
Oral dysbiosis, a key driver of many oral diseases that impact over three billion people worldwide, is the shift away from health-associated homeostatic microbial populations that inhabit the oral cavity.1-3 Indeed, the oral microbial consortia associated with dental health is characteristic of having high diversity, both in genetic composition and in metabolic capability.4-7 Antibiotics, chronic antiseptic exposure, iatrogenic immunosuppression, medical interventions that disrupt the mucocutaneous or gingival barriers, daily consumption of preservatives and highly processed ingredients in the diet, and stress, can rapidly narrow the diversity of the oral microbiome. This shift favors keystone pathogens or pathobionts that change the oral environment and modulate the immune system to evade clearance, modify tissue and bone, lead to ligament and bone loss, and increase the risk for cancers of the oral cavity.15-20
It is widely appreciated that individuals have their own oral microbial fingerprint, wherein, diet, genetics, disease status and lifestyle all play an influential role in guiding the composition of the oral microbiome.8-11 This, and the broadening availability of commercial salivary tests, enables the dental professional to be at the forefront of personalized dentistry.12 However, the identification of a patient with Candida spp., S. mutans, or Red Complex overgrowth informs the practitioner that there is a problem, but the results are not actionable beyond continuing to remind the patient of regular brushing, flossing, professional cleanings, and diet modification away from processed foods and sugar. There has been little innovation in solutions that make a significant difference, until recently.
I was thrust into resolving this problem in 2012, when my grandmother suffered a stroke shortly after a tooth extraction. As a microbiologist and immunologist, I postulated the possible mechanism giving rise to her stroke. The gaping wound created in her mouth when the tooth was extracted may have allowed millions of her oral microbes to gain direct access to her bloodstream, giving her transient bacteremia. This potentially caused the triggering of a massive cytokine cascade, resulting in the activation of a clotting cascade to bind and neutralize the bacteria in her bloodstream. A thrombo-embolic event occurred, and one or more clot(s) circulating in her blood made its way to her brain, lodging in a vessel and cutting off circulation to a rather large section of her prefrontal cortex.13-14 I was concerned that those bacteria were likely very dangerous as testing showed high levels of Red Complex bacteria in her mouth.
Looking into her options for daily oral hygiene, post-stroke, I was dismayed at the lack of good options. Suggestions were either too toxic to take daily or were completely ineffective at selectively modulating her oral microbiome.
I took the matter into my own hands. I cultured her oral microbes and performed a lot of in vitro screening with her bacteria to identify naturally safe ingredients to feed the good microbes and steer their metabolism to not secrete any more damaging effectors that can lead to tooth loss. I stumbled upon a combination of ingredients that did just that. In fact, this same combination also leads to the reduction of Red Complex bacterial activity and burdens, as well as sugar-mediated cariogenic pathogen growth.
Briefly, we found a combination of ingredients that: (i) blocked universal uptake and processing of sugar (and sugar alcohols) by bacteria and yeasts, whilst simultaneously (ii) stimulating beneficial microbes, that naturally inhabit the mouth, to start metabolizing proteins in saliva. The combination of these two metabolic shifts transforms not only the microbial population in the mouth but also the metabolites that these little metabolic factories make on a daily basis. Basically, instead of microbes producing acids when they metabolize our dietary sugars, the ingredients are forcing the microbes to sugar-starve and switch to eating the proteins in our saliva and crevicular fluid; the metabolites being produced from this are nitric oxide, nitrate, nitrite and urea.
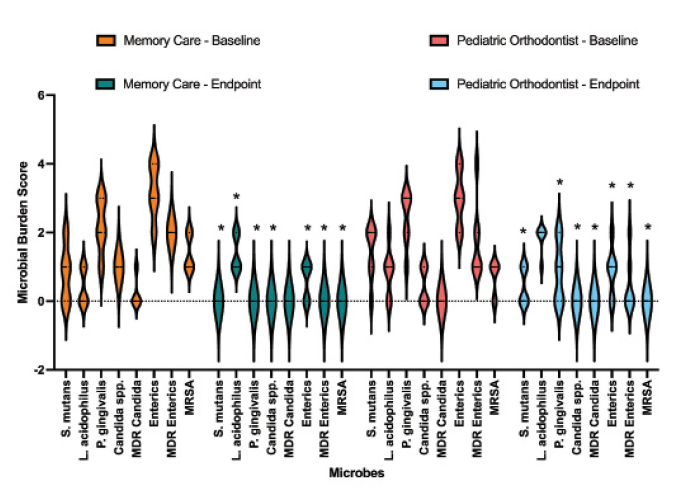
Over the past decade, these naturally safe ingredients have been formulated as PTx800, or PTx325, and have been evaluated in numerous clinical case studies, randomized and double-blinded controlled clinical studies examining clinical efficacy on gingivitis, periodontal disease, halitosis, and in oral and gut dysbiosis cases. Studies have included companion and zoo animals, pediatric orthodontic patients, memory care patients, older adults with gingivitis, public health populations of 500+ subjects between 18-80 years, and even a study up in the International Space Station, sponsored by the European Space Agency. Some study participants are presented in Figure 1.
Fig. 1A

Fig. 1B

Fig. 1C

Here are a few things we have learned when the oral microbiome is blocked from sugar metabolism while simultaneously activated to metabolize protein:
Finding #1 – The oral microbiome shifts composition very quickly. Data show that a significant shift can be observed by salivary testing within days (depending upon the study timepoints). In canine periodontal disease studies, we observed a shift in 5 days, with Porphyromonas spp. burdens reducing by over 50% in all animals tested (Fig. 2) and gingival tissue inflammation reducing in 14 and 30 days (Fig. 3).
Fig. 2A
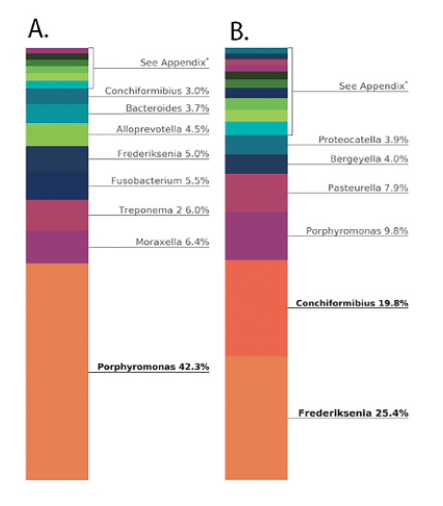
Fig. 2B
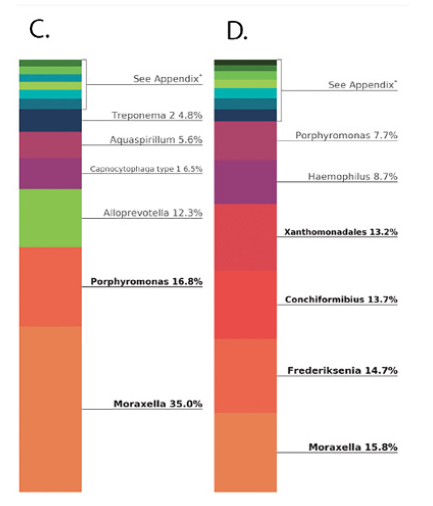
Fig. 3A
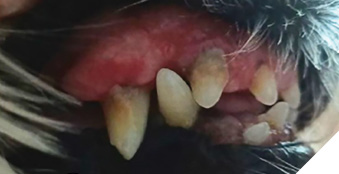
Fig. 3B
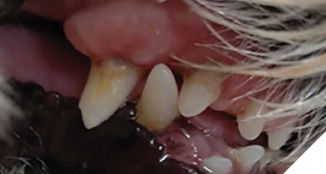
Fig. 3C
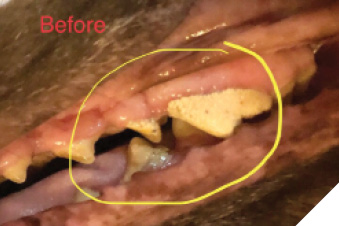
Fig. 3D
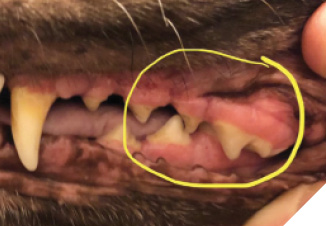
Finding #2 – The plaque and tartar burdens change within 30 to 45 days of daily administration in canines and humans with gingivitis and/or periodontal disease (Fig. 3 and Fig. 6).
Finding #3 – The subgingival anaerobic microbial burdens significantly shift when animals and humans’ oral microbiome shifts from primarily sugar fermentation to protein metabolism (Fig. 5 & 7).
Finding #4 – The amount of bad breath-associated volatile aromatic compounds significantly reduces (Fig. 4).
Fig. 4
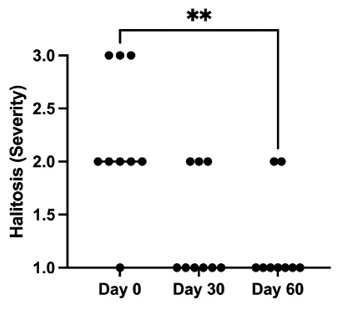
Fig. 5
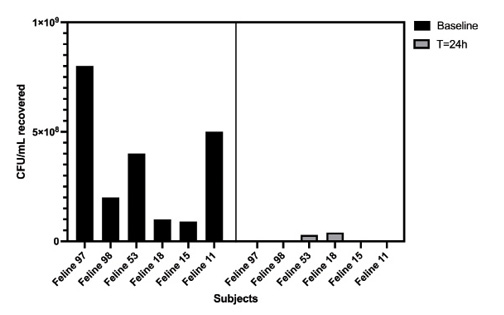
Fig. 6

Fig. 7
Finding #5 – Salivary acids and cariogenic pathogen-acid production, S. mutans growth and subgingival plaque are blocked for a prolonged period of time, even in the presence of sugar, and that can be reflected below the gumline in a manner that is differentiated from sugar alcohols, as well (Fig. 6).
Taken together, these and many other data gathered over the past 13 years suggest that a novel means of microbial control can be driven through the modulation of the oral microbiome using metabolic influencers and that this can have clinical benefit in diseased patients. Indeed, a growing amount of academic research points to the importance of microbial metabolites that play a role in cariogenesis, mucosal inflammation, periodontal disease, and, most recently, carcinogenesis.21-24
The findings from our studies highlight the potential of leveraging naturally safe ingredients to influence the oral microbiome, reducing levels of certain bacteria and promoting a healthier microbial balance. The significant shifts observed in both subgingival microbial composition and overall oral health markers suggest that targeting microbial metabolism can be an effective strategy for supporting better oral health outcomes. By modulating the microbiome to decrease sugar metabolism while promoting protein utilization, we observed not only a reduction in the presence of Porphyromonas species but also improvements in gingival health, plaque, and tartar levels.
In canines, the use of PTx325 and PTx800 resulted in a reduction of volatile organic compounds associated with bad breath, as well as improved oral microbiome diversity. These outcomes demonstrate the potential of microbiome modulation in addressing common dental concerns, such as gingivitis, plaque buildup, and halitosis, without relying on traditional methods like antibiotics or harsh antiseptics.
The ability to support reductions in plaque and tartar through microbial management alone, without mechanical interventions like toothbrushing, suggests benefits for populations with limited access to dental care or those who may face challenges performing routine oral hygiene, such as the elderly, people with disabilities, or individuals with cognitive decline. Clinical case studies and trials involving companion animals, pediatric orthodontic patients, and memory care populations highlight the broad applicability of these formulations across various patient demographics.
Our data also underscore the rapid response of the microbiome to intervention, with significant microbial shifts occurring within days of treatment. This timely modulation may be valuable in supporting oral health for at-risk populations and minimizing the need for more invasive dental procedures.
The results from these studies suggest that PTx325 and PTx800, same composition with animal and human-grades respectively, provide effective means to modulate the oral microbiome, contributing to better oral health outcomes. By encouraging a shift in the metabolic pathways of oral bacteria from sugar fermentation to protein metabolism, these formulations support a reduction in certain bacterial loads and improvements in oral health markers such as gingival condition, breath quality, and plaque levels. The ability to achieve these results through naturally safe ingredients offers a promising approach to oral health care, particularly for populations with limited access to traditional dental interventions.
Future studies will aim to refine our understanding of how these products interact with the oral microbiome and evaluate their role in supporting oral health for individuals with different backgrounds, including those managing various health conditions. 
Oral Health welcomes this original article.
About the Author

Emily Stein, Ph.D. is CEO of Primal Health, LLC. She completed a Fellowship in the Department of Rheumatology and Immunology at Stanford University School of Medicine. She holds a PhD in Microbial Biology from the University of California-Berkeley and a B.S. in Microbiology and Immunology from the University of Iowa.
