self-assembly for synthesis of highly-loaded antimicrobial drug-silica particles. Scientific Reports. 2018;8:895.
- Stewart CA, Hong JH, Hatton BD, Finer Y. Responsive antimicrobial dental adhesive based on drug-silica co-assembled particles. Acta Biomater. 2018;76:283-94.
- Stewart CA, Hong JH, Hatton BD, Finer Y. Antimicrobial antidegradative dental adhesive preserves restoration-tooth bond. Dental Materials. 2020.
- Stewart CA, Ngai K, Gouveia Z, Rao S, Abuquteish D, Mandelis A, et al. Evaluation of a long-term antimicrobial dental adhesive via in vitro biodegradation and in vivo rodent secondary caries models. Dental Materials. 2025.
- Stewart C, Siu A, Tsui C, Finer Y, Hatton B. Rapid synthesis of drug-encapsulated films by evaporation-induced self-assembly for highly-controlled drug release from biomaterial surfaces. Journal of Materials Chemistry B. 2022;10:6453-63.
- Marashdeh MQ, Stewart CA, Kishen A, Levesque C, Finer Y. Assessment of Root Canal Sealers Loaded with Drug-Silica Coassembled Particles Using an In Vitro Tooth Model. Journal of endodontics. 2021;47:1775-82.
About the author

Dr. Cameron Stewart is founder and CEO of Mesosil, a lab-based startup company, developing and manufacturing anti-infective materials for dental and healthcare applications. He is a former PhD student and post-doctoral fellow at the Faculty of Dentistry, University of Toronto, in Dr. Yoav Finer’s Dental Biomaterials research group. He obtained his PhD from the University of Toronto in Biomedical Engineering and holds a BASc in Nanotechnology Engineering from the University of Waterloo.