
Hypochlorous acid, nitric oxide, sulfur, uric acid, and heme in oral redox signaling
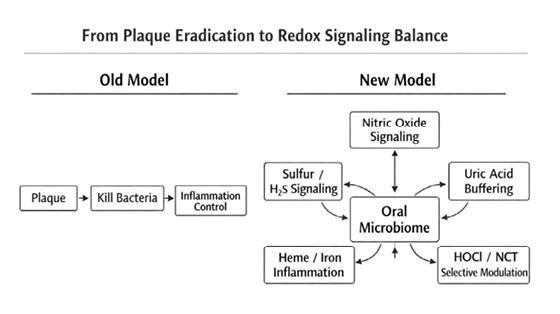
The oral cavity is increasingly recognized as a critical interface between microbial ecology, host immunity, and systemic health. While dentistry has traditionally focused on mechanical plaque control and antimicrobial suppression, emerging evidence suggests that oral health is governed less by eradication and more by biochemical signaling balance.
Previous discussions have highlighted the relationship between oral nitrate-reducing bacteria and nitric oxide (NO) bioavailability, and how indiscriminate antiseptic use may impair this pathway. However, a third signaling system — sulfur metabolism — has received comparatively little attention in dentistry, despite its central role in halitosis, periodontal tissue injury, and microbial dysbiosis.1,2
This article proposes a novel multi-axis signaling model of oral health involving at least:
- Oxidative signaling (hypochlorous acid; HOCl)
- Nitrogen signaling (nitrate ➞ nitrite ➞ nitric oxide)
- Sulfur signaling (hydrogen sulfide and volatile sulfur compounds)
An additional, clinically relevant modifier of this redox signaling triad is uric acid (urate), the most abundant low–molecular weight antioxidant in human saliva. Unlike plasma urate, which is often elevated in metabolic disease, salivary urate appears to function primarily as a local redox buffer, scavenging reactive species generated during host–microbe interactions. Emerging evidence suggests that salivary urate levels are reduced in periodontitis and inversely associated with disease severity, consistent with increased oxidative demand rather than diminished production. This local depletion may reflect consumption of urate during active inflammation, further supporting the concept that periodontal disease represents a state of redox imbalance rather than microbial burden alone.17,18
Fig. 1
Vagal tone and systemic inflammatory control
An often-overlooked component of oral–systemic health is the intimate neuro-immune connection between the oral cavity and the body’s primary anti-inflammatory control system: the vagus nerve. The posterior tongue, soft palate, tonsillar pillars, and pharyngeal tissues are densely innervated by vagal afferents that continuously relay immune status from the oral environment to the brainstem. Persistent exposure to lipopolysaccharides (LPS), proteolytic enzymes such as gingipains, volatile sulfur compounds, and oxidative metabolites within dysbiotic oral biofilms may chronically stimulate toll-like receptors (TLRs) at this interface, sending sustained inflammatory signals centrally and impairing the cholinergic anti-inflammatory pathway — the mechanism by which vagal signaling regulates cytokine production. Restoration of oral redox balance and microbial ecology therefore has implications beyond local periodontal health. By reducing inflammatory bioburden while preserving commensal nitrate-reducing species, hypochlorous acid (HOCl) and its downstream derivative N-chlorotaurine (NCT) may help normalize this vagal feedback loop, permitting appropriate cytokine regulation and systemic inflammatory control. This perspective reframes oral biofilm management not merely as antimicrobial therapy, but as modulation of a critical neuro-immune gateway influencing whole-body inflammatory tone.
Understanding how these systems interact might provide a unifying framework for periodontal inflammation, halitosis, and emerging diet-related oral changes.
Hypochlorous acid: Physiologic oxidation, not just chemical disinfection
Hypochlorous acid is endogenously produced by neutrophils via the myeloperoxidase (MPO) pathway as part of innate immunity. 3,4 At physiologic concentrations, HOCl functions as a redox signaling molecule, not merely as a microbicidal agent. To note: most literature considers it primarily as a microbial agent.
HOCl:
- Disrupts biofilm architecture
- Suppresses pathogenic anaerobes
- Modulates microbial gene expression
- Preserves epithelial barrier integrity
Importantly, HOCl is rapidly neutralized after action, leaving no persistent residue. This differentiates it from conventional antiseptics such as chlorhexidine or alcohol, which bind oral tissues, alter pellicle structure, and exert prolonged non-selective antimicrobial pressure. Of note: during active periodontitis, neutrophil-derived HOCl has been considered a marker of tissue damage, not homeostasis.
Considering HOCl as selective, signaling-restoring molecule rather than antiseptic:
HOCl ↔ Nitric Oxide (NO) ↔ nitrate-reducing bacteria
- HOCl ↔ Uric acid ↔ metabolic inflammation
- HOCl ↔ TLR (Toll-like receptor) hijacking ↔ neuroinflammation
- HOCl ↔ oral microbiome ↔ systemic disease
- HOCl as selective, signaling-restoring molecule rather than antiseptic
Uric Acid (Urate)
From a biochemical perspective, urate represents a major salivary redox sink and is a known target of oxidation by reactive species, including hypochlorous acid. HOCl rapidly oxidizes uric acid to allantoin and related products, a reaction that has been proposed as a mechanism by which oxidative inflammatory activity can lower measurable salivary urate concentrations. This provides a plausible biochemical link between neutrophil activation, HOCl chemistry, and the observed reduction of salivary urate in periodontal disease — supporting the interpretation of urate depletion as a marker of excessive local oxidant flux rather than a pathogenic driver itself.19
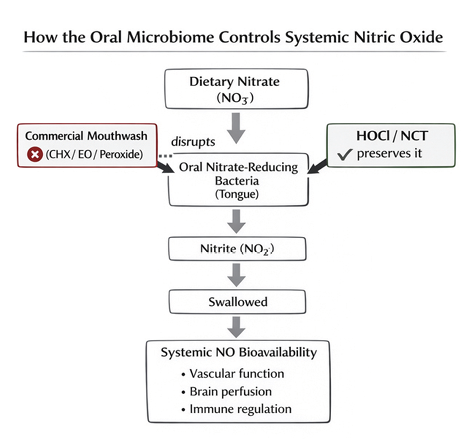
The nitrate–nitric oxide pathway: A beneficial microbial function
A healthy oral microbiome supports systemic nitric oxide production via dietary nitrate reduction. Commensal proteolytic bacteria such as Neisseria, Rothia, and Actinomyces reduce nitrate to nitrite, which is further converted to NO in the oral cavity and gastrointestinal tract.5–7 It should be noted that some periodontal pathogens (Fusobacterium) actually possess nitrate-reducing capacity too.
Nitric oxide:
- Regulates vascular tone
- Enhances mitochondrial efficiency
- Supports epithelial repair
- Suppresses periodontal pathogens
Clinical studies demonstrate that antiseptic mouthwashes can reduce salivary nitrite and impair systemic NO bioavailability, with measurable cardiovascular consequences. This highlights the importance of preserving functional microbial pathways, not simply reducing bacterial load.
Fig. 2
Sulfur signaling: From physiologic gasotransmitter to local toxin
Sulfur metabolism represents a double-edged sword in oral biology.
At low concentrations, hydrogen sulfide (H₂S) functions as a gasotransmitter, contributing to redox balance and cellular signaling. However, in dysbiotic states, sulfur-reducing bacteria metabolize sulfur-containing amino acids into excessive levels of volatile sulfur compounds (VSCs), including H₂S and methyl mercaptan.10–12
Key oral pathogens involved include:
- Porphyromonas gingivalis
- Fusobacterium nucleatum
- Treponema denticola
- Prevotella intermedia
Elevated local H₂S concentrations have been shown to:13
- Inhibit cytochrome c oxidase
- Impair mitochondrial respiration
- Induce gingival fibroblast apoptosis
- Increase epithelial permeability
- Promote collagen degradation
This provides a mechanistic explanation for the long-observed association between halitosis, periodontal inflammation, and tissue breakdown.14
Fig. 3

The heme–iron redox axis: An under-recognized driver of periodontal ecology
A further, under-recognized signaling system in periodontal ecology involves heme and iron metabolism. Several key periodontal pathogens — including Porphyromonas gingivalis, Fusobacterium nucleatum, Prevotella intermedia, and Treponema denticola — are heme-dependent organisms that actively scavenge iron-porphyrin complexes from inflamed gingival tissues.4,9
This phenomenon helps explain long-recognized clinical observations in periodontology: bleeding gums, dark pigmentation within plaque and calculus, and the strong association between inflammation and anaerobic dominance. These are not merely coincidental findings but reflections of a heme-driven microbial ecology.
Free heme and iron are not biologically inert. They catalyze Fenton chemistry, generate hydroxyl radicals, amplify oxidative stress, impair epithelial and mitochondrial function, and create conditions that favor obligate anaerobes and sulfur-reducing bacteria. In this way, heme availability acts as a redox amplifier within the periodontal pocket, reinforcing the dysbiotic shift.
From a biochemical perspective, hypochlorous acid is known to oxidize heme groups and disrupt iron-porphyrin chemistry. Neutrophil-derived HOCl can inactivate heme-containing enzymes, modify porphyrin rings, and limit iron-driven radical propagation. This suggests that HOCl may modulate not only sulfur and nitrogen signaling but also this heme-iron redox pathway within periodontal tissues.
Recognition of this axis further supports the concept that periodontal disease represents a self-reinforcing redox environment, where bleeding, microbial metabolism, oxidative stress, and anaerobic selection feed into one another.
Fig. 4
Diet, protein fermentation, and revealed dysbiosis
High-protein or sulfur-rich diets — including meat-dominant diets — do not inherently cause periodontal disease. Rather, they increase substrate availability for microbial metabolism.1,15
In a balanced oral ecosystem:
- Sulfur metabolism remains within physiologic signaling range
- Nitrate-reducing bacteria dominate
- NO signaling prevails
In dysbiosis:
- Proteolytic anaerobes proliferate
- Sulfur metabolism shifts toward toxicity
- VSC production increases
- Inflammation accelerates
Thus, dietary patterns may reveal underlying microbial imbalance, rather than create pathology de novo.
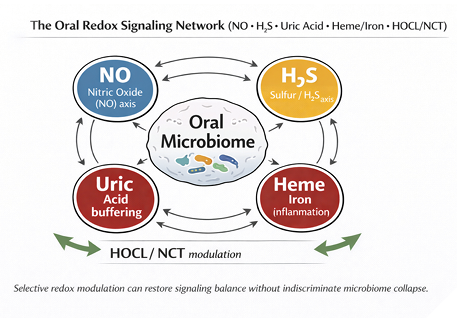
HOCl hypothesized as a redox gatekeeper between nitrogen and sulfur pathways
Hypochlorous acid occupies a unique regulatory position between nitrogen and sulfur signaling.
HOCl:
HOCl is also known to oxidize heme and disrupt iron-porphyrin chemistry, potentially limiting the heme-driven redox amplification that favors anaerobic periodontal pathogens.
- Neutralizes excess H₂S locally
- Suppresses sulfur-reducing anaerobes
- Preserves nitrate-reducing commensals
- Raises local redox potential, discouraging anaerobic dominance
This allows the oral cavity to shift away from sulfur-driven toxicity and toward nitric-oxide–mediated homeostasis. Unlike traditional antiseptics, HOCl achieves this without collapsing microbial diversity or impairing beneficial signaling pathways in the long term. Although the HOCL is not selective as an antiseptic, the NCT that persists long after HOCL is gone, allows for the selective claim as the commensal bacteria population tends to bounce back more readily.
- There are no studies demonstrating that endogenous oral HOCl levels are sufficient to neutralize pathologic H₂S concentrations in periodontal pockets
- Similarly, we found no data on the spatial and temporal distribution of HOCl vs H₂S in periodontal tissues
- To note, the reaction generates polysulfides, which themselves have biological activity
While the HOCl-H₂S reaction occurs rapidly in vitro, its regulatory role in vivo remains speculative. This would have to be confirmed to support the given hypothesis.
Clinical implications for periodontal therapy
Systemic metabolic factors may further modulate this oral redox environment. Hyperuricemia has been shown to impair endothelial nitric oxide bioavailability, in part through inhibition of endothelial nitric oxide synthase activity and increased oxidative stress. In patients with metabolic syndrome or elevated serum urate, diminished NO signaling may compound local oral effects, including reduced microvascular perfusion and delayed epithelial repair. This systemic–local interaction reinforces the concept that periodontal inflammation cannot be fully understood without considering broader redox and metabolic context.20
This signaling-based framework suggests several important clinical considerations:
- Periodontal disease may reflect redox imbalance, not bacterial quantity alone
- Halitosis could be a marker of pathologic sulfur metabolism
- Long-term antiseptic use may worsen dysbiosis by disrupting NO pathways
- Redox-modulating strategies may support healing without resistance or rebound effects
Rather than viewing plaque as an enemy to be eradicated, clinicians may benefit from considering how therapeutic interventions influence microbial function and host signaling.15,16
Conclusion
I have introduced a hypothesis based on clinical observation and supported by scientific evidence.
Oral health emerges from the dynamic balance of oxidative, nitrogen, and sulfur signaling. Hypochlorous acid, nitric oxide, and hydrogen sulfide are not inherently harmful or beneficial; their effects depend on concentration, localization, and microbial context.
Recognizing the oral cavity as a biochemical signaling environment rather than a sterile field opens new avenues for understanding periodontal disease, halitosis, and host–microbiome interactions. This framework aligns dentistry more closely with immunology, microbiology, and systems biology — and may help explain why physiologic approaches often succeed where aggressive antimicrobial strategies fail. 
Oral Health welcomes this original article.
Disclosure: The author has a financial interest in DentalRebel Inc., which produces and sells products including HOCL which is mentioned in this article.
References
- Winter SE, Lopez CA, Bäumler AJ. The dynamics of gut-associated microbial communities during inflammation. Nat Rev Microbiol. 2013;11(9):593–603.
- Lundberg JO, Weitzberg E. Biology of nitrogen oxides in the gastrointestinal tract. Gut. 2013;62(4):616–629.
- Thomas EL, Jefferson MM, Grisham MB. Myeloperoxidase-catalyzed oxidation of chloride and other halides. Free Radic Biol Med. 1982;3(3):139–146.
- Hawkins CL, Davies MJ. Hypochlorous acid-induced damage to proteins. Free Radic Biol Med. 1998;25(7):766–778.
- Kapil V, Haydar SM, Pearl V, Lundberg JO, Weitzberg E, Ahluwalia A. Physiological role for nitrate-reducing oral bacteria in blood pressure control. Free Radic Biol Med. 2013;55:93–100.
- Hyde ER, Andrade F, Vaksman Z, et al. Metagenomic analysis of nitrate-reducing bacteria in the oral cavity. PLoS One. 2014;9(3):e88645.
- Carey RM, Kapoor S, Wakabayashi H, et al. Role of oral bacteria in nitric oxide bioavailability. Hypertension. 2020;76(3):740–749.
- Bescos R, Ashworth A, Cutler C, et al. Effects of chlorhexidine mouthwash on the oral microbiome. Sci Rep. 2020;10:5254.
- Kapil V, Haydar SM, Lundberg JO, Weitzberg E, Ahluwalia A. Oral nitrate reduction and blood pressure. Free Radic Biol Med. 2013;55:93–100.
- Persson S, Edlund MB, Claesson R, Carlsson J. The formation of hydrogen sulfide and methyl mercaptan by oral bacteria. Oral Microbiol Immunol. 1990;5(4):195–201.
- Persson S, Claesson R, Carlsson J. The capacity of subgingival microbiotas to produce volatile sulfur compounds. Oral Microbiol Immunol. 1989;4(4):169–172.
- Söderling E, Meurman JH, Tengvall P. Volatile sulfur compounds in relation to oral health status. J Periodontol. 1989;60(10):527–531.
- Takeuchi H, Setoguchi T, Machigashira M, et al. Hydrogen sulfide inhibits mitochondrial respiration in gingival fibroblasts. J Periodontal Res. 2008;43(4):432–438.
- Ratcliff PA, Johnson PW. The relationship between oral malodour, gingivitis, and periodontitis. J Periodontol. 1999;70(5):485–489.
- Marsh PD. Microbial ecology of dental plaque and its significance in health and disease. Adv Dent Res. 1994;8(2):263–271.
- Rosier BT, De Jager M, Zaura E, Krom BP. Historical and contemporary hypotheses on oral disease development. Front Cell Infect Microbiol. 2014;4:92.
- Ye L, et al. Association between periodontitis and uric acid levels in blood, saliva, and gingival crevicular fluid: a systematic review and meta-analysis. Clin Oral Investig. 2023;27
- Lorente L, et al. Low salivary uric acid levels are independently associated with periodontitis severity. J Clin Periodontol. 2025
- Yardim-Akaydin S, et al. Reaction of uric acid with oxidants such as hypochlorous acid: formation of allantoin as a marker of oxidative stress. Free Radic Biol Med. 2004;36(5)
- Park JH, et al. Uric acid attenuates nitric oxide production by decreasing endothelial nitric oxide synthase activity. Hypertension. 2013;61(5)
- Tracey KJ. The inflammatory reflex. Nature. 2002;420:853-859.
- Pavlov VA, Tracey KJ. Neural regulation of immunity: molecular mechanisms and clinical translation. Nat Neurosci. 2017;20:156-166.
- Wang H, Yu M, Ochani M, et al. Nicotinic acetylcholine receptor α7 subunit is an essential regulator of inflammation. Nature. 2003;421:384-388.
- Breit S, Kupferberg A, Rogler G, Hasler G. Vagus nerve as modulator of the brain–gut axis. Front Psychiatry. 2018;9:44.
- Bonaz B, Sinniger V, Pellissier S. The vagus nerve in the neuro-immune axis: implications in inflammatory bowel disease. Front Immunol. 2017;8:1452.
- Olsen I, Singhrao SK. Oral infection, inflammation, and systemic diseases: role of Porphyromonas gingivalis. J Oral Microbiol. 2015;7:27598.
- Hajishengallis G. Periodontitis: from microbial immune subversion to systemic inflammation. Nat Rev Immunol. 2015;15:30-44.
About the author

Janice Goodman is on the Oral Health Editorial Board and practices general dentistry in downtown Toronto. She is passionate about disseminating knowledge on the significance of the recent emergence and availability of Hypochlorous acid in a stable form and can be reached at jangoodman@rogers.com.
